Abstract
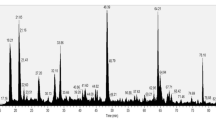
Chromobacterium violaceum, a component of tropical soil microbiota, is an opportunistic pathogenic bacterium that can infect humans and other animals. In addition to identifying a large number of genes that demonstrate the vast biotechnological potential of this bacterium, genome sequencing revealed several virulence factors, including different cytolysins, which can be related to its pathogenicity. Here we confirmed these predictions from genomic analyses by identifying, through mass spectrometry, proteins present in the culture supernatant of C. violaceum that may constitute secreted virulence factors. Among them, we identified a secreted collagenase and the product of a gene with sequence similarity to previously characterized bacterial porins.
Similar content being viewed by others
References
Andrews, N.W. and Whitlow, M.B. 1989. Secretion by Trypanosoma cruzi of a hemolysin active at low pH. Mol. Biochem. Parasitol. 33, 249–256.
Beaufort, N., Corvazier, E., Hervieu, A., Choqueux, C., Dussiot, M., Louedec, L., Cady, A., de Bentzmann, S., Michel, J., and Pidard, D. 2011. The thermolysin-like metalloproteinase and virulence factor LasB from pathogenic Pseudomonas aeruginosa induces anoikis of human vascular cells. Cell. Microbiol. 13, 1149–1167.
Bernardini, M.L., Sanna, M.G., Fontaine, A., and Sansonetti, P.J. 1993. OmpC is involved in invasion of epithelial cells by Shigella flexneri. Infect. Immun. 61, 3625–3635.
Brito, C.F.A., Carvalho, C.M.B., Santos, F.R., Gazzinelli, R.T., Oliveira, S.C., Azevedo, V., and Teixeira, S.M.R. 2004. Chromobacterium violaceum genome: molecular mechanism associated with pathogenicity. Genet. Mol. Res. 3, 148–161.
Brumbach, K.C., Eason, B.D., and Anderson, L.K. 2007. The serratia-type hemolysin of Chromobacterium violaceum. FEMS Microbiol. Lett. 267, 243–250.
Chatfield, S.N., Dorman, C.J., Hayward, C., and Dougan, G. 1991. Role of ompR-dependent genes in Salmonella typhimurium virulence: mutants deficient in both OmpC and OmpF are attenuated in vivo. Infect. Immun. 59, 449–452.
Chattopadhyay, A., Kumar, V., Bhat, N., and Rao, P. 2002. Chromobacterium violaceum infection: a rare but frequently fatal disease. J. Pediatr. Surg. 37, 108–110.
Cluss, R.G., Silverman, D.A., and Stafford, T.R. 2004. Extracellular secretion of the Borrelia burgdorferi Oms28 porin and Bgp, a glycosaminoglycan binding protein. Infect. Immun. 72, 6279–6286.
Durán, N. and Menck, C.F. 2001. Chromobacterium violaceum: a review of pharmacological and industrial perspectives. Crit. Rev. Microbiol. 27, 201–222.
Fukuda, Y. and Schuetz, J.D. 2012. ABC transporters and their role in nucleoside and nucleotide drug resistance. Biochem. Pharmacol. 83, 1073–1083.
Han, X.Y., Han, F.S., and Segal, J. 2008. Chromobacterium haemolyticum sp. nov., a strongly haemolytic species. Int. J. Syst. Evol. Microbiol. 58, 1398–1403.
Hoshino, T. 2011. Violacein and related tryptophan metabolites produced by Chromobacterium violaceum: biosynthetic mechanism and pathway for construction of violacein core. Appl. Microbiol. Biotechnol. 91, 1463–1475.
Khan, M.A., Bouzari, S., Ma, C., Rosenberger, C.M., Bergstrom, K.S.B., Gibson, D.L., Steiner, T.S., and Vallance, B.A. 2008. Flagellin-dependent and independent inflammatory responses following infection by enteropathogenic Escherichia coli and Citrobacter rodentium. Infect. Immun. 76, 1410–1422.
Lima-Bittencourt, C.I., Costa, P.S., Barbosa, F.A.R., Chartone-Souza, E., and Nascimento, A.M.A. 2011. Characterization of a Chromobacterium haemolyticum population from a natural tropical lake. Lett. Appl. Microbiol. 52, 642–650.
Miller, D.P., Blevins, W.T., Steele, D.B., and Stowers, M.D. 1988. A comparative study of virulent and avirulent strains of Chromobacterium violaceum. Can. J. Microbiol. 34, 249–255.
Miyoshi, S. and Shinoda, S. 2000. Microbial metalloproteases and pathogenesis. Microbes Infect. 2, 91–98.
Mougous, J.D., Cuff, M.E., Raunser, S., Shen, A., Zhou, M., Gifford, C.A., Goodman, A.L., Joachimiak, G., Ordoñez, C.L., Lory, S., and et al. 2006. A virulence locus of Pseudomonas aeruginosa encodes a protein secretion apparatus. Science 312, 1526–1530.
Nikaido, H. 2003. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev. 67, 593–656.
Osipiuk, J., Xu, X., Cui, H., Savchenko, A., Edwards, A., and Joachimiak, A. 2011. Crystal structure of secretory protein Hcp3 from Pseudomonas aeruginosa. J. Struct. Funct. Genomics 12, 21–26.
Piddock, L.J. 2006. Multidrug-resistance efflux pumps — not just for resistance. Nat. Rev. Microbiol. 4, 629–636.
Pukatzki, S., Ma, A.T., Sturtevant, D., Krastins, B., Sarracino, D., Nelson, W.C., Heidelberg, J.F., and Mekalanos, J.J. 2006. Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl. Acad. Sci. USA 103, 1528–1533.
Rolhion, N., Barnich, N., Claret, L., and Darfeuille-Michaud, A. 2005. Strong decrease in invasive ability and outer membrane vesicle release in Crohn’s disease-associated adherent-invasive Escherichia coli strain LF82 with the yfgL gene deleted. J. Bacteriol. 187, 2286–2296.
Vasconcelos, A.T.R. and the Brazilian National Genome Project Consortium. 2003. Complete genome sequence of Chromobacterium violaceum reveals remarkable and exploitable bacterial adaptability. Proc. Natl. Acad. Sci. USA 100, 11660–11665.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Castro-Gomes, T., Cardoso, M.S., DaRocha, W.D. et al. Identification of secreted virulence factors of Chromobacterium violaceum . J Microbiol. 52, 350–353 (2014). https://doi.org/10.1007/s12275-014-3202-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-014-3202-5