Abstract
The psychoneuroimmunology of panic disorder is relatively unexplored. Alterations within brain stress systems that secondarily influence the immune system have been documented. A recent report indicated elevations of serotonin (5-HT) and ganglioside antibodies in patients with primary fibromyalgia, a condition with documented associations with panic disorder. In line with our interest in dysregulated 5-HT systems in panic disorder (PD), we wished to assess if antibodies directed at the 5-HT system were elevated in patients with PD in comparison to healthy volunteers. Sixty-three patients with panic disorder and 26 healthy volunteers were diagnosed by the SCID. Employing ELISA, we measured anti-5-HT and 5-HT anti-idiotypic antibodies (which are directed at 5-HT receptors). To include all subjects in one experiment, three different batches were run during the ELISA. Plasma serotonin anti-idiotypic antibodies: there was a signficiant group effect [patients > controls (p = .007)] and batch effect but no interaction. The mean effect size for the three batches was .76. Following Z-score transformation of each separate batch and then combining all scores, patients demonstrated significantly elevated levels of plasma serotonin anti-idiotypic antibodies. Neither sex nor age as covariates affected the significance of the results. There was a strong correlation between anti-serotonin antibody and serotonin anti-idiotypic antibody measures. Plasma anti-serotonin antibodies: there was a signficiant diagnosis effect [patients > controls (p = .037)]. Mean effect size for the three batches was .52. Upon Z-score transformation, there was a diagnosis effect with antibody elevations in patients. Covaried for sex and age, the result falls below significance to trend levels. The data raise the possibility that psycho-immune dysfunction, specifically related to the 5-HT system, may be present in PD. Potential interruption of 5-HT neurotransmission through autoimmune mechanisms may be of pathophysiologic significance in certain patients with panic disorder. It remains to be demonstrated if the peripheral autoimmunity is representative of CNS 5-HT neuronal alterations. Replication appears warranted.
Similar content being viewed by others
Main
Recent reports document extensive links between neurally-mediated stress responses and immune activation. Psychoimmunological studies in panic disorder are therefore of potential relevance to understanding the pathophysiology of panic disorder. Available studies indicate that patients with panic disorder, in relation to healthy volunteers, exhibit activation of the immune response on a variety of measures: Ramesh et al. (1991) reports that “significantly increased IgA levels and tendency toward increase in other immunoglobulins in panic disorder patients suggest an immune dysfunction”; Schmidt-Traub (1995) describes a high frequency of allergic reaction of the IgE-mediated type I in panic disorder. Panic disorder, agoraphobia, allergic disorder, and vasomotor reactions are viewed within the framework of a psycho-endocrino-immunological spectrum of disorders. Andreoli et al. (1992) found that patients with major depression and panic disorder had greater numbers of T cells and phytohemagglutinin (PHA) mitogen responses than depressive patients without PD, as well as increased PHA and concanavalin A mitogen responses compared to healthy controls. This suggests that panic disorder comorbidity significantly contributes to the variance of immunologic parameters in major depression. Brambilla et al. 1994 report elevations of plasma interleukin-1β in panic disorder, a key cytokine mediating multiple CNS effects. Finally, Marazziti et al. (1992) state that patients with panic attacks differed from healthy controls and depressives with respect to CD4+ cells, which were significantly lowered.
With respect to other diagnoses, it is now reasonably established that obsessive compulsive disorder and Tourette's syndrome in children can be triggered by a post-streptococcal autoimmune process (Allen et al. 1995). Work by Maes et al. (1995) have shown increased cytokine activity, including interleukin-6 elevations, in patients suffering from depression.
A report by Klein et al. (1992) indicates elevations of antibodies to serotonin and gangliosides in patients with primary fibromyalgia. A documented association between panic disorder and fibromyalgia (Hudson et al. 1985), as well as a strong overlap in clinical symptomatology (Waylonis and Heck 1992), raises the possibility of autoimmunity involving the serotonin system in panic disorder.
In general, the variable region of a circulating antibody may often act like an antigen and may induce the generation of self-directed antibodies (described as anti-idiotypic or anti-id) (see Figure 1 ). In our specific case (Tamir et al. 1991), we have previously induced formation of antibodies to serotonin in rabbits, isolated these antibodies and after purification, reinjected them into rabbits. The injected anti-serotonin antibodies then generates serotonin anti-idiotypic antibodies.
Flowchart depicts the experimental formation of a serotonin anti-idiotypic (anti-id) antibody (Tamir et al. 1991). Abbreviations Inj. = active immunization of rabbit with antigenic material linked to serotonin; Prod. = immunological response of rabbit to injected antigens. Finally, a 5-HT anti-idiotypic antibody can be developed experimentally, which recognizes and binds serotonin receptor sites, either as an agonist or antagonist. As the current study suggests, there exists the possibility that aspects of this process may occur spontantaneously in the clinical situation
If ligand binding sites of anti-serotonin antibodies produced through immunization are sufficiently similar to binding sites on serotonin receptors, then the anti-idiotypic antibody will react with the related epitope and bind to serotonin receptors (Tamir et al. 1991, Wade et al. 1994). These anti-idiotypic antibodies which recognize serotonin receptors may act as serotonin agonists or antagonists (Wade et al. 1994). A spontaneous anti-idiotypic reaction involving the serotonin system may be therefore be analagous to the pathophysiology of myasthenia gravis, where antibodies directed at the neuromuscular junction are produced (Wasserman et al. 1982). Given our interest in the role of serotonin in the pathophysiology of panic disorder (Coplan et al. 1992), we measured plasma levels of anti-serotonin and serotonin anti-id antibodies in patients and healthy controls.
METHODS
Subjects
Sixty-three patients meeting present DSM-III-R criteria (American Psychiatric Association 1987) for panic disorder with or without agoraphobia were recruited by advertisement in local newspapers and were diagnostically assessed by experienced research psychiatrists [and authors of this article (JDC, LAP, JMG)] at the Biological Studies Unit, New York State Psychiatric Institute by standard psychiatric interview. Diagnoses were confirmed by administration of the structured clinical interview for DSM-III-R (SCID) (Spitzer et al. 1990) by a trained research nurse.
Inclusion and exclusion criteria are discussed in detail previously (Coplan et al. 1995). Twenty-six healthy subjects, assessed by experienced research psychiatrists at New York State Psychiatric Institute and confirmed by a trained research nurse by the SCID, served as a healthy volunteer control group. Informed consent was obtained from all participants.
The mean age of the patient group was older [32.29 years (SD = 8.1)] than the control mean [27.4 years (SD = 7.6); t = 2.63; df = 87; p = .01]. There were more females in the patient group (56%) than in the control group (35%) at a trend level; χ2 = 3.05; p = .08. Medication status was not necessarily an exclusion criteria; 21 (33%) patients were on medications. A total of 10 patients were taking benzodiazepines (alprazolam 5, lorazepam 3, diazepam 1, temazepam 1); five patients were taking antidepressants (fluoxetine 2, paroxetine 1, imipramine 1, trimipramine 1), one patient was taking buspirone, two patients were taking anti-inflammatories (naprosyn, 2), two patients were taking H-2 blocker antacids (ranitidine, 2), and one patient was taking a bronchodilator (albuterol, 1). As bloods were performed before patients were tapered off medications, to be eligible for participation in biological studies and treatment, patients were therefore taking their routine doses of medications.
The routine baseline screening for all subjects included physical examination, blood tests for liver profile and electrolyte function, complete blood count and thyroid function tests, electrocardiogram, pregnancy test, and urinalysis.
15 ccs of blood was collected into “SST Gel and Clot Activator tubes” (Beckton-Dickinson), inverted six times and allowed to clot in an upright position at room temperature. Samples were allowed to clot for between three to four hours. Plasma was then pippetted into separate tubes for centrifugation at 2800 RPM for 20 minutes. Centrifuged plasma was then placed into Nunc cryotubes (Intermed) and stored in a −80° C refrigerator.
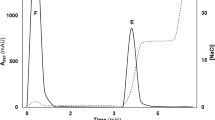
The measurement of serotonin anti-id can be accomplished by using enzyme-linked immunosorbent assay (ELISA) and anti-serotonin as an antigen (Tamir et al. 1991). ELISA was used to determine whether antibodies that react with serotonin or with anti-serotonin sera are present in the plasma of control and patients as previously described (Tamir et al. 1991). Briefly, serotonin bound to bovine serum albumin was used as the antigen for detecting anti-serotonin antibodies. Affinity purified antibodies to serotonin were used as antigen for detecting serotonin anti-idiotypic antibodies. The immune complex was measured with goat anti-human immunoglobulin conjugated to alkaline phosphatase. Alkaline phosphotase was visualized using p-nitrophenol phosphate as substrate and an ELISA reader fitted with 405 nm filter. Because the methodology did not permit measurement of all subjects in one batch, three separate ELISA experiments were performed. All measurements were performed in duplicate and the mean computed for data analytic purposes. Staff performing laboratory experiments were blind to the diagnosis attached to the specimens.
DATA ANALYSIS
The first analysis ascertained the magnitude of group and batch differences by both ANOVA and also by determining the mean effect size of the three batches for both type of antibody. The next strategy was to convert each batch's scores to standardized Z-scores, permitting combining of the three batches in a single analysis, thereby controlling for potential batch effects. As laboratory analyses were performed blind, there was a possibility that the proportion of patients to controls for each of the three ELISA procedures were significantly different from each other. Thus, the analyses required that the proportion of patients to controls was equivalent for each batch. Elisa scores of the dependent variables were then compared using one-way ANCOVA while the independent variables, sex, and age were used as covariates. Unpaired T-tests were used to assess the effects of medication within the patient group.
Significance was determined at an α level of p < .05, two-tailed.
RESULTS
Plasma Serotonin Anti-idiotypic Antibodies
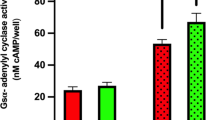
There was a significant diagnosis effect (F = 7.57; df = 1;83; p = .007). Patients had greater ELISA values (.53 SD = .12) than did healthy volunteers (.48, SD = .16) (see Figure 2 ). A significant batch effect (F = 12.6; df = 2;83; p = .00001) with highest ELISA values evident during the last batch (mean ELISA value = .47 (n = 33),.49 (n = 29) and .6 (n = 27), respectively) was noted but no batch by diagnosis effect (see Figure 3 ). The effect size for each batch was 1.2, .88, and .2, respectively, with a mean effect size for the three batches of .76, indicating elevations of antibody in patients with a strong effect size. A substantially greater spread of healthy volunteer values accounted for the lack of high effect size in the third batch.
Elevated 5-HT receptor plasma autoantibody levels on panic disorder. Group effect: p = .007; batch effect: p < .001. Graph depicts the effect size for the patient/control differences for the three separate batch procedures (1.2, .88, and .2, respectively). A significant batch effect (F = 12.6; df = 2;83; p < .0001) with highest ELISA values evident during the last batch (mean ELISA value = .47, .49, and .6, respectively) was noted but no batch × diagnosis effect. Several high values in healthy volunteers of the third batch diminished its effect size
Elevations of 5HT-receptor anti-idiotypic antibodies in panic disorders. Graph depicts the scatterplot, means and standard deviations (SD) indicating elevations of the Z-score converted ELISA values of serotonin anti-idiotypic antibodies in patients with panic disorder in comparison to healthy volunteers. This effect remain significant when covaried for both age and sex (t = 2.99; df = 87; p = .004; effect size = .6)
The proportion of patients to controls in each of the three ELISA procedures was not significantly different from each other. Each batch's scores were converted to Z-scores so that all scores could be combined. Patients demonstrated elevated levels of plasma serotonin anti-idiotypic antibodies [t = 2.99; df = 87, p = .004; .22 (SD = .93) versus −.43 (SD = .93) on Z-score transformation]. Neither entering sex or age as covariates affected the significance of the results. Using ANOVA, no sex or sex by diagnosis effect was present. Age was not related to anti-serotonin idiotypic antibody levels within the group as a whole, the patients considered alone or the controls considered alone. Patient's medication status did not have a significant effect on serotonin anti-idiotypic antibody level, even with respect to medications with specific actions on the serotonin system (three patients were on selective serotonin reuptake inhibitors and one was on buspirone), as determined by visual inspection. In support of the view that the elevations of serotonin anti-idiotypic were not an artifact of medication status, when all patients on medications were excluded from the analysis, patients continued to demonstrate elevations of Z-score converted serotonin anti-idiotypic antibody levels in comparison to healthy volunteers (t = 2.6; df = 66; p = .01). The result remained significant when age and sex covariates were introduced.
Plasma Anti-Serotonin Antibodies
There was a significant diagnosis effect (F = 4.45; df = 1;83; p = .037); patients exhibited greater ELISA values (.58 SD = .125) than did healthy volunteers (.54, SD = .11). A significant batch effect (F = 4.75; df = 2;83; p = .01) with highest ELISA values evident during the last batch was noted (.55 (n = 33), .53 (N = 29) and .62 (n = 27) respectively) but no batch by diagnosis effect. Effect sizes for each batch were .8, .7, and .05, respectively, with a mean effect size for the three batches of .52, indicating that patients exhibited antibody elevations in comparison to healthy volunteers with, on average, moderate effect size. A substantially greater spread of healthy volunteer values accounted for the lack of high effect size in the third batch.
Following Z-score transformation of each separate batch and then combining all scores, there was a diagnosis effect [t = 2.1; df = 87; p = .04; .20 (SD = 1.0) versus −.27 (SD = .8)], indicating significant elevations of antibodies towards serotonin. When the Z-score analysis is covaried for sex and age, the result falls below significance levels (p < .07 and p < .051, respectively). No sex or sex by diagnosis effect was evident. Medication status had no effect on plasma antibody levels to serotonin. When patients on medications were excluded from the analysis, patients continued to demonstrate elevations of Z-score converted anti-serotonin antibody levels in comparison to healthy volunteers (t = 2.0; df = 66; p < .05) but this effect failed to reach significance when sex and age were used as covariates. There was no relationship between age and plasma antibody levels, either within the combined group or when each group is considered separately.
There was a strong correlation between anti-serotonin antibody versus serotonin anti-idiotypic antibody measures (Pearson r = .7, df = 88; p < .0001). Supporting the reproducibility of the methodology, there was a high correlation between experiment 1 and experiment 2 measures for anti-serotonin antibodies (r = .84; df = 88, p < .0001) and serotonin anti-idiotypic antibodies (r = .88; df = 88, p < .0001).
DISCUSSION
The data raise the possibility of psychoimmune dysfunction in panic disorder, specifically involving the serotonin system. Elevations of antibodies, directed at the serotonin neurotransmitter itself, as has been reported for fibromyalgia (Klein et al. 1992), as well as serotonin receptors, may be a potential mechanism for interruption of serotonin neurotransmission. Elevations of serotonin anti-idiotypic antibodies were significant when covaried for age and sex whereas elevations of antiserotonin antibodies were only at trend levels when covaried for age and sex. Cerebrospinal fluid examinations for serotonin antibodies may facilitate further characterization of CNS involvement.
Cytokines, such as interleukins, are important messengers (immunotransmitters) linking the immune system to the CNS. As mentioned previously, Brambilla et al. (1994) report elevations of plasma interleukin-1β in panic disorder. Activation of the cytokine cascade could possibly play a role in the generation of elevated levels of anti-serotonin antibodies, which may in turn provide an antigenic stimulus for the production of serotonin anti-idiotypic antibodies, hypotheses requiring further investigation. As a strong positive correlation between the two forms of auto-antibodies against differing elements of the serotonin system was observed, it remains unclear why only serotonin anti-idiotypic antibodies were significantly elevated.
The significant production of serotonin anti-idiotypic antibodies could conceivably be stimulated following exposure of serotonin receptors to immune surveillance during periods of low receptor occupancy by serotonin, thus reflecting 5-HT dysfunction and not necessarily immune dysfunction.
In summary, the report provides evidence for elevations of serotonin anti-idiotypic antibodies in patients with panic disorder. This form of immune disturbance may have implications for serotonin neurotransmission. The data are sufficiently intriguing to warrant replication.
References
Allen AJ, Leonard HL, Swedo SE . (1995): Case study: A new infection-triggered, autoimmune of pediatric OCD and Tourette's syndrome. J Am Acad Child & Adoles Psychiatry 34: (3): 307–311
American Psychiatric Association. (1987): Diagnostic and Statistical Manual of Mental Disorders, Third edition, revised. Washington, DC, American Psychiatric Association.
Andreoli A, Keller SE, Rabaeus M, Zaugg L, Garrone G, Taban C . (1992): Immunity, major depression and panic disorder comorbidity. Biol Psychiatry 31: (9): 896–908
Brambilla F, Bellodi L, Perna G, Battaglia M, Sciuto G, Diaferia G, Petraglia F, Panerai A, Sacerdote P . (1994): Plasma interleukin-1 beta concentrations in panic disorder. Neuropsychobiol 26(1–2):12–22
Coplan JD, Gorman JM, Klein DF . (1992): Serotonin-related function in panic disorder: A critical overview. Neuropsychopharmacology 6: (3): 189–200
Coplan JD, Papp LA, Martinez J, Rosenblum LA, Cooper T, Liebowitz MR, Gorman JM . (1995): Persistence of blunted human growth hormone response to clonidine in panic disorder following fluoxetine treatment. Am J Psychiatry 152: (4): 619–622
Hudson JI, Hudson SH, Pliner LF, Goldenberg DL, Pope HG, Jr . (1985): Fibromyalgia and major affective disorder: A controlled phenomenology and family history study. Am J Psychiatry 142: 441–446
Klein R, Bansch M, Berg PA . (1992): Clinical relevance of antibodies against serotonin and gangliosides in patients with primary fibromyalgia syndrome. Psychoneuroendocrinology 17: (6): 593–598
Maes M, Meltzer HY, Bosmans E, Bergmans R, Vandoolaeghe E, Ranjan R, Desnyder R . (1995): Increased plasma concentrations of interleukin-6, soluble interleukin-6, soluble interleukin-2 and transferrin receptor in major depression. J Affect Disorders 34: (4): 301–309
Marazziti D, Ambrogi F, Vanacore R, Mignani V, Savino M, Palego L, Cassano GB, Akiskal HS . (1992): Immune cell imbalance in major depressive and panic disorders. Neuropsychobiology 26: (1–2): 23–26
Ramesh C, Yeragani VK, Balon R, Pohl R . (1991): A comparative study of immune status in panic disorder patients and controls. Acta Psychiatrica Scandinavica 84 (4): 396–397
Schmidt-Traub S . (1995): The psychoimmunological network of panic disorders, agoraphobia and allergic reactions (German). Therapeutische Umschau (2):123–128
Spitzer RL, Williams JBW, Gibon M, First M . (1990): SCID: User's Guide for the Structured Clinical Interview for DSM-III-R. Washington DC, American Psychiatric Press.
Tamir H, Liu K, Hsiung S, Yu P, Kirchgessner AL, Gershon MD . (1991): Identification of serotonin receptors recognized by anti-idiotypic antibodies. J Neurochem 57: 930–942
Wade PR, Tamir H, Kirchgessner AL, Gershon MD . (1994): Analysis of the role of serotonin in the enteric nervous system using anti-idiotypic antibodies to serotonin receptors. Am J Physiol 266 (Gastrointest Liver Physiol 29): G403–G416
Wasserman NH, Penn AS, Freimuth PI, Treptow N, Wentzel S, Cleveland WL, Erlanger BF . (1982): Anti-idiotypic route to anti-acetylcholine receptor antibodies and myasthenia gravis. Proc Natl Acad Sci USA 79: 4810–4814
Waylonis GW, Heck W . (1992): Fibromyalgia syndrome: New associations. Am J Phys Med Rehabil 71: 343–348
Acknowledgements
Supported in part by NIMH-CRC 30906, NIMH Research Scientist Award NH-00416 (to Dr Gorman), NIMH Research Scientist Development Award MH-00858 (to Dr Papp), and Clinical training grant MH-18641 and NIMH Research Scientist Development Award MH-01039 (to Dr. Coplan).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coplan, J., Tamir, H., Calaprice, D. et al. Plasma Anti-Serotonin and Serotonin Anti-Idiotypic Antibodies Are Elevated in Panic Disorder. Neuropsychopharmacol 20, 386–391 (1999). https://doi.org/10.1016/S0893-133X(98)00130-4
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S0893-133X(98)00130-4
Keywords
This article is cited by
-
Effects of the immunoglobulin/histamine complex on panic disorder concurrent with chronic spontaneous urticaria: a case report
Journal of Medical Case Reports (2023)
-
In vivo Monitoring of Serotonin by Nanomaterial Functionalized Acupuncture Needle
Scientific Reports (2016)
-
Differential responses to anxiogenic drugs in a mouse model of panic disorder as revealed by Fos immunocytochemistry in specific areas of the fear circuitry
Amino Acids (2007)
-
The Management of Anxiety and Depressive Disorders: A Review
International Journal of Mental Health and Addiction (2006)
-
Entstehung und Verlauf der Panikstörung
Psychotherapeut (2005)