Abstract
Background
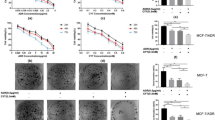
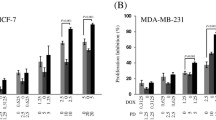
Recent studies have revealed that artesunate (ART) has clear anti-tumor activity, suggesting that it could be a good candidate chemotherapeutic agent. In this study, we researched the inhibitory effect of ART on MCF7 cells and explored the possible mechanisms.
Methods
MTT assay was used to detect the effect of ART on the proliferation of MCF7 cells. Crystal violet staining was used to observe morphological and quantitative changes. Flow cytometry was used to detect the cell cycle of the drug-acting MCF7 cells. In addition, western blotting was used to detect the drug influence on expression of the ATM, phospho-ATM(S1981), H2AX, γH2AX(S139), CHK2 and phospho-CHK2(T68), cdc25C, and phospho-cdc25C(S216).
Results
In the experimental groups, the proliferation of MCF7 cells was inhibited in a dose-dependent manner and the original cell morphology was lost. The number of G2/M phase cells in the experimental groups increased significantly, and the expression of DNA damage response-associated proteins was significantly increased, such as phospho-ATM(S1981), γH2AX(S139), phospho-CHK2(T68), and phospho-cdc25C(S216).
Conclusions
ART can inhibit cell proliferation and promote G2/M arrest in MCF7 cells through ATM activation and the ensuing “ATM-Chk2-Cdc25C” pathway, thus implicating ART as a novel candidate for breast cancer chemotherapy.




Similar content being viewed by others
References
Chisti A, Sharara N, Gupta M, et al. A global cancer project map integrating global cancer statistics to guide international efforts. J Glob Oncol. 2016. https://doi.org/10.1200/JGO.2016.004523.
Zeichner SB, Herna S, Mani A, et al. Survival of patients with de-novo metastatic breast cancer: analysis of data from a large breast cancer-specific private practice, a university-based cancer center and review of the literature. Breast Cancer Res Treat. 2015;153(3):617–24.
Brocato T, Dogra P, Koay EJ, et al. Understanding drug resistance in breast cancer with mathematical oncology. Curr Breast Cancer Rep. 2014;6(2):110–20.
Michaelis M, Kleinschmidt MC, Barth S, et al. Anti-cancer effects of artesunate in a panel of chemoresistant neuroblastoma cell lines. Biochem Pharmacol. 2010;79(2):130–6.
Efferth T. I10 the anticancer activity of the antimalarial artesunate. Biochem Pharmacol. 2017;139:107.
Holien T, Olsen OE, Misund K, et al. Lymphoma and myeloma cells are highly sensitive to growth arrest and apoptosis induced by artesunate. Eur J Haematol. 2013;91(4):339–46.
Rasheed SA, Efferth T, Asangani IA, et al. First evidence that the antimalarial drug artesunate inhibits invasion and in vivo metastasis in lung cancer by targeting essential extracellular proteases. Int J Cancer. 2010;127(6):1475–85.
Li LN, Zhang HD, Yuan SJ, et al. Differential sensitivity of colorectal cancer cell lines to artesunate is associated with expression of beta-catenin and E-cadherin. Eur J Pharmacol. 2008;588(1):1–8.
Zhou X, Sun WJ, Wang WM, et al. Artesunate inhibits the growth of gastric cancer cells through the mechanism of promoting oncosis both in vitro and in vivo. Anticancer Drugs. 2013;24(9):920–7.
Conconi A, Bell B. Molecular biology: the long and short of a DNA-damage response. Nature. 2017;545(7653):165–6.
Morio T. Recent advances in the study of immunodeficiency and DNA damage response. Int J Hematol. 2017;106:357–65.
Liu M, Hu W. Functional role of ATM in the cellular response to DNA damage. Front Chem Sci Eng. 2011;5(2):179–87.
Matt S, Hofmann TG. The DNA damage-induced cell death response: a roadmap to kill cancer cells. Cell Mol Life Sci CMLS. 2016;73(15):2829–50.
Bhaw-Luximon A, Jhurry D. Artemisinin and its derivatives in cancer therapy: status of progress, mechanism of action, and future perspectives. Cancer Chemother Pharmacol. 2017;79(3):451.
Roussel C, Caumes E, Thellier M, et al. Artesunate to treat severe malaria in travellers: review of efficacy and safety and practical implications. J Travel Med. 2017;24(2):1–9.
Rutteman GR, Erich SA, Mol JA, et al. Safety and efficacy field study of artesunate for dogs with non-resectable tumours. Anticancer Res. 2013;33(5):1819–27.
Deeken JF, Wang H, Hartley M, et al. A phase I study of intravenous artesunate in patients with advanced solid tumor malignancies. Cancer Chemother Pharmacol. 2018;81(3):587–96.
Otto T, Sicinski P. Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer. 2017;17(2):93.
Lin X, Gu Y, Tang D. BMI1, ATM and DDR. Oncoscience. 2015;2(8):665–6.
Barba M, Vici P, Pizzuti L, et al. Body mass index modifies the relationship between γ-H2AX, a DNA damage biomarker, and pathological complete response in triple-negative breast cancer. BMC Cancer. 2017;17(1):101.
Manic G, Obrist F, Sistigu A, et al. Trial watch: targeting ATM–CHK2 and ATR–CHK1 pathways for anticancer therapy. Mol Cell Oncol. 2015;2(4):e1012976.
Acknowledgements
This work was supported by Natural Science Foundation of Guangdong Province (2014A03031749), Medical Science and Technology Research Foundation of Guangdong Province (A2017359), Shenzhen Science and Technology Project Foundation (JCYJ20150403094227974), and Longgang Science and Technology Project Foundation (YLWS20140610155624914).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wen, L., Liu, L., Wen, L. et al. Artesunate promotes G2/M cell cycle arrest in MCF7 breast cancer cells through ATM activation. Breast Cancer 25, 681–686 (2018). https://doi.org/10.1007/s12282-018-0873-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-018-0873-5