Abstract
Introduction
Recent medical investigations suggest that HLA-G, due to its tolerogenic properties, can be used as a biomarker in the diagnosis, treatment, and prognosis of different neoplasms. This observational prospective pilot study aims at detecting sHLA-G in the serum and saliva of patients diagnosed with colorectal cancer (CRC). For this purpose, we compared the expression of sHLA-G from patients with a control sample from a healthy population.
Materials and methods
Using the specific enzyme-linked immunosorbent assay (ELISA) method, the expression of sHLA-G in the serum and saliva samples from patients affected by CRC (n = 20) and in a control sample (n = 10) were analyzed.
Results
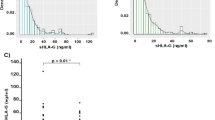
The data showed that in patients with CRC, salivary sHLA-G values were significantly higher than in the control group (18.84 U/ml versus 6.3 U/ml, p = 0.036). In addition, higher levels of sHLA-G were observed in the saliva of patients with CRC in more advanced stages, compared with patients in early stages (24.2 U/ml vs. 8.1 U/ml, p = 0.019). A significant correlation was observed between the concentration of sHLA-G in the serum and saliva of the analyzed samples (Spearman correlation 0.7, p = 0.004).
Conclusions
This study demonstrates, for the first time, the possibility of detecting sHLA-G in the saliva of patients with CRC, resulting in a less invasive alternative to venipuncture. Likewise, we propose that sHLA-G could be an attractive molecular target based on its significant high levels in advanced stages.





Similar content being viewed by others
References
World Health Organization. Global status report on Noncommunicable Diseases 2014. Geneva: WHO; 2014.
Lorusso G, Ruegg C. New insights into the mechanisms of organ-specific breast cancer metastasis. Semin Cancer Biol. 2012;22:226–33.
Chen DS, Mellman I. Elements of cancer immunity and the cancer-immune set point. Nature. 2017;541:321–30.
International Agency for Research on Cancer: GLOBOCAN 2018: Estimated cancer incidence, mortality and prevalence worldwide in 2018-Cancer fact sheets; Available from: https://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx. Accessed 14 Mar 2019.
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69.
Favoriti P, Carbone G, Greco M, Pirozzi F, Pirozzi RE, Corcione F. Worldwide burden of colorectal cancer: a review. Updates Surg. 2016;68:7–11.
Chumbley G, King A, Holmes N, Loke YW. In situ hybridization and northern blot demonstration of HLA-G mRNA in human trophoblast populations by locus-specific oligonucleotide. Hum Immunol. 1993;37:17–22.
Yao YQ, Barlow DH, Sargent IL. Differential expression of alternatively spliced transcripts of HLA-G in human preimplantation embryos and inner cell masses. J Immunol. 1950;175:8379–85.
Shiroishi M, Tsumoto K, Amano K, Shirakihara Y, Colonna M, Braud VM, et al. Human inhibitory receptors Ig-like transcript 2 (ILT2) and ILT4 compete with CD8 for MHC class I binding and bind preferentially to HLA-G. Proc Nat Acad Sci USA. 2003;100:8856–61.
Shiroishi M, Kuroki K, Rasubala L, Tsumoto K, Kumagai I, Kurimoto E, et al. Structural basis for recognition of the nonclassical MHC molecule HLA-G by the leukocyte Ig-like receptor B2 (LILRB2/LIR2/ILT4/CD85d). Proc Nat Acad Sci USA. 2006;103:16412–7.
HLA Nomenclature @ hla.alleles.org. https://hla.alleles.org/nomenclature/stats.html. Accessed 9 Mar 2016
Ishitani A, Geraghty DE. Alternative splicing of HLA-G transcripts yields proteins with primary structures resembling both class I and class II antigens. Proc Nat Acad Sci USA. 1992;89:3947–51.
Le Bouteiller P, Blaschitz A. The functionality of HLA-G is emerging. Immunol Rev. 1999;167:233–44.
HoWangYin K-Y, Loustau M, Wu J, Alegre E, Daouya M, Caumartin J, et al. Multimeric structures of hla-g isoforms function through differential binding to lilrb receptors. Cell Mol Life Sci. 2012;69:4041–9.
Pan YQ, Ruan YY, Peng JB, Han QY, Zhang X, Lin A, Yan WH. Diagnostic significance of soluble human leukocyte antigen-G for gastric cancer. Hum Immunol. 2016;77:317–24.
Ben Amor A, Beauchemin K, et al. Leukocyte antigen G polymorphism and expression are associated with an increased risk of non-small-cell lung cancer and advanced disease stage. PLoS ONE. 2016;11:e0161210.
Lin A, Zhu C, Chen H, et al. Clinical relevance and functional implications for human leucocyte antigen-g expression in non-small-cell lung cancer. J Cell Mol Med. 2010;14:2318–9.
Lin A, Yan W. Human leuokocite antigen G (HLA-G) expression in cancers: roles in inmune evasion, metastasis and targer for therapy. Mol Med. 2015;21:782–91.
Loumagne L, Baudhuin J, Favier B, Montespan F, Carosella ED, Rouas-Freiss N. In vivo evidence that secretion of HLA-G by immunogenic tumor cells allows their evasion from immunosurveillance. Int J Cancer. 2014;135:2107–17.
Fridman WH, Pagès F, Sautès-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer. 2012;12:298–306.
Menier C, Rouas-Freiss N, Carosella ED. The HLA-G non-classical MHC class I molecule is expressed in cancer with poor prognosis. Implications in tumour escape from immune system and clinical applications. Atlas Genet Cytogenet Oncol Haematol. 2009;6:879–900.
Morandi F, Rizzo R, Fainardi E, Rouas-Freiss N, and Pistoia V. Recent advances in our understanding of HLA-G biology: lessons from a wide spectrum of human diseases. J Inmunol Res. 2016;4326495. https://doi.org/10.1155/2016/4326495.
Vinay DS, Ryan EP, Pawelec G, Talib WH, Stagg J, Elkord E, Lichtor T, Decker WK, Whelan RL, Kumara HM, Signori E, Honoki K, Georgakilas AG, et al. Inmune evasión in cáncer. Mechanistic basis and therapeutic strategies. Semin Cancer Biol. 2015;35:S185–S198198.
Yan WH. Human leukocyte antigen-G in cancer: are they clinically relevant? Cancer Lett. 2011;311:123–30.
Carosella ED, Rouas-Freiss N, Tronik-Le Roux D, Moreau P, LeMaoult J. HLA-G: an immune checkpoint molecule. Adv Immunol. 2015;127:33–144.
Amiot L, Ferrone S, Grosse-Wilde H, Seliger B. Biology of HLA-G in cancer: a candidate molecule for therapeutic intervention? Cell Mol Life Sci. 2011;68:417–31.
Ye S-R, Yang H, Li K, et al. Human leukocyte antigen G expression: as a significant prognostic indicator for patients with colorectal cancer. Mod Pathol. 2007;20:375–83.
Nosho K, Baba Y, Tanaka N, Shima K, Hayashi M, Meyerhardt JA, Giovannucci E, Dranoff G, Fuchs CS, Ogino S. Tumour-infiltrating T-cell subsets, molecular changes in colorectal cancer and prognosis: cohort study and literature review. J Pathol. 2010;222:350–66.
Pourhoseingholi MA, Fazeli Z, Ashtari S, Bavand-Pour FS. Mortality trends of gastrointestinal cancers in Iranian population. Gastroenterol Hepatol Bed Bench. 2013;6:S52–S5757.
Farjadian S, Tabebordbar M, Mokhtari M, et al. HLA-G Expression in tumor tissues and soluble HLA-G. Plasma levels in patients with gastrointestinal cancer. Asian Pac J Cancer Prev. 2018;19:2731–5.
Zhu CB, Wang CX, et al. Serum sHLA-G levels: a useful indicator in distinguishing colorectal cancer from benign colorectal diseases. Int J Cancer. 2011;128:617–22.
Gonçalves AS, Mosconi C, Jaeger F, et al. Overexpression of immunomodulatory mediators in oral precancerous lesions. Hum Immunol. 2017;78:752–7.
Gonçalves AS, Arantes DA, Bernardes VF, et al. Immunosuppressive mediators of oral squamous cell carcinoma in tumour samples and saliva. Hum Immunol. 2015;76:52–8.
Ravaglia G, Forti P, Maioli F, Bastagli L, Facchini A, Mariani E, Savarino L, Sassi S, Cucinotta D, Lenaz G. Effect of micronutrient status on natural killer cell immune function in healthy free-living subjects aged %3e 90 years. Am J Clin Nutr. 2000;71:590–8.
Grubeck-Loebenstein B. Changes in the aging immune system. Biologicals. 1997;25:205–8.
Acknowledgements
We are grateful for the collaboration of the study participants, the Department of Clinical Medicine of the Miguel Hernández University, as well as the nursing team, administrative staff, and physicians of the Medical Oncology Service of the Sant Joan d'Alacant Hospital. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no confilct of interest with respect to the publication of this document.
Ethical approval
The study was approved by the Ethics Committee of the Department of Health of the University Hospital of Sant Joan d'Alacant (Committee code: 17/332). All procedures performed in the study were in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lázaro-Sánchez, A.D., Salces-Ortiz, P., Velásquez, L.I. et al. HLA-G as a new tumor biomarker: detection of soluble isoforms of HLA-G in the serum and saliva of patients with colorectal cancer. Clin Transl Oncol 22, 1166–1171 (2020). https://doi.org/10.1007/s12094-019-02244-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02244-2