Abstract
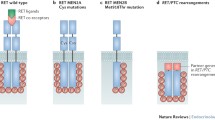
Rearrangements of the protooncogene RET (RET/PTC) and somatic mutations of the gene BRAF are the most common events in the etiopathogenesis of papillary thyroid carcinoma (PTC). The rates of RET/PTC rearrangements and BRAF mutations in different nodular formations of the thyroid gland (TG) have been estimated. Comparative expression analysis of the extracellular (EC) and tyrosine kinase (TK) domains of RET has shown that 14% (12 out of 85) of PTC cases are RET/PTC-positive, including one ΔRFP/RET-, two RET/PTC3-, and seven RET/PTC1-positive tumors, as well as two unidentified chimeric RET/PTC oncogenes. The standard T1796A transversion in the gene BRAF has been found in 60% (55 out of 91) PTC cases with the use of amplification refractory mutation system–polymerase chain reaction (ARMS–PCR). Somatic mutation G1753A and deletion del1800_1811 have been identified in PTC for the first time. The absence of the BRAF mutations in RET/PTC-positive tumors allows these two genetic defects to be regarded as alternative mechanisms of the RAS–RAF–MEK–ERK mitogen-activated protein (MAP) kinase cascade activation in PTCs. In none of the three follicular thyroid carcinomas (FTCs), 11 follicular thyroid adenomas (FTAs), and 13 nodular goiters have either BRAF mutations or RET/PTC rearrangements been found. Thus, the RET/PTC chimeric oncogenes and BRAF somatic mutations are specific markers of PTC and can be used for the preoperative diagnosis of these tumors.
Similar content being viewed by others
REFERENCES
Robbins J. 1991. Thyroid cancer, a lethal endocrine neoplasm. Ann. Intern. Med. 115, 133–147.
Farid N.R., Shi Y., Zou M. 1994. Molecular basis of thyroid cancer. Endocr. Rev. 15, 202–232.
Learoyd D.L., Messina M., Zedenius J., Robinson B.G. 2000. Molecular genetics of thyroid tumors and surgical decision-making. World J. Surg. 24, 923–933.
Goretzki P.E., Dotzenrath C., Simon D., Roher H.-D. 1999. Studies of oncogenes and tumor-suppressor genes in human thyroid carcinomas, and their clinical implications. Langenbeck's Arch. Surg. 384, 1–8.
Sarlis N.J. 2000. Expression patterns of cellular growthcontrolling genes in non-medullary thyroid cancer: Basic aspects. Rev. Endocr. Metab. Disorders. 1, 183-196.
Komminoth P. 1997. The RET proto-oncogene in medullary and papillary thyroid carcinoma. Virchows Arch. 431, 1–9.
Nakamura T., Ishizaka Y., Nagao M., Ishikawa T. 1994. Expression of the ret proto-oncogene product in human normal and neoplastic tissues of neural crest origin.J. Pathol. 172, 255–260.
Baloh R.H., Enomoto H., Johnson E.M., Milbrandt J. 2000.The GDNF family ligands and receptors: Implications for neural development. Curr. Opin. Neurobiol. 10, 103–110.
Eng C. 1999. RET proto-oncogene in the development of human cancer.J. Clin. Oncol. 17, 380–393.
Grieco M., Santoro M., Berlingieri M.T., Melillo R.M., Donghi R., Bongarzone I., Pierotti M.A., Dell Porta G., Fusco A., Vecchio G. 1990. PTC is a novel rearranged form of ret proto-oncogene and frequently detected in vivo in human thyroid papillary carcinomas. Cell. 60, 557–563.
Santoro M., Carlomagno F., Hay I.D., Herrmann M.A., Grieco M., Melillo R., Pierotti M.A., Bongarzone I., Della Porta G., Berger N., Peix, J.L., Paulin C., Fabien N., Vecchio G., Jenkins R.B., Fusco A. 1992. RET oncogene activation in thyroid neoplasms is restricted to the papillary cancer subtype. J. Clin. Invest.89, 1517–1522.
Tallini G., Asa S.L. 2001. RET oncogene activation in papillary thyroid carcinoma.Adv. Anat. Pathol.8, 345-354.
Pierotti M.A., Santoro M., Jenkins R.B., Sozzi G., Bongarzone I., Grieco M., Monzini N., Miozzo M., Herrmann M.A., Fusco A., Hay I.D., Della Porta G., Vecchio G. 1992. Characterization of an inversion on the long arm of chromosome 10 juxtaposes D10S170 and ret and creating the oncogenic sequence Ret/PTC. Proc.Natl. Acad. Sci. USA. 89, 1616–1620.
Bongarzone I., Butti M.G., Coronelli S., Borrello M.G., Santoro M., Mondellini P., Pilotti S., Fusco A., Della Porta G., Pierotti M.A. 1994. Frequent activation of ret protooncogene by fusion with a new activating gene in papillary thyroid carcinomas. Cancer Res.54, 2979-2985.
Bongarzone I., Monzini N., Borrello M.G., Carcano C., Ferraresi G., Arighi E., Mondellini P., Della Porta G., Pierotti M.A. 1993. Molecular characterization of a thyroid tumor-specific transforming sequence formed by the fusion of Ret tyrosine kinase and the regulatory subunit RI a of cyclic AMP-dependent protein kinase A.Mol. Cell Biol. 13, 358–366. _
Tuttle R.M., Becker D.V. 2000. The Chernobyl and its consequences: Update at the millennium. Semin. Nucl.Med. 30, 133–140.
Greco A., Pierotti M.A., Bongarzone I., Pagliardini S., Lanzi C., Della Porta G. 1992. TRK-T1 is a novel oncogene formed by the fusion of TPR and TRK genes in human papillary thyroid carcinomas. Oncogene. 7, 237-242.
Greco A., Mariani C., Miranda C., Lupas A., Pagliardini S., Pomati M., Pierotti M.A. 1995. The DNA rearrangement that generates the TRK-T3 oncogene involves a novel gene on chromosome 3 whose product has a potential coiled-coil domain. Mol. Cell. Biol. 15, 6118–6127.
Pearson G., Robinson F., Gibson T.B., Xu B.-E., Karandikar M., Berman K., Cobb M.H. 2001. Mitogenactivated protein (MAP) kinase pathways: Regulation and physiological functions. Endocr. Rev. 22, 153–183.
Suarez H.G., du Villard J.A., Severino M., Caillou B., Schlumberger M., Tubiana M., Parmentier C., Monier R. 1990. Presence of mutations of all three ras genes in human thyroid tumors. Oncogene.5, 565–570.
Karga H., Lee J.-K., Vickery A.L., Thor A., Gaz R.D., Jameson J.L. 1991. Ras oncogene mutations in benign and malignant thyroid tumors. J. Clin. Endocr. Metab. 73, 832–836.
Rapp U.R., Goldsborough M.D., Mark G.E., Bonner T.I., Groffen J., Reynolds Jr. F.H., Stephenson J.R. 1983. Structure and biological activity of v-raf, a unique oncogene transduced by a retrovirus. Proc.Natl. Acad. Sci. USA. 80, 4218–4222.
Jansen H.W., Lurz R., Bister K., Bonner T.I., Mark G.E., Rapp U.R. 1984. Homologous cell-derived oncogenes in avian carcinoma virus MH2 and murine sarcoma virus 3611. Nature. 307, 281–284.
Davies H., Bignell G.R., Cox C., Stephens P., Edkins S., Clegg S., Teague J., Woffendin H., Garnett M.J., Bottomley W., Davis N., Dicks E., Ewing R., Floyd Y., Gray K., Hall S., Hawes R., Hughes J., Kosmidou V., Menzies A., Mould C., Parker A., Stevens C., Watt S., Hooper S., Wilson R., Jayatilake H., Gusterson B.A., Cooper C., Shipley J., Hargrave D., Pritchard-Jones K., Maitland N., Chenevix-Trench G., Riggins G.J., Bigner D.D., Palmieri G., Cossu A., Flanagan A., Nicholson A., Ho J.W., Leung S.Y., Yuen S.T., Weber B.L., Seigler H.F., Darrow T.L., Paterson H., Marais R., Marshall C.J., Wooster R., Stratton M.R., Futreal P.A. 2002. Mutations of the BRAF gene in human cancer. Nature. 417, 949–954.
Tannapfel A., Sommerer F., Benicke M., Katalinic A., Uhlmann D., Witzigmann H., Hauss J., Wittekind C. 2003. Mutations of the BRAF gene in cholangiocarcinoma but not in hepatocellular carcinoma. Gut. 52, 706-712.
Soares P., Trovisco V., Rocha A.S., Lima J., Castro P., Preto A., Maximo V., Botelho., Seruca R., Sobrinho-Sim es M. 2003. BRAF mutations and RET/PTC rearrangements are alternative events in the etiopathogenesis of PTC. Oncogene. 22, 4578–4580.
Kimura E.T., Nikiforova M.N., Zhu Z., Knauf J.A., Nikiforov Y.E., Fagin J.A. 2003. High prevalence of BRAF mutations in thyroid cancer. Genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma.Cancer Res. 63, 1454–1457.
Xu X., Quiros R.M., Gattuso P., Ain K.B., Prinz R.A. 2003. High prevalence of BRAF gene mutation in papillary thyroid carcinomas and thyroid tumor cell lines. Cancer Res. 63, 4561–4567.
Cohen Y., Xing M., Mambo E., Guo Z., Wu G., Trink B., Beller U., Westra W.H., Ladenson P.W., Sidransky D. 2003. BRAF mutation in papillary thyroid carcinoma. J. Natl. Cancer Inst. 95, 625–627.
Namba H., Nakashima M., Hayashi T., Hayashida N., Maeda S., Rogounovitch T.I., Ohtsuru A., Saenko V.A., Kanematsu T., Yamashita S. 2003.Clinical implication of hot spot BRAF mutation, V599E, in papillary thyroid cancers. J. Clin. Endocr. Metab. 88, 4393–4397.
Fukushima T., Suzuki S., Mashiko M., Ohtake T., Endo Y., Takebayashi Y., Sekikawa K., Hagiwara K., Takenoshita S. 2003. BRAF mutations in papillary carcinomas of the thyroid. Oncogene. 22, 6455–6457.
Nikiforova M.N., Kimura E.T., Gandhi M., Biddinger P.W., Knauf J.A., Basolo F., Zhu Z., Giannini R., Salvatore G., Fusco A., Santoro M., Fagin J.A., Nikiforov Y.E. 2003. BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J. Clin. Endocrinol. Metab. 88, 5399–5404.
Landegren U. 1996. Laboratory Protocols for Mutation Detection. Oxford: Oxford Univ. Press.
Smanik P.A., Furminger T.L., Mazzaferri E.L., Jhiang S.M. 1995. Breakpoint characterization of the ret/PTC oncogene in human papillary thyroid carcinoma. Hum. Mol.Genet. 4, 2313–2318.
Bunone G., Uggeri M., Mondellini P., Pierotti M.A., Bongarzone I. 2000. RET receptor expression in thyroid follicular epithelial cell-derived tumors. Cancer Res. 60, 2845–2849.
Saenko V., Rogounovitch T., Shimizu-Yoshida Y., Abrosimov A., Lushnikov E., Roumiantsev P., Matsumoto N., Nakashima M., Meirmanov S., Ohtsuru A., Namba H., Tsyb A., Yamashita S. 2003. Novel tumorigenic rearrangement, delta rfp/ret, in a papillary thyroid carcinoma from externally irradiated patient. Mutat. Res. 527, 81-90.
Takahashi M., Ritz J., Cooper G.M. 1985. Activation of a novel human transforming gene, ret, by DNA rearrangement.Cell.42, 581–588.
Nikiforov Y.E., Rowland J.M., Bove K.E., Monforte-Munoz H., Fagin J.A. 1997. Distinct pattern of ret oncogene rearrangement in morphological variants of radiation-induced and sporadic thyroid papillary thyroid carcinomas in children. Cancer Res. 57, 1690–1694.
Bongarzone I., Vigneri P., Mariani L., Collini P., Pilotti S., Pierotti M.A. 1998. RET/NTRK1 rearrangements in thyroid gland tumors of the papillary carcinoma family: Correlation with clinopathological features. Clin. Cancer Res. 4, 223–228.
Thomas G.A., Bunnell H., Cook H.A., Williams E.D., Nerovnya A., Cherstvoy E.D., Tronko N.D., Bogdanova T.I., Chiappetta G., Viglietto G., Pentimalli F., Salvatore G., Fusco A., Santoro M., Vecchio G. 1999.High prevalence of RET/PTC rearrangements in Ukrainian and Belarussian post-Chernobyl thyroid papillary carcinomas: A strong correlation between RET/PTC3 and the solid-follicular variant.J. Clin. Endocr. Metab. 84, 4232–4238.
Cinti R., Yin L., Ilc K., Berger N., Basolo F., Cuccato S., Giannini R., Torre G., Miccoli P., Amati P., Romeo G., Corvi R.2000.RET rearrangements in papillary thyroid carcinomas and adenomas detected by interphase FISH.Cytogenet. Cell Genet.88, 56–61.
Ishizaka Y., Kobayashi S., Ushijima T., Hirohashi S., Sugimura T., Nagao M. 1991. Detection of retTPC/PTC transcripts in thyroid adenomas and adenomatous goiter by an RT-PCR method. Oncogene. 6, 1667–1672.
Sugg S.L., Ezzat S., Rosen I.B., Freeman J.L., Asa S.L. 1998. Distinct multiple RET/PTC gene rearrangements in multifocal papillary thyroid neoplasia. J. Clin.Endocr. Metab. 83, 4116–4122.
Viglietto G., Chiappetta G., Martinez-Tello F.J., Fukunaga F.H., Tallini G., Rigopoulou D., Visconti R., Mastro A., Santoro M., Fusco A. 1995. RET/PTC oncogene activation is an early event in thyroid carcinogenesis.Oncogene. 11, 1207–1210.
Corvi R., Martinez-Alfaro M., Harach H.R., Zini M., Papotti M., Romeo G. 2001. Frequent RET rearrangements in thyroid papillary microcarcinoma detected by interphase fluorescence in situ hybridization. Lab.Invest.81, 1639–1645.
Harach H.R., Franssila K.O., Wasenius V.M. 1985.Occult papillary carcinoma of the thyroid. A "normal" finding in Finland. A systematic autopsy study. Cancer. 56, 531–538.
Newton C.R., Graham A., Heptinstall L.E., Powell S.J., Summers C., Kalsheker N., Smith J.C., Markham A.F. 1989. Analysis of any point mutation in DNA. The amplification refractory mutation system (ARMS).Nucleic Acids Res.17, 2503–2516.
Mercer K.E., Pritchard C.A. 2003. Raf proteins and cancer: B-Raf is identified as a mutational target. Biochim.Biophys. Acta. 1653, 25–40.
Marais R., Light Y., Paterson H.F., Mason C.S., Marshall C.J. 1997.Differential regulation of Raf-1, ARaf and B-Raf by oncogenic Ras and tyrosine kinases.J. Biol. Chem. 272, 4378–4383.
Pollock P.M., Harper U.L., Hansen K.S., Yudt L.M., Stark M., Robbins C.M., Moses T.Y., Hostetter G., Wagner U., Kakareka J., Salem G., Pohida T., Heenan P., Duray P., Kallioniemi O., Hayward N.K., Trent J.M., Meltzer P.S. 2003. High frequency of BRAF mutations in nevi. Nature Genet. 33, 19–20.
Flores J.F., Walker G.J., Glendening J.M., Haluska F.G., Castresana J.S., Rubio M.P., Pastorfide G.C., Boyer L.A., Kao W.H., Bulyk M.L., Barnhill R.L., Hayward N.K., Housman D.E., Fountain J.W. 1996.Loss of the p16INK4a and p15INK4b genes, as well as neighboring 9p21 markers, in sporadic melanoma. Cancer Res. 56, 5023–5032.
Kumar R., Sauroja I., Punnonen K., Jansen C., Hemminki K. 1998. Selective deletion of exon 1 beta of the p19ARF gene in metastatic melanoma cell lines. Genes Chromosomes Cancer.23,273–277.
Zuo L., Weger J., Yang Q., Goldstein A.M., Tucker M.A., Walker G.J., Hayward N., Dracopoli N.C. 1996. Germline mutations in the p16INK4a binding domain of CDK4 in familial melanoma. Nature Genet. 12, 97–99.
Lackey K., Cory M., Davis R., Frye S.V., Harris P.A., Hunter R.N., Jung D.K., McDonald O.B., McNutt R.W., Peel M.R., Rutkowske R.D., Veal J.M., Wood E.R. 2000.The discovery of potent cRaf1 kinase inhibitors. Bioorg.Med. Chem. Lett. 10, 223–226.
Kolch W. 2002. Ras/Raf signalling and emerging pharmacotherapeutic targets. Expert. Opin. Pharmacother. 3, 709–718.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasil'ev, E.V., Roumiantsev, P.O., Saenko, V.A. et al. Molecular Analysis of Structural Abnormalities in Papillary Thyroid Carcinoma Genome. Molecular Biology 38, 538–548 (2004). https://doi.org/10.1023/B:MBIL.0000037006.90193.f6
Issue Date:
DOI: https://doi.org/10.1023/B:MBIL.0000037006.90193.f6