Abstract
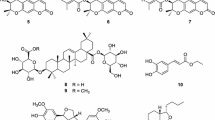
A new phenylpropanoid ester mixture, (E)-geranylferulic acid (1a) and (Z)-geranylferulic acid (1b), along with 13 known compounds, [6]-gingerol (2), [8]-gingerol (3), [10]-gingerdione (4), 1-dehydro-[6]-gingerdione (5), 1-dehydro-[8]-gingerdione (6), [6]-paradol (7), [8]-paradol (8), [6]-gingeroldiacetate (9), 6-hydroxy-[6]-shogaol (10), galanolactone (11), trans-®-sesquiphellandrol (12), trans-sesquipiperitol (13), and 4α,5β-dihydroxybisabola-2,10-diene (14) were isolated from ethanol extract of Zingiber officinale. Their structures were determined based on the spectroscopic (1D, 2D-NMR and MS) and chemical evidence. All of the isolates were evaluated for their potential to inhibit LPS-induced production of nitric oxide in murine macrophage RAW264.7 cells. Compounds 1–12 were found to inhibit nitric oxide production with IC50 values ranging from 5.5 to 28.5 μM.
Similar content being viewed by others
References
Alderton, W. K., Cooper, C. E., and Knowles, R. G., Nitric oxide synthases: structure, function and inhibition. Biochem. J., 357, 593–615 (2001).
Bednarczyk, A. A., Galetto, W. G., and Kramer, A., cis- and trans-β-Sesquiphellandrol. Two new sesquiterpene alcohols from oil of ginger (Zingiber officinale, Roscoe). J. Agric. Food Chem., 23, 499–501 (1975).
Bohlmann, F., Tsankova, E., and Jakupovic, J., Sesquiterpenes and acetylenes from Argyranthemum adauctum ssp. jacobaeifolium. Phytochemistry, 23, 1103–1104 (1984).
But, P. P. H., Kimura, T., Guo, J.-X., and Sung, C. K., International collation of traditional and folk medicine. World Scientific, Singapore, p. 400, (1997).
Cool, L. G., Sesquiterpene alcohols from foliage of Fitzroya cupressoldes. Phytochemistry, 42, 1015–1019 (1996).
Delazar, A., Shoeb, M., Kumarasamy, Y., Byres, M., Nahar, L., Modarresi, M., and Sarker, S. D., Two bioactive ferulic acid derivatives from Eremostachys glabra. DARU, 12, 49–53 (2004).
Denyer, C. L., Jackson, P., and Loakes, D. M., Isolation of antirhinoviral sesquiterpenes from ginger. J. Nat. Prod., 57, 658–662 (1994).
Dugasani, S., Pichika, M. R., Nadarajah, V. D., Balijepalli, M. K., Tandra, S., and Korlakunta, J. N., Comparative antioxidant and anti-inflammatory effects of [6]-gingerol, [8]-gingerol, [10]-gingerol and [6]-shogaol. J. Ethnopharmacol., 127, 515–520 (2010).
ElRokh, E. S. M., Yassin, N. A. Z., El-Shenawy, S. M. A., and Ibrahim, B. M. M., Antihypercholesterolaemic effect of ginger rhizome (Zingiber officinale) in rats. Inflammopharmacology, 18, 309–315 (2010).
He, X. G., Bernart, M. W., Lian, L. Z., and Lin, L. Z., Highperformance liquid chromatography-electrospray mass spectrometric analysis of pungent constituents of ginger. J. Chromatogr. A, 796, 327–334 (1998).
Hong, S. S., Lee, S. A., Han, X. H., Hwang, J. S., Lee, C., Lee, D., Hong, J. T., Kim, Y., Lee, H., and Hwang, B. Y., ent-Kaurane diterpenoids from Isodon japonicus. J. Nat. Prod., 71, 1055–1058 (2008).
Hsu, Y. L., Chen, C. Y., Hou, M. F., Tsai, E. M., Jong, Y. J., Hung, C. H., and Kuo, P. L., 6-Dehydrogingerdione, an active constituent of dietary ginger, induces cell cycle arrest and apoptosis through reactive oxygen species/c-Jun N-terminal kinase pathways in human breast cancer cells. Mol. Nutr. Food Res., 54, 1307–1317 (2010).
Jiang, H., Solyom, A. M., Timmermann, B. N., and Gang, D. R., Characterization ofgingerol-related compounds in ginger rhizome (Zingiber officinale Rosc.) by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrum., 19, 2957–2964 (2005).
Kikuzaki, H., Tsai, S. M., and Nakatani, N., Gingerdiol related compounds from the Rhizomes of Zingiber officinale. Phytochemistry, 31, 1783–1786 (1992).
Kim, J. S., Lee, S. I., Park, H. W., Yang, J. H., Shin, T. Y., Kim, Y. C., Baek, N. I., Kim, S. H., Choi, S. U., Kwon, B. M., Leem, K. H., Jung, M. Y., and Kim, D. K., Cytotoxic components from the dried rhizomes of Zingiber officinale Roscoe. Arch. Pharm. Res., 31, 415–418 (2008).
Kim, S. O., Kundu, J. K., Shin, Y. K., Park, J. H., Cho, M. H., Kim, T. Y., and Surh, Y. J., [6]-Gingerol inhibits COX-2 expression by blocking the activation of p38 MAP kinase and NF-κB in phorbol ester-stimulated mouse skin. Oncogene, 24, 2558–2567 (2005).
Kiuchi, F., Shibuya, M., and Sankawa, U., Inhibitors of prostaglandin biosynthesis from ginger. Chem. Pharm. Bull., 30, 754–757 (1982).
Kuo, P. C., Damu, A. G., Cherng, C. Y., Jeng, J. F., Teng, C. M., Lee, E. J., and Wu, T. S., Isolation of a naturalantioxidant, dehydrozingerone from Zingiber officinale and synthesis of its analogues for recognition of effective antioxidant and antityrosinase agents. Arch. Pharm. Res., 28, 518–528 (2005).
Lai, Y. Y., Huang, L. J., Lin, H. C., Wu, T. S., Teng, C. M., and Kuo, S. C., Synthesis of gingerdione derivatives as potent antiplatelet agents. Chin. Pharm. J., 54, 259–269 (2002).
Ma, J. P., Jin, X. L., Yang, L., and Liu, Z. L., Two new diarylheptanoids from the rhizomes of Zingiber officinale. Chin. Chem. Lett., 15, 1306–1308 (2004).
MacMicking, J., Xie, Q. W., and Nathan, C., Nitric oxide and macrophage function. Annu. Rev. Immunol., 15, 323–350 (1997).
Morita H. and Itokawa, H., Cytotoxic and antifungal diterpenes from the seeds of Alpinia galangal. Planta Med., 54, 117–120 (1988).
Sharma, J. N., Srivastava, K. C., and Gan E. K., Suppressive effects of eugenol and ginger oil on arthritic rats. Pharmacology, 49, 314–318 (1994)
Singh, G., Kapoor, I. P. S., Singh, P., Heluani, C. S., Lampasona, M. P., and Catalan, C. A. N., Chemistry, antioxidant and antimicrobial investigations on essential oil and oleoresins of Zingiber officinale. Food Chem. Toxicol., 46, 3295–3302 (2008).
Tao, Q. F., Xu, Y., Lam, R. Y. Y., Schneider, B., Dou, H., Leung, P. S., Shi, S. Y., Zhou, C. X., Yang, L. X., Zhang, R. P., Xiao, Y. C., Wu, X., Stockigt, J., Zeng, S., Cheng, C. H. K., and Zhao, Y., Diarylheptanoids and a monoterpenoid from rhizomes of Zingiber officinale: antioxidant and cytoprotective properties. J. Nat. Prod., 71, 12–17 (2008).
Vishwakarma, S. L., Pal, S. C., Kasture, V. S., and Kasture, S. B., Anxiolytic and antiemetic activity of Zingiber officinale. Phytother. Res., 16, 621–626 (2002).
Wei, Q. Y., Ma, J. P., Cai, Y. J., Yang, L., and Liu, Z. L., Cytotoxic and apoptotic activities of diarylheptanoids and gingerol-related compounds from the rhizome of Chinese ginger. J. Ethnopharmacol., 102, 177–184 (2005).
Yang, E. J., Yim, E. Y., Song, G., Kim, G. O., and Hyun, C. G., Inhibition of nitric oxide production in lipopolysaccharideactivated RAW 264.7macrophages by Jeju plant extracts. Interdisc. Toxicol., 2, 245–249 (2009).
Yoon, W. J., Ham, Y. M., Kim, K. N., Park, S. Y., Lee, N. H., Hyun, C. G., and Lee, W. J., Anti-inflammatory activity of brown alga Dictyota dichotoma in murine macrophage RAW 264.7 cells. J. Med. Plant Res., 3, 1–8 (2009).
Zdero, C., Bohlmann, F., and Scott, R., Germacranolides, guaianolides and eudesmanolides from Greenmaniella resinosa. Phytochemistry, 26, 1999–2006 (1987).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, S.S., Oh, J.S. Phenylpropanoid ester from Zingiber officinale and their inhibitory effects on the production of nitric oxide. Arch. Pharm. Res. 35, 315–320 (2012). https://doi.org/10.1007/s12272-012-0211-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0211-y