Abstract
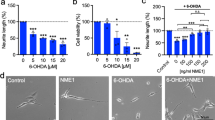
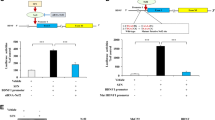
The transduction of dopaminergic (DA) neurons with human ras homolog enriched in brain, which has a S16H mutation [hRheb(S16H)] protects the nigrostriatal DA projection in the 6-hydroxydopamine (6-OHDA)-treated animal model of Parkinson’s disease (PD). However, it is still unclear whether the expression of active hRheb induces the production of neurotrophic factors such as glial cell line-derived neurotrophic factor (GDNF) and brain-derived neurotrophic factor (BDNF), which are involved in neuroprotection, in mature neurons. Here, we show that transduction of nigral DA neurons with hRheb(S16H) significantly increases the levels of phospho-cyclic adenosine monophosphate (cAMP) response element-binding protein (p-CREB), GDNF, and BDNF in neurons, which are attenuated by rapamycin, a specific inhibitor of mammalian target of rapamycin complex 1 (mTORC1). Moreover, treatment with specific neutralizing antibodies for GDNF and BDNF reduced the protective effects of hRheb(S16H) against 1-methyl-4-phenylpyridinium (MPP+)-induced neurotoxicity. These results show that activation of hRheb/mTORC1 signaling pathway could impart to DA neurons the important ability to continuously produce GDNF and BDNF as therapeutic agents against PD.







Similar content being viewed by others
Abbreviations
- 6-OHDA:
-
6-Hydroxydopamine
- AAV1:
-
Adeno-associated virus 1
- ANOVA:
-
Analysis of variance
- AP:
-
Anterior-posterior
- BDNF:
-
Brain-derived neurotrophic factor
- DA:
-
Dopaminergic
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DV:
-
Dorsal-ventral
- FLAG:
-
FLAG-tag
- GDNF:
-
Glial cell line-derived neurotrophic factor
- GFAP:
-
Glial fibrillary acidic protein
- GFP:
-
Green fluorescent protein
- hRheb(S16H):
-
Human Ras homolog enriched in brain with a S16H mutation
- ip:
-
Immunopositive
- MFB:
-
Medial forebrain bundle
- ML:
-
Medial-lateral
- MPP+ :
-
1-Methyl-4-phenylpyridinium
- mTORC1:
-
Mammalian target of rapamycin complex 1
- p-CREB:
-
Phospho-cAMP response element binding protein
- PBS:
-
Phosphate-buffered saline
- PD:
-
Parkinson’s disease
- SD:
-
Sprague-Dawley
- SN:
-
Substantia nigra
- SNpc:
-
Substantia nigra pars compacta
- STR:
-
Striatum
- TH:
-
Tyrosine hydroxylase
References
Burke RE, O'Malley K (2013) Axon degeneration in Parkinson’s disease. Exp Neurol 246:72–83
Savitt JM, Dawson VL, Dawson TM (2006) Diagnosis and treatment of Parkinson disease: molecules to medicine. J Clin Invest 116(7):1744–1754
Allen SJ, Watson JJ, Shoemark DK, Barua NU, Patel NK (2013) GDNF, NGF and BDNF as therapeutic options for neurodegeneration. Pharmacol Ther 138(2):155–175
Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F (1993) GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science 260(5111):1130–1132
Manfredsson FP, Okun MS, Mandel RJ (2009) Gene therapy for neurological disorders: challenges and future prospects for the use of growth factors for the treatment of Parkinson’s disease. Curr Gene Ther 9(5):375–388
Siegel GJ, Chauhan NB (2000) Neurotrophic factors in Alzheimer’s and Parkinson’s disease brain. Brain Res Brain Res Rev 33(2–3):199–227
Howells DW, Porritt MJ, Wong JY, Batchelor PE, Kalnins R, Hughes AJ, Donnan GA (2000) Reduced BDNF mRNA expression in the Parkinson’s disease substantia nigra. Exp Neurol 166(1):127–135
Studer L, Spenger C, Seiler RW, Othberg A, Lindvall O, Odin P (1996) Effects of brain-derived neurotrophic factor on neuronal structure of dopaminergic neurons in dissociated cultures of human fetal mesencephalon. Exp Brain Res 108(2):328–336
Chauhan NB, Siegel GJ, Lee JM (2001) Depletion of glial cell line-derived neurotrophic factor in substantia nigra neurons of Parkinson’s disease brain. J Chem Neuroanat 21(4):277–288
Hyman C, Hofer M, Barde YA, Juhasz M, Yancopoulos GD, Squinto SP, Lindsay RM (1991) BDNF is a neurotrophic factor for dopaminergic neurons of the substantia nigra. Nature 350(6315):230–232
Kholodilov N, Kim SR, Yarygina O, Kareva T, Cho JW, Baohan A, Burke RE (2011) Glial cell line-derived neurotrophic factor receptor-alpha1 expressed in striatum in trans regulates development and injury response of dopamine neurons of the substantia nigra. J Neurochem 116(4):486–498
Creedon DJ, Tansey MG, Baloh RH, Osborne PA, Lampe PA, Fahrner TJ, Heuckeroth RO, Milbrandt J, Johnson EM Jr (1997) Neurturin shares receptors and signal transduction pathways with glial cell line-derived neurotrophic factor in sympathetic neurons. Proc Natl Acad Sci U S A 94(13):7018–7023
Bhave SV, Ghoda L, Hoffman PL (1999) Brain-derived neurotrophic factor mediates the anti-apoptotic effect of NMDA in cerebellar granule neurons: signal transduction cascades and site of ethanol action. J Neurosci 19(9):3277–3286
Selvaraj S, Sun Y, Watt JA, Wang S, Lei S, Birnbaumer L, Singh BB (2012) Neurotoxin-induced ER stress in mouse dopaminergic neurons involves downregulation of TRPC1 and inhibition of AKT/mTOR signaling. J Clin Invest 122(4):1354–1367
Kim SR, Chen X, Oo TF, Kareva T, Yarygina O, Wang C, During M, Kholodilov N, Burke RE (2011) Dopaminergic pathway reconstruction by Akt/Rheb-induced axon regeneration. Ann Neurol 70(1):110–120
Kim SR, Kareva T, Yarygina O, Kholodilov N, Burke RE (2012) AAV transduction of dopamine neurons with constitutively active Rheb protects from neurodegeneration and mediates axon regrowth. Mol Ther 20(2):275–286
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates, 4th edn. Academic Press, San Diego
Park ES, Kim SR, Jin BK (2012) Transient receptor potential vanilloid subtype 1 contributes to mesencephalic dopaminergic neuronal survival by inhibiting microglia-originated oxidative stress. Brain Res Bull 89(3–4):92–96
Kurauchi Y, Hisatsune A, Isohama Y, Mishima S, Katsuki H (2012) Caffeic acid phenethyl ester protects nigral dopaminergic neurons via dual mechanisms involving haem oxygenase-1 and brain-derived neurotrophic factor. Br J Pharmacol 166(3):1151–1168
Zhou J, Blundell J, Ogawa S, Kwon CH, Zhang W, Sinton C, Powell CM, Parada LF (2009) Pharmacological inhibition of mTORC1 suppresses anatomical, cellular, and behavioral abnormalities in neural-specific Pten knock-out mice. J Neurosci 29(6):1773–1783
Chung YC, Kim SR, Jin BK (2010) Paroxetine prevents loss of nigrostriatal dopaminergic neurons by inhibiting brain inflammation and oxidative stress in an experimental model of Parkinson’s disease. J Immunol 185(2):1230–1237
Kim SR, Lee DY, Chung ES, Oh UT, Kim SU, Jin BK (2005) Transient receptor potential vanilloid subtype 1 mediates cell death of mesencephalic dopaminergic neurons in vivo and in vitro. J Neurosci 25(3):662–671
Pascual A, Hidalgo-Figueroa M, Piruat JI, Pintado CO, Gomez-Diaz R, Lopez-Barneo J (2008) Absolute requirement of GDNF for adult catecholaminergic neuron survival. Nat Neurosci 11(7):755–761
Cen X, Nitta A, Ohya S, Zhao Y, Ozawa N, Mouri A, Ibi D, Wang L, Suzuki M, Saito K, Ito Y, Kawagoe T, Noda Y, Furukawa S, Nabeshima T (2006) An analog of a dipeptide-like structure of FK506 increases glial cell line-derived neurotrophic factor expression through cAMP response element-binding protein activated by heat shock protein 90/Akt signaling pathway. J Neurosci 26(12):3335–3344
Jeon SJ, Rhee SY, Seo JE, Bak HR, Lee SH, Ryu JH, Cheong JH, Shin CY, Kim GH, Lee YS, Ko KH (2011) Oroxylin A increases BDNF production by activation of MAPK-CREB pathway in rat primary cortical neuronal culture. Neurosci Res 69(3):214–222
Altar CA, Heikkila RE, Manzino L, Marien MR (1986) 1-Methyl-4-phenylpyridine (MPP+): regional dopamine neuron uptake, toxicity, and novel rotational behavior following dopamine receptor proliferation. Eur J Pharmacol 131(2–3):199–209
Yan L, Findlay GM, Jones R, Procter J, Cao Y, Lamb RF (2006) Hyperactivation of mammalian target of rapamycin (mTOR) signaling by a gain-of-function mutant of the Rheb GTPase. J Biol Chem 281(29):19793–19797
Pochon NA, Menoud A, Tseng JL, Zurn AD, Aebischer P (1997) Neuronal GDNF expression in the adult rat nervous system identified by in situ hybridization. Eur J Neurosci 9(3):463–471
Gash DM, Zhang Z, Gerhardt G (1998) Neuroprotective and neurorestorative properties of GDNF. Ann Neurol 44(3Suppl1):S121–S125
Peterson AL, Nutt JG (2008) Treatment of Parkinson’s disease with trophic factors. Neurotherapeutics 5(2):270–280
Seroogy KB, Lundgren KH, Tran TM, Guthrie KM, Isackson PJ, Gall CM (1994) Dopaminergic neurons in rat ventral midbrain express brain-derived neurotrophic factor and neurotrophin-3 mRNAs. J Comp Neurol 342(3):321–334
Mogi M, Togari A, Kondo T, Mizuno Y, Komure O, Kuno S, Ichinose H, Nagatsu T (1999) Brain-derived growth factor and nerve growth factor concentrations are decreased in the substantia nigra in Parkinson’s disease. Neurosci Lett 270(1):45–48
Porritt MJ, Batchelor PE, Howells DW (2005) Inhibiting BDNF expression by antisense oligonucleotide infusion causes loss of nigral dopaminergic neurons. Exp Neurol 192(1):226–234
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (Nos. 2008-0061888 and 2012R1A1A1039140).
Conflict of Interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jin Han Nam Eunju Leem and Min-Tae Jeon contributed equally to this work.
Rights and permissions
About this article
Cite this article
Nam, J.H., Leem, E., Jeon, MT. et al. Induction of GDNF and BDNF by hRheb(S16H) Transduction of SNpc Neurons: Neuroprotective Mechanisms of hRheb(S16H) in a Model of Parkinson’s Disease. Mol Neurobiol 51, 487–499 (2015). https://doi.org/10.1007/s12035-014-8729-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8729-2