Abstract
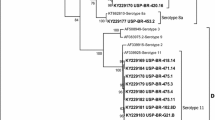
Ten fowl adenoviruses (FAVs), isolated from suspected cases of inclusion body hepatitis (IBH) in quails and broilers, were characterized by a hexon-based polymerase chain reaction (PCR) combined with restriction enzyme analysis (REA) of the amplified DNA fragments. All the isolates could be detected using H1/H2 and H3/H4 primer sets. Amplification of DNA with H1/H2 and H3/H4 primer sets resulted in fragments of approximately 1219 bp and 1319 bp, respectively. HaeII digestion of the H1/H2 PCR products and HpaII digestion of the H3/H4 PCR products characterized all the isolates in FAV groups, known from genomic typing using the whole DNA. For some of the isolates, neutralization tests were used to confirm these results. The results revealed that, as well as FAV serotype 1, which is the sole member of DNA group A, FAVs of DNA group E are also associated with IBH in poultry in northern India. The FAV specific PCR combined with REA was found to be very useful in investigating the epidemiological situation in the field. It was even possible to define mixed infections with more than one FAV.
Similar content being viewed by others
REFERENCES
Erny, K.M., Barr, D.A. and Fahey, K.J., 1991. Molecular characterization of highly virulent fowl adenoviruses associated with outbreaks of inclusion body hepatitis. Avian Pathology, 20, 597-606
Erny, K.M., Pallister, J. and Sheppard, M., 1995. Immunological and molecular comparison of fowl adenovirus serotype 4 and 10. Archives of Virology, 140, 491-501
Grewal, G.S., Sharma, S.N. and Deka, B.C., 1981. Inclusion body hepatitis in broiler chickens. Indian Journal of Poultry Science, 16, 51-56
Grewal, G.S., Singh, A., Singh, B. and Oberoi, M.S., 1994. Inclusion body hepatitis in Japanese quail (Coturnix coturnix japonica). Indian Journal of Animal Sciences, 64, 665-667
Grimes, T.M., King, D.J. and Kleven, S.H., 1976. Application of a microtiter cell-culture method to characterization of avian adenoviruses. Avian Diseases, 20, 299-310
Grimes, T.M., King, D.J., Kleven, S.H. and Fletcher, O.J., 1977. Involvement of a type-8 avian adenovirus in the etiology of inclusion body hepatitis. Avian Diseases, 21, 26-38
Hess, M., 2000. Detection and differentiation of avian adenoviruses: a review. Avian Pathology, 29, 195-206
Hess, M., Raue, R. and Prusas, C., 1999. Epidemiological studies on fowl adenoviruses isolated from cases of infectious hydropericardium. Avian Pathology, 28, 433-439
Jack, S.W. and Reed, W.M., 1990. Further characterization of an avian adenovirus associated with inclusion body hepatitis in Bobwhite quails. Avian Diseases, 34, 526-530
Jack, S.W., Reed, W.M. and Bryan, T.A., 1987. Inclusion body hepatitis in Bobwhite quail (Colinus virginianus). Avian Diseases, 31, 661-665
Khanna, M., Oberoi, M.S., Sawhney, S.M.S. and Sharma, N., 1992. Application of dot-enzyme immunoassay for the detection of avian adeno and avian adeno-associated viruses. Indian Journal of Animal Sciences, 62, 830-831
King, D.J., Pursglove, S.R. (Jr.) and Davidson, W.R., 1981. Adenovirus isolation and serology from wild Bobwhite quail (Colinus virginianus). Avian Diseases, 25, 678-682
McFerran, J.B., 1997. Group I adenovirus infections. In: B.W. Calnek, H.J. Barnes, C.W. Beard, L.R. McDougald and Y.M. Saif (eds), Diseases of Poultry, 10th edn, (Iowa State University Press, Ames, IA), 608-620
Monreal, G., 1992. Adenovirus and adeno-associated viruses of poultry. Poultry Science Reviews, 4, 1-27
Monreal, G., Dorn, R. and Kassim, M., 1980. Detection of neutralizing antibodies against avian adenovirus in a microtitre cell-culture system. Berliner Münchner Tierärztliche Wochenschrift, 93, 125-128
Nagal, K.B., Maiti, N.K., Oberoi, M.S. and Sharma, S.N., 1990. Antigenic characterization of fowl adenovirus strains by neutralization test and dot-ELISA. Indian Journal of Animal Sciences, 60, 1023-1026
Oberoi, M.S., Sharma, S.N. and Maiti, N.K., 1990. Detection of avian adenovirus by counterimmunoelectrophoresis. Indian Journal of Animal Sciences, 60, 1307-1308
Raue, R. and Hess, M., 1998. Hexon based PCRs combined with restriction enzyme analysis for rapid detection and differentiation of fowl adenoviruses and egg drop syndrome virus. Journal of Virological Methods, 73, 211-217
Reece, R.L., Barr, D.A., Grix, D.C., Forsyth, W.M., Condron, R.J. and Hindmarsh, M., 1986. Observations of naturally occurring inclusion body hepatitis in Victorian chickens. Australian Veterinary Journal, 63, 201-202
Reed, L.J. and Muench, H.A., 1938. A simple method of estimating fifty percent endpoints. American Journal of Hygiene, 27, 493
Saifuddin, Md. and Wilks, C.R., 1992. Effects of fowl adenovirus infection on the immune system of chickens. Journal of Comparative Pathology, 107, 285-294
Singh, B. and Oberoi, M.S., 1994. Studies on avian adenoviruses with special reference to epidemiology, pathology and immunology. Final Technical Report of PL-480 project. (Punjab Agricultural University, Ludhiana, India), 1-88
Singh, A., Oberoi, M.S. and Singh, B., 1995. Pathogenicity of quail's inclusion body hepatitis virus (avian adenovirus-1) for Japanese quails and broiler chicks. Veterinary Research Communications, 19, 545-551
Singh, A., Oberoi, M.S., Jand, S.K. and Singh, B., 1996. Epidemiology of inclusion body hepatitis in poultry in northern India from 1990 to 1994. Revue Scientifique et Technique de l'Office International des Epizooties, 15, 1053-1060
Zsak, L. and Kisray, J., 1984. Grouping of fowl adenoviruses based upon the restriction patterns of DNA generated by BamHI and HindIII. Intervirology, 22, 110-114
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, A., Oberoi, M., Grewal, G. et al. The Use of PCR Combined with Restriction Enzyme Analysis to Characterize Fowl Adenovirus Field Isolates from Northern India. Vet Res Commun 26, 577–585 (2002). https://doi.org/10.1023/A:1020299700907
Issue Date:
DOI: https://doi.org/10.1023/A:1020299700907