Abstract
Background
MicroRNAs (miRs) have been implicated in the etiology of various human cancers. The aim of this study was to investigate the association of the expression of three members—miR 200a, miR 200b, and miR 200c belonging to the miR-200 family with clinicopathological characteristics and their impact on the progression of epithelial ovarian cancer (EOC).
Materials and methods
Total RNA from serum was isolated by Trizol method, polyadenylated, and reverse transcribed into cDNA. Expression levels of miR-200a, miR-200b, and miR-200c were detected by using miRNA qRT-PCR. We measured miR expression in 70 serum samples of EOC patients with matched controls using U6 snRNA as a reference. Levels of miR expression was compared with distinct clinicopathological features.
Results
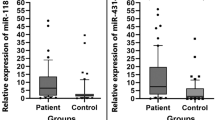
Expression of miR-200a was found to be greater than six-fold (p = 0.01), miR-200b and miR-200c greater than three-fold (p = 0.01) in comparison with matched normal controls. Association of miRNA expression with clinicopathological factors and progression was statistically evaluated. The expression levels of miR-200a and miR-200c were found to be significantly associated with disease progression (p = 0.04 and p < 0.001, respectively). miR-200a overexpression was found be associated with tumor histology and stage. Patients with lymph node metastasis showed significant elevation of miR-200c (p = 0.006). The AUC in ROC curve also indicated that serum levels of miR-200a and miR-200c might be worthwhile as a diagnostic tool in the near future.
Conclusion
Our findings suggest that miR-200a, miR-200b, and miR-200c overexpressions are associated with the aggressive tumor progression and be recognized as reliable markers to predict the prognosis and survival in EOC patients.



Similar content being viewed by others
References
Coward JI, Middleton K, Murphy F. New perspectives on targeted therapy in ovarian cancer. Int J Womens Health. 2015;7:189–203.
Hoyer T, Bekkers R, Gooszen H, Massuger L, Rovers M, Grutters JPC. Cost-effectiveness of early-initiated treatment for advanced-stage epithelial ovarian cancer patients: a modeling study. Int J Gynecol Cancer. 2014;24(1):75–84.
Toiyama Y, Tanaka K, Inoue Y, Mohri Y, Kusunoki M. Circulating cell-free microRNAs as biomarkers for colorectal cancer. Surg Today. 2015. doi:10.1007/s00595-015-1138-y.
Sarode SC, Sarode GS, Patil S. MicroRNA in oral cancer research: future prospects. J Contemp Dent Pract. 2014;15(5):i–ii.
Ning X, Shi Z, Liu X, Zhang A, Han L, Jiang K, et al. DNMT1 and EZH2 mediated methylation silences the microRNA-200b/a/429 gene and promotes tumor progression. Cancer Lett. 2015;359(2):198–205.
Dhayat SA, Mardin WA, Köhler G, Bahde R, Vowinkel T, Wolters H, et al. The microRNA-200 family—a potential diagnostic marker in hepatocellular carcinoma? J Surg Oncol. 2014;110(4):430–8.
Hur K, Toiyama Y, Takahashi M, Balaguer F, Nagasaka T, Koike J, et al. MicroRNA-200c modulates epithelial-to-mesenchymal transition (EMT) in human colorectal cancer metastasis. Gut. 2013;62(9):1315–26.
Bojmar L, Karlsson E, Ellegård S, Olsson H, Björnsson B, Hallböök O, et al. The role of microRNA-200 in progression of human colorectal and breast cancer. PLoS One. 2013;8(12):e84815.
Kan CWS, Hahn MA, Gard GB, Maidens J, Huh JY, Marsh DJ, et al. Elevated levels of circulating microRNA-200 family members correlate with serous epithelial ovarian cancer. BMC Cancer. 2012;12:627.
Pacurari M, Addison JB, Bondalapati N, Wan Y-W, Luo D, Qian Y, et al. The microRNA-200 family targets multiple non-small cell lung cancer prognostic markers in H1299 cells and BEAS-2B cells. Int J Oncol. 2013;43(2):548–60.
Kurashige J, Kamohara H, Watanabe M, Hiyoshi Y, Iwatsuki M, Tanaka Y, et al. MicroRNA-200b regulates cell proliferation, invasion, and migration by directly targeting ZEB2 in gastric carcinoma. Ann Surg Oncol. 2012;19(Suppl 3):S656–64.
Minn Y-K, Lee DH, Hyung WJ, Kim JE, Choi J, Yang S-H, et al. MicroRNA-200 family members and ZEB2 are associated with brain metastasis in gastric adenocarcinoma. Int J Oncol. 2014;45(6):2403–10.
Mei Q, Li F, Quan H, Liu Y, Xu H. Busulfan inhibits growth of human osteosarcoma through miR-200 family microRNAs in vitro and in vivo. Cancer Sci. 2014;105(7):755–62.
Zhou X, Wang Y, Shan B, Han J, Zhu H, Lv Y, et al. The downregulation of miR-200c/141 promotes ZEB1/2 expression and gastric cancer progression. Med Oncol Northwood Lond Engl. 2015;32(1):428.
Katz B, Reis ST, Viana NI, Morais DR, Moura CM, Dip N, et al. Comprehensive study of gene and microRNA expression related to epithelial–mesenchymal transition in prostate cancer. PLoS One. 2014;9(11):e113700.
Cao Q, Lu K, Dai S, Hu Y, Fan W. Clinicopathological and prognostic implications of the miR-200 family in patients with epithelial ovarian cancer. Int J Clin Exp Pathol. 2014;7(5):2392–401.
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G, et al. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008;10(5):593–601.
Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial–mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem. 2008;283(22):14910–4.
Park S-M, Gaur AB, Lengyel E, Peter ME. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008;22(7):894–907.
Burk U, Schubert J, Wellner U, Schmalhofer O, Vincan E, Spaderna S, et al. A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Rep. 2008;9(6):582–9.
Bracken CP, Gregory PA, Kolesnikoff N, Bert AG, Wang J, Shannon MF, et al. A double-negative feedback loop between ZEB1-SIP1 and the microRNA-200 family regulates epithelial–mesenchymal transition. Cancer Res. 2008;68(19):7846–54.
Kim TH, Kim YK, Kwon Y, Heo JH, Kang H, Kim G, et al. Deregulation of miR-519a, 153, and 485-5p and its clinicopathological relevance in ovarian epithelial tumours. Histopathology. 2010;57(5):734–43.
Zhang L, Volinia S, Bonome T, Calin GA, Greshock J, Yang N, et al. Genomic and epigenetic alterations deregulate microRNA expression in human epithelial ovarian cancer. Proc Natl Acad Sci USA. 2008;105(19):7004–9.
Nam EJ, Yoon H, Kim SW, Kim H, Kim YT, Kim JH, et al. MicroRNA expression profiles in serous ovarian carcinoma. Clin Cancer Res. 2008;14(9):2690–5.
Iorio MV, Visone R, Di Leva G, Donati V, Petrocca F, Casalini P, et al. MicroRNA signatures in human ovarian cancer. Cancer Res. 2007;67(18):8699–707.
Dahiya N, Sherman-Baust CA, Wang T-L, Davidson B, Shih I-M, Zhang Y, et al. MicroRNA expression and identification of putative miRNA targets in ovarian cancer. PLoS One. 2008;3(6):e2436.
Yang H, Kong W, He L, Zhao J-J, O’Donnell JD, Wang J, et al. MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res. 2008;68(2):425–33.
Wyman SK, Parkin RK, Mitchell PS, Fritz BR, O’Briant K, Godwin AK, et al. Repertoire of microRNAs in epithelial ovarian cancer as determined by next generation sequencing of small RNA cDNA libraries. PLoS One. 2009;4(4):e5311.
Chao A, Lin C-Y, Lee Y-S, Tsai C-L, Wei P-C, Hsueh S, et al. Regulation of ovarian cancer progression by microRNA-187 through targeting disabled homolog-2. Oncogene. 2012;31(6):764–75.
Resnick KE, Alder H, Hagan JP, Richardson DL, Croce CM, Cohn DE. The detection of differentially expressed microRNAs from the serum of ovarian cancer patients using a novel real-time PCR platform. Gynecol Oncol. 2009;112(1):55–9.
Marchini S, Cavalieri D, Fruscio R, Calura E, Garavaglia D, Fuso Nerini I, et al. Association between miR-200c and the survival of patients with stage I epithelial ovarian cancer: a retrospective study of two independent tumour tissue collections. Lancet Oncol. 2011;12(3):273–85.
Peng D-X, Luo M, Qiu L-W, He Y-L, Wang X-F. Prognostic implications of microRNA-100 and its functional roles in human epithelial ovarian cancer. Oncol Rep. 2012;27(4):1238–44.
Toiyama Y, Hur K, Tanaka K, Inoue Y, Kusunoki M, Boland CR, et al. Serum miR-200c is a novel prognostic and metastasis-predictive biomarker in patients with colorectal cancer. Ann Surg. 2014;259(4):735–43.
Wang H-Y, Shen J, Jiang C-P, Liu B-R. How to explain the contradiction of microRNA 200c expression and survival in solid tumors? A meta-analysis. Asian Pac J Cancer Prev. 2014;15(8):3687–90.
Xue X, Zhang Y, Zhi Q, Tu M, Xu Y, Sun J, et al. MiR200-upregulated Vasohibin 2 promotes the malignant transformation of tumors by inducing epithelial–mesenchymal transition in hepatocellular carcinoma. Cell Commun Signal. 2014;12(1):62.
Gregory PA, Bracken CP, Bert AG, Goodall GJ. MicroRNAs as regulators of epithelial–mesenchymal transition. Cell Cycle. 2008;7(20):3112–8.
Le MTN, Hamar P, Guo C, Basar E, Perdigão-Henriques R, Balaj L, et al. miR-200-containing extracellular vesicles promote breast cancer cell metastasis. J Clin Invest. 2014;124(12):5109–28.
Tuomarila M, Luostari K, Soini Y, Kataja V, Kosma V-M, Mannermaa A. Overexpression of microRNA-200c predicts poor outcome in patients with PR-negative breast cancer. PLoS One. 2014;9(10):e109508.
Kinose Y, Sawada K, Nakamura K, Kimura T. The role of microRNAs in ovarian cancer. BioMed Res Int. 2014;2014:249393.
Zhu C-L, Gao G-S. miR-200a overexpression in advanced ovarian carcinomas as a prognostic indicator. Asian Pac J Cancer Prev. 2014;15(20):8595–601.
Tejero R, Navarro A, Campayo M, Viñolas N, Marrades RM, Cordeiro A, et al. miR-141 and miR-200c as markers of overall survival in early stage non-small cell lung cancer adenocarcinoma. PLoS One. 2014;9(7):e101899.
Adam L, Zhong M, Choi W, Qi W, Nicoloso M, Arora A, et al. miR-200 expression regulates epithelial-to-mesenchymal transition in bladder cancer cells and reverses resistance to epidermal growth factor receptor therapy. Clin Cancer Res. 2009;15(16):5060–72.
Ladeiro Y, Couchy G, Balabaud C, Bioulac-Sage P, Pelletier L, Rebouissou S, et al. MicroRNA profiling in hepatocellular tumors is associated with clinical features and oncogene/tumor suppressor gene mutations. Hepatology. 2008;47(6):1955–63.
Ali S, Ahmad A, Banerjee S, Padhye S, Dominiak K, Schaffert JM, et al. Gemcitabine sensitivity can be induced in pancreatic cancer cells through modulation of miR-200 and miR-21 expression by curcumin or its analogue CDF. Cancer Res. 2010;70(9):3606–17.
Wagner A, Mayr C, Bach D, Illig R, Plaetzer K, Berr F, et al. MicroRNAs associated with the efficacy of photodynamic therapy in biliary tract cancer cell lines. Int J Mol Sci. 2014;15(11):20134–57.
Lee J-W, Park Y-A, Choi J-J, Lee YY, Kim C-J, Choi C, et al. The expression of the miRNA-200 family in endometrial endometrioid carcinoma. Gynecol Oncol. 2011;120(1):56–62.
Braun J, Hoang-Vu C, Dralle H, Hüttelmaier S. Downregulation of microRNAs directs the EMT and invasive potential of anaplastic thyroid carcinomas. Oncogene. 2010;29(29):4237–44.
Koutsaki M, Spandidos DA, Zaravinos A. Epithelial–mesenchymal transition-associated miRNAs in ovarian carcinoma, with highlight on the miR-200 family: prognostic value and prospective role in ovarian cancer therapeutics. Cancer Lett. 2014;351(2):173–81.
Xu S, Xu P, Wu W, Ou Y, Xu J, Zhang G, et al. The biphasic expression pattern of miR-200a and E-cadherin in epithelial ovarian cancer and its correlation with clinicopathological features. Curr Pharm Des. 2014;20(11):1888–95.
Ye X, Jiang F, Li Y, Mu J, Si L, Wang X, et al. Glabridin attenuates the migratory and invasive capacity of breast cancer cells by activating microRNA-200c. Cancer Sci. 2014;105(7):875–82.
Mostert B, Sieuwerts AM, Martens JWM, Sleijfer S. Diagnostic applications of cell-free and circulating tumor cell-associated miRNAs in cancer patients. Expert Rev Mol Diagn. 2011;11(3):259–75.
Mo M-H, Chen L, Fu Y, Wang W, Fu SW. Cell-free Circulating miRNA biomarkers in cancer. J Cancer. 2012;3:432–48.
Acknowledgments
We gratefully acknowledge the assistance of University Grants Commission for supporting this work.
Conflict of interest
None.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zuberi, M., Mir, R., Das, J. et al. Expression of serum miR-200a, miR-200b, and miR-200c as candidate biomarkers in epithelial ovarian cancer and their association with clinicopathological features. Clin Transl Oncol 17, 779–787 (2015). https://doi.org/10.1007/s12094-015-1303-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1303-1