Abstract
α-Synuclein (α-syn) has been recognized to induce neuroinflammation and to disturb nerve repair process in Parkinson’s disease. However, the potential mechanisms underlying α-syn-induced impairment of adult neurogenesis remain unclear. In the present study, A53T mutant α-synuclein transgenic (A53Ttg/tg) mice, caspase-1 knockout mice, and A53Ttg/tg;caspase-1−/− double transgenic mice were used to prepare adult neural stem cells (ANSCs) and to investigate inflammasome-related mechanism for α-syn-impaired neurogenesis in mouse subventricular zone (SVZ). We showed that α-syn inhibited neurogenesis in the SVZ of A53Ttg/tg mice and impaired proliferation and differentiation in ANSCs cultured in vitro, accompanied by reduced microRNA-7 (miR-7) expression levels. We further found that ANSC expressed NLRP3-containing inflammasome and α-syn activated both TLR4/NF-κB and NLRP3/caspase-1 signals in ANSCs. Either Nlrp3 knockdown or Caspase-1 knockout could attenuate the inhibition of proliferation in ANSCs induced by α-syn. Furthermore, we demonstrated that miR-7 post-transcriptionally controlled Nlrp3 expression besides targeting α-syn. Most notably, stereotactic injection of miR-7 mimics into lateral ventricles significantly inhibited NLRP3 inflammasome activation and improved adult neurogenesis in mouse SVZ. Our study provides a direct link between NLRP3 inflammasome activation and α-syn-impaired neurogenesis in the pathogenesis of α-synucleinopathies.





Similar content being viewed by others
References
Giasson BI, Duda JE, Quinn SM, Zhang B, Trojanowski JQ, Lee VM (2002) Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein. Neuron 34(4):521–533
Gasser T (2009) Molecular pathogenesis of Parkinson disease: insights from genetic studies. Expert reviews in molecular medicine 11, e22
Schapira AH, Bezard E, Brotchie J, Calon F, Collingridge GL, Ferger B, Hengerer B, Hirsch E et al (2006) Novel pharmacological targets for the treatment of Parkinson’s disease. Nat Rev Drug Discov 5(10):845–854
Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, Hulihan M, Peuralinna T et al (2003) alpha-Synuclein locus triplication causes Parkinson’s disease. Science 302(5646):841
Nishioka K, Hayashi S, Farrer MJ, Singleton AB, Yoshino H, Imai H, Kitami T, Sato K et al (2006) Clinical heterogeneity of alpha-synuclein gene duplication in Parkinson’s disease. Ann Neurol 59(2):298–309
Alvarez-Buylla A, Lim DA (2004) For the long run: maintaining germinal niches in the adult brain. Neuron 41(5):683–686
Gould E (2007) How widespread is adult neurogenesis in mammals? Nat Rev Neurosci 8(6):481–488
O’Sullivan SS, Johnson M, Williams DR, Revesz T, Holton JL, Lees AJ, Perry EK (2011) The effect of drug treatment on neurogenesis in Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society 26(1):45–50
Hirsch EC, Vyas S, Hunot S (2012) Neuroinflammation in Parkinson’s disease. Parkinsonism Relat Disord 18(Suppl 1):S210–S212
Koprich JB, Reske-Nielsen C, Mithal P, Isacson O (2008) Neuroinflammation mediated by IL-1beta increases susceptibility of dopamine neurons to degeneration in an animal model of Parkinson’s disease. J Neuroinflammation 5:8
Walsh JG, Muruve DA, Power C (2014) Inflammasomes in the CNS. Nat Rev Neurosci 15(2):84–97
Dinarello CA (2007) A signal for the caspase-1 inflammasome free of TLR. Immunity 26(4):383–385
Schroder K, Zhou R, Tschopp J (2010) The NLRP3 inflammasome: a sensor for metabolic danger? Science 327(5963):296–300
Franchi L, Munoz-Planillo R, Nunez G (2012) Sensing and reacting to microbes through the inflammasomes. Nat Immunol 13(4):325–332
Bryant C, Fitzgerald KA (2009) Molecular mechanisms involved in inflammasome activation. Trends Cell Biol 19(9):455–464
Jin C, Flavell RA (2010) Molecular mechanism of NLRP3 inflammasome activation. J Clin Immunol 30(5):628–631
Sayed D, Abdellatif M (2011) MicroRNAs in development and disease. Physiol Rev 91(3):827–887
Gangaraju VK, Lin H (2009) MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol 10(2):116–125
Kawahara H, Imai T, Okano H (2012) MicroRNAs in Neural Stem Cells and Neurogenesis. Front Neurosci 6:30
Junn E, Lee KW, Jeong BS, Chan TW, Im JY, Mouradian MM (2009) Repression of alpha-synuclein expression and toxicity by microRNA-7. Proc Natl Acad Sci U S A 106(31):13052–13057
Kong H, Sha LL, Fan Y, Xiao M, Ding JH, Wu J, Hu G (2009) Requirement of AQP4 for antidepressive efficiency of fluoxetine: implication in adult hippocampal neurogenesis. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology 34(5):1263–1276
Kong H, Fan Y, Xie J, Ding J, Sha L, Shi X, Sun X, Hu G (2008) AQP4 knockout impairs proliferation, migration and neuronal differentiation of adult neural stem cells. J Cell Sci 121(Pt 24):4029–4036
Baudry A, Mouillet-Richard S, Schneider B, Launay JM, Kellermann O (2010) miR-16 targets the serotonin transporter: a new facet for adaptive responses to antidepressants. Science 329(5998):1537–1541
Shen X, Fang J, Lv X, Pei Z, Wang Y, Jiang S, Ding K (2011) Heparin impairs angiogenesis through inhibition of microRNA-10b. J Biol Chem 286(30):26616–26627
John B, Enright AJ, Aravin A, Tuschl T, Sander C, Marks DS (2004) Human MicroRNA targets. PLoS Biol 2(11), e363
Westerlund U, Svensson M, Moe MC, Varghese M, Gustavsson B, Wallstedt L, Berg-Johnsen J, Langmoen IA (2005) Endoscopically harvested stem cells: a putative method in future autotransplantation. Neurosurgery 57(4):779–784, discussion 779–784
Cave JW, Baker H (2009) Dopamine systems in the forebrain. Adv Exp Med Biol 651:15–35
Crews L, Mizuno H, Desplats P, Rockenstein E, Adame A, Patrick C, Winner B, Winkler J et al (2008) Alpha-synuclein alters Notch-1 expression and neurogenesis in mouse embryonic stem cells and in the hippocampus of transgenic mice. J Neurosci 28(16):4250–4260
Tani M, Hayakawa H, Yasuda T, Nihira T, Hattori N, Mizuno Y, Mochizuki H (2010) Ectopic expression of alpha-synuclein affects the migration of neural stem cells in mouse subventricular zone. J Neurochem 115(4):854–863
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140(6):918–934
Yin Y, Yan Y, Jiang X, Mai J, Chen NC, Wang H, Yang XF (2009) Inflammasomes are differentially expressed in cardiovascular and other tissues. Int J Immunopathol Pharmacol 22(2):311–322
Lee EJ, Woo MS, Moon PG, Baek MC, Choi IY, Kim WK, Junn E, Kim HS (2010) Alpha-synuclein activates microglia by inducing the expressions of matrix metalloproteinases and the subsequent activation of protease-activated receptor-1. J Immunol 185(1):615–623
Rolls A, Shechter R, London A, Ziv Y, Ronen A, Levy R, Schwartz M (2007) Toll-like receptors modulate adult hippocampal neurogenesis. Nat Cell Biol 9(9):1081–1088
Kalinowski FC, Brown RA, Ganda C, Giles KM, Epis MR, Horsham J, Leedman PJ (2014) microRNA-7: a tumor suppressor miRNA with therapeutic potential. Int J Biochem Cell Biol 54:312–317
de Chevigny A, Core N, Follert P, Gaudin M, Barbry P, Beclin C, Cremer H (2012) miR-7a regulation of Pax6 controls spatial origin of forebrain dopaminergic neurons. Nat Neurosci 15(8):1120–1126
Acknowledgments
The work reported herein was supported by the grants from the National Natural Science Foundation of China (No. 81473196, No. 81573403, No. 81202514, and No. 81202512), the National Science & Technology Major Project (No. 2012ZX09304-001), the Natural Science Foundation of Jiangsu Province (BK20130039), and the key project of Natural Science Foundation of the Higher Education Institutions of Jiangsu Province (No. 15KJA310002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Zheng Fan and Ming Lu contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
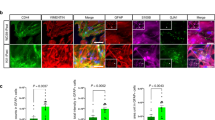
Phenotypic identification of cultured adult neural stem cells (ANSCs). (A) α-syn immunostaining in ANSCs. (B) Nestin immunostaining in ANSCs. Scale bar: 100 μm. (PDF 110 kb)
ESM 2
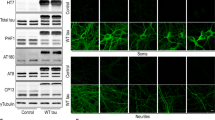
Phenotypic identification of ANSCs isolated from caspase-1+/+ and caspase-1−/− mice. (A) The morphological photo of neurospheres isolated from SVZ of caspase-1+/+ and caspase-1−/− mice. Scale bar: 100 μm. (B & C) Statistical analysis the number and diameter of neurospheres in both genotypic ANSCs. Data are expressed as mean ± S.E.M. from four independent experiments. (PDF 109 kb)
ESM 3
Luciferase report data show that miR-7 failed to inhibit Tlr4 gene expression. Data are expressed as mean ± S.E.M. from four independent experiments. (PDF 97 kb)
ESM 4
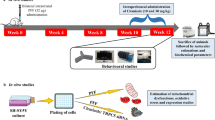
The protocol illustration of stereotactic injection and representative photo of injected miR-7 mimics in SVZ and striatum. Scale bar: 200 μm. (PDF 100 kb)
Rights and permissions
About this article
Cite this article
Fan, Z., Lu, M., Qiao, C. et al. MicroRNA-7 Enhances Subventricular Zone Neurogenesis by Inhibiting NLRP3/Caspase-1 Axis in Adult Neural Stem Cells. Mol Neurobiol 53, 7057–7069 (2016). https://doi.org/10.1007/s12035-015-9620-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9620-5