Abstract
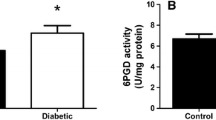
The aim of the present study was to investigate the effects of treatment with antioxidant stobadine (ST) on the activities of enzymes related with pentose phosphate pathway and glutathione-dependent metabolism and the other markers of oxidative stress in brain and peripheral organs of diabetic rats, and to compare the effects of ST treatment alone with the effects of treatments with another antioxidant vitamin E and ST plus vitamin E. Rats were made diabetic by the injection of streptozotocin (STZ; 55 mg/kg IP), and, 2 days later, some control and diabetic rats were left untreated or treated with ST (24.7 mg/kg/day, orally), vitamin E (400–500 U/kg/day, orally), or both substances together. In the brain, although 6-phosphogluconate dehydrogenase activity (6-PGD) did not change, glucose-6-phosphate dehydrogenase activity (G-6PD) was markedly increased in diabetic rats compared with controls; only combined treatment with ST and vitamin E produced a partial prevention on this alteration. The aorta G-6PD and 6-PGD of diabetic rats were 52% and 36% of control values, respectively. Neither single treatments with each antioxidant nor their combination altered the G-6PD and 6-PGD in aorta of diabetic rats. Glutathione peroxidase (GSHPx) activity was increased by STZ-diabetes in brain, heart, and kidney. In diabetic brain, vitamin E alone or combination with ST kept GSHPx at normal levels. Diabetes-induced stimulation in GSHPx did not decrease in response to the treatment with vitamin E in heart and kidney, but was greatly prevented by ST alone. The activity of glutathione reductase (GR) was decreased in brain and heart of diabetic rats. The treatment with each antioxidant or with a combination of both agents completely prevented this deficiency and resulted in further activation of GR in diabetic tissues. Glutathione S-transferase (GST) activity did not significantly change in diabetic brain and aorta. GST was stimulated by all treatment protocols in the brain of diabetic rats and was depressed in aorta of control rats. Catalase (CAT) was activated in diabetic heart but depressed in diabetic kidney. Diabetes-induced abnormalities in CAT activity did not respond to vitamin E alone in heart, was moderately ameliorated by the treatment with this vitamin in kidney, and was completely prevented by ST alone in both tissues. Superoxide dismutase (SOD) activity of brain and heart was unchanged by the diabetes but inhibited in diabetic kidney after the treatment ST alone or ST plus vitamin E. The lipid peroxidation (MDA) was increased in diabetic brain and heart. ST or vitamin E alone partly prevented diabetes-induced increase in MDA in brain and heart; however, antioxidant combination achieved a completely amelioration in MDA of these tissues of diabetic rats. Kidney MDA levels were similar in control and untreated diabetic animals. ST and vitamin E treatments, when applied separately or together, significantly reduced kidney MDA in both control and diabetic rats; and the combined effect of antioxidants was greater than that of each alone. These results are consistent with the degenerative role of hyperglycemia on cellular reducing equivalent homeostasis and antioxidant defense, and provide further evidence that pharmacological intervention of different antioxidants may have significant implications in the prevention of the prooxidant feature of diabetes and protects redox status of the cells.
Similar content being viewed by others
REFERENCES
Bayners, J. W. 1991. Role of oxidative stress in development of complications in diabetes. Diabetes 40:405–412.
Karasu, Ç., Dewhurst, M., Stevens, E. J., and Tomlinson, D. R. 1995. Effects of anti-oxidant treatment on sciatic nerve dysfunction in streptozotocin-diabetic rats: Comparison with essential fatty acids. Diabetologia 38:129–134.
Giugliano, D., Ceriello, A., and Paolisso, G. 1996. Oxidative stress and diabetic vascular complications. Diabetes Care 19:257–267.
Karasu, Ç. 2000. Time course of changes in endothelium-dependent and-independent relaxation of chronically diabetic aorta: Role of reactive oxygen species. Eur. J. Pharmacol. 392:163–173.
Karasu, Ç., Ozansoy, G., Bozkurt, O., Erdogan, D., and ömeroglu, S. 1997. Antioxidant and triglyceride lowering effects of vitamin E associated with the prevention of abnormalities in the reactivity and morphology of aorta from streptozotocin-diabetic rats. Metabolism 46:872–879.
Martins, R. N., Stokes, G. B., and Masters, C. L. 1986. Regulation of liver and brain hexose monophosphate dehydrogenases by insulin and dietary intake in the female rat. Mol. Cell Biochem. 70:169–175.
Roy, S., Sen, C. K., Tritschler, H. J., and Packer, L. 1997. Modulation of cellular reducing equivalent homeostasis by alphalipoic acid: Mechanisms and implications for diabetes and ischemic injury. Biochem. Pharmacol. 53:393–399.
Hink, U., Li, H., Mollnau, H., Oelze, M., Matheis, E., Hartmann, M., Skatchkov, M., Thaiss, F., Stahl, R. A., Warnholtz, A., Meinertz, T., Griendling, K., Harrison, D. G., Forstermann, U., and Munzel, T. 2001. Mechanisms underlying endothelial dysfunction in diabetes mellitus. Circ. Res. 88:E14-E22.
Onozato, M. L., Tojo, A., Goto, A., Fujita, T., and Wilcox, C. S. 2002. Oxidative stress and nitric oxide synthase in rat diabetic nephropathy: Effects of ACEI and ARB. Kidney Int. 61:186–194.
Abdel-Rahim, E. A., El-Saadany, S. S., Abo-Eytta, A. M., and Wasif, M. M. 1992. The effect of Sammo administration on some fundamental enzymes of pentose phosphate pathway and energy metabolites of alloxanized rats. Die. Nahrung. 36:8–14.
Mak, D. H. F., Ip, S. P., Li, P. C., Poon, M. K. T., and Ko, K. M. 1996. Alterations in tissue glutathione antioxidant system in streptozotocin-induced diabetic rats. Mol. Cell Biochem. 162:153–158.
Ramanathan, M., Jaiswal, A. K., and Bhattacharya, S. K. 1999. Superoxide dismutase, catalase and glutathione peroxidase activities in the brain of streptozotocin induced diabetic rats. Ind. J. Exp. Biol. 37:182–183.
Kaul, N., Siveski-Iliskovic, N., Hill, M., Khaper, N., Seneviratne, C., and Singal, P. K. 1996. Probucol treatment reverses antioxidant and functional deficit in diabetic cardiomyopathy. Mol. Cell Biochem. 160/161:283–288.
Koçak, G., Aktan, F., Canbolat, O., özoğul, C., Elbeğ, Ş., Yildizoğlu-Ari, N., and Karasu, Ç. 2000. Alpha-lipoic acid treatment ameliorates metabolic parameters, blood pressure, vascular reactivity and morphology of vessels already damaged by streptozotocin-diabetes (The ADIC Study Group). Diabetes Nutr. Metab. 13:308–319.
Horakova, L. and Stolc, S. 1998. Antioxidant and pharmacodynamic effects of pyridoindole stobadine. Gen. Pharmacol. 30:627–638.
Pekiner, B., Ulusu, N. N., Das-Evcimen, N., Shilli, M., Aktan, F., Stefek, M., Stolc, S., and Karasu, Ç. 2002. In vivo treatment with stobadine prevents lipid peroxidation, protein glycation and calcium overload but does not ameliorate Ca2+-ATPase activity in heart and liver of streptozotocin-diabetic rats: Comparison with vitamin E. Biochim. Biophys. Acta. 62143, 588:71–78.
Stefek, M., Sotnikova, R., Okruhlicova, L., Volkovova, K., Kucharska, J., Gajdosik, A., Gajdosikova, A., Mihalova, D., Hozova, R., Tribulova, N., and Gvozdjakova, A. 2000. Effect of dietary supplementation with the pyridoindole antioxidant stobadine on antioxidant state and ultrastructure of diabetic rat myocardium. Acta Diabetol. 37:111–117.
Sotnikova, R., Stefek, M., Okruhlicova, L., Navarova, J., Bauer, V., Gajdosik, A., and Gajdosikova, A. 2001. Dietary supplementation of the pyridoindole antioxidant stobadine reduces vascular impairment in streptozotocin-diabetic rats. Methods Find. Exp. Clin. Pharmacol. 23:121–129.
Karasu, Ç., Ulusu, N., Avci, A., Sahilli, M., Canbolat, O., Ozansoy, G., Ari, N., Stefek, S., Stolc, S., and Gojdosik, A. 2001. Effects of ST treatment on pentose phosphate pathway, glutathione-dependent enzymes, superoxide dismutase, and lipid peroxidation in the brain of diabetic rat; comparison with vitamin E. Diabetologia 44:(Suppl. 1) A155–592.
Karasu, Ç., Avi, A., Canbolat, O., Bali, M., Ozansoy, G., Ceylan, A., Ari, N., and Stefek, M. 2002. Comparative effects of stobadine and vitamin E treatments on oxidative stress markers in heart and kidney of streptozotocin-diabetic rats. Free Radic. Biol. Med. 33(Suppl. 1):S188, 494.
Ulusu, N. N., Kus, M. S., Acan, N. L., and Tezcan, E. F. 1999. A rapid method for the purification of glucose-6-phosphate dehydrogenase from bovine lens. Int. J. Biochem. Cell Biol. 31:787–796.
Pearse, B. M. F. and Rosemeyer, M. A. 6-Phosphogluconate dehydrogenase from human erytrocytes. Page 220, in Colowich, S. R. and Kaplan, N. O. (eds.), Methods Enzymology. XLI Academic Press Inc., London.
Lawrence, R. A. and Burk, R. F. 1976. Glutathione peroxidase activity in selenium-deficient rat liver. Biochem. Biophys. Res. Commun. 71:952–958.
Acan, N. L. and Tezcan, E. F. 1989. Sheep brain glutathione reductase: Prufication and general properties. FEBS Lett. 250:72–74.
Habig, W. H., Pabst, M. J., and Jakoby, W. B. 1974. Glutathione S-transferase. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249:7130–7139.
Durak, I., Yurtarslan, Z., Canbolat, O., and Akyol, O. 1993. A methodological approach to superoxide dismutase (SOD) activity assay based on inhibition of nitroblue tetrazolium (NBT) reduction. Clin. Chim. Acta. 214:103–104.
Aebi, H. 1983. Catalase in vitro. Methods Enzymol. 105:121–126.
Jain, S. K. and Levine, S. N. 1995. Elevated lipid peroxidation and vitamin E-quinone levels in heart ventricles of streptozotocin-treated diabetic rats. Free Radic. Biol. Med. 18:337–341.
Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Pereira, B., Rosa, L. F., Safi, D. A., Bechara, E. J., and Curi, R. 1994. Superoxide dismutase, catalase and glutathione peroxidase activities in the lymphoid organs of diabetic rats. J. Endocrinol. 142:161–165.
M cD ermott, B. M., Flatt, P. R., and Strain, J. J. 1994. Effects of copper deficiency and experimental diabetes on tissue antioxidant enzyme levels in rats. Ann. Nutr. Metab. 38:263–269.
Thurston, J. H., M cD ougal, D. B., Jr, Hauhart, R. E., and Schulz, D. W. 1995. Effects of acute, subacute, and chronic diabetes on carbohydrate and energy metabolism in rat sciatic nerve: Relation to mechanisms of peripheral neuropathy. Diabetes 44:190–195.
Lakhman, S. S., Sharma, P., Kaur, G., and Kaur, G. 1994. Changes in glucose metabolism from discrete regions of rat brain and its relationship to reproductive failure during experimental diabetes. Mol. Cell Biochem. 141:97–102.
Berg, E. A., Wu, J. Y., Campbell, L., Kagey, M., and Stapleton, S. R. 1995. Insulin-like effects of vanadate and selenate on the expression of glucose-6-phosphate dehydrogenase and fatty acid synthase in diabetic rats. Biochimie 77:919–924.
Zhang, Z., Apse, K., Pang, J., and Stanton, R. C. 2000. High glucose inhibits glucose-6-phosphate dehydrogenase via cAMP in aortic endothelial cells. J. Biol. Chem. 275:40042–40047.
Guzik, T. J., Mussa, S., Gastaldi, D., Sadowski, J., Ratnatunga, C., Pillai, R., and Channon, K. M. 2002. Mechanisms of increased vascular superoxide production in human diabetes mellitus: Role of NAD(P)H oxidase and endothelial nitric oxide synthase. Circulation 105:1656–1662.
Gupta, B. L. and Baquer, N. Z. 1998. Hexokinase, glucose-6-phosphate dehydrogenase and antioxidant enzymes in diabetic reticulocytes: Effects of insulin and vanadate. Biochem. Mol. Biol. Int. 46:1145–1152.
Gaskin, R. S., Estwick, D., and Peddi, R. 2001. G6PD deficiency: Its role in the high prevalence of hypertension and diabetes mellitus. Ethn. Dis. Fall. 1:749–754.
Yadav, P., Sarkar, S., and Bhatnagar, D. 1997. Action of capparis decidua against alloxan-induced oxidative stress and diabetes in rat tissues. Pharmacol. Res. 36:221–228.
Raza, H., Ahmed, I., John, A., and Sharma, A. K. 2000. Modulation of xenobiotic metabolism and oxidative stress in chronic streptozotocin-induced diabetic rats fed with momordica charantia fruit extract. J. Biochem. Mol. Toxicol. 14:132–139.
Agha, A. M., El-Fattah, A. A., Al-Zuhair, H. H., and Al-Rikabi, A. C. 2001. Chemopreventive effect of Ginkgo biloba extract against benzo(a)pyrene-induced forestomach carcinogenesis in mice: Amelioration of doxorubicin cardiotoxicity. J. Exp. Clin. Cancer Res. 20:39–50.
Sanders, R. A., Rauscher, F. M., and Watkins, J. B., 3rd, 2001. Effects of quercetin on antioxidant defense in streptozotocin-induced diabetic rats. J. Biochem. Mol. Toxicol. 15:143–149.
Gupta, D., Raju, J., Prakash, J., and Baquer, N. Z. 1999. Change in the lipid profile, lipogenic and related enzymes in the livers of experimental diabetic rats: Effect of insulin and vanadate. Diabetes Res. Clin. Pract. 46:1–7.
Volkovova, K., Chorvathova, V., Jurcovicova, M., Koszeghyova, L., and Bobek, P. 1993. Antioxidative state of the myocardium and kidneys in acute diabetic rats. Physiol. Res. 42:251–255.
Karasu, Ç. 1999. Increased activity of H2O2 in aorta isolated from chronically streptozotocin-diabetic rats: Effects of antioxidant enzymes and enzymes inhibitors. Free Radic. Biol. Med. 27:16–27.
Zobali, F., Avci, A., Canbolat, O., and Karasu, C. 2002. Effects of vitamin A and insulin on the antioxidative state of diabetic rat heart: A comparison study with combination treatment (antioxidants in diabetes-induced complications) The ADIC Study Group. Cell Biochem. Funct. 20:75–80.
Kowluru, R. A., Kern, T. S., and Engerman, R. L. 1997. Abnormalities of retinal metabolism in diabetes or experimental galactosemia: IV. Antioxidant defense system. Free Radic. Biol. Med. 22:587–592.
Gokkusu, C., Palanduz, S., Ademoglu, E., and Tamer, S. 2001. Oxidant and antioxidant systems in NIDDM patients: influence of vitamin E supplementation. Endocr. Res. 27:377–386.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ulusu, N.N., Sahilli, M., Avci, A. et al. Pentose Phosphate Pathway, Glutathione-Dependent Enzymes and Antioxidant Defense During Oxidative Stress in Diabetic Rodent Brain and Peripheral Organs: Effects of Stobadine and Vitamin E. Neurochem Res 28, 815–823 (2003). https://doi.org/10.1023/A:1023202805255
Issue Date:
DOI: https://doi.org/10.1023/A:1023202805255