Abstract
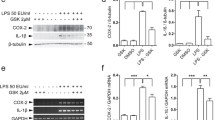
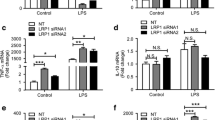
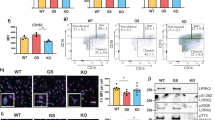
The leucine-rich repeat kinase 2 (LRRK2) gene was discovered to encode a member of an evolutionarily conserved family of proteins marked by GTPase domains usually in combination with kinase domains. Missense mutations in both the kinase and GTPase domains in LRRK2 have been found to cause late-onset Parkinson’s disease (PD). In this study, we investigated the effects of the LRRK2 on endothelial inflammation. We first demonstrated that the LRRK2 is expressed in endothelial cells. We also report here that IL-1β can possibly increase LRRK2 expression in human umbilical vein endothelial cells (HUVECs). Wild-type LRRK2 (LRRK2wt) expression induces expression of vascular cell adhesion molecule 1 (VCAM-1) which is further exacerbated in cells expressing PD-associated LRRK2 G2019S mutants (LRRK2G2019S). Importantly, induction of VCAM-1 is almost completely blocked in cells expressing the GTP-binding-deficient mutant K1347A of LRRK2 (LRRK2K1347A). In addition, overexpression of LRRK2wt and LRRK2G2019S were found to cause an increase in monocyte attachment to endothelial cells. Mechanistically, we found that LRRK2 increases the transcriptional activity of nuclear factor κB (NF-κB) by increasing phosphorylation levels of IκBα. These findings suggest that inhibition of LRRK2 kinase activity may be a potential target for treatment of endothelial dysfunction.






Similar content being viewed by others
References
Bosgraaf L, Van Haastert PJ (2003) Roc, a Ras/GTPase domain in complex proteins. Biochim Biophys Acta 1643:5–10
Dächsel JC, Farrer MJ (2010) LRRK2 and Parkinson disease. Arch Neurol 67:542–547
Farkas E, De Jong GI, de Vos RA, Jansen Steur EN, Luiten PG (2000) Pathological features of cerebral cortical capillaries are doubled in Alzheimer’s disease and Parkinson’s disease. Acta Neuropathol 100:395–402
Gardet A, Benita Y, Li C, Sands BE, Ballester I, Stevens C, Korzenik JR, Rioux JD, Daly MJ, Xavier RJ, Podolsky DK (2010) LRRK2 is involved in the IFN-gamma response and host response to pathogens. J Immunol 185(9):5577–5585
Gimbrone MA Jr, Topper JN, Nagel T, Anderson KR, Garcia-Cardena G (2000) Endothelial dysfunction, hemodynamic forces, and atherogenesis. Ann N Y Acad Sci 902:230–239, discussion 239–240
Greggio E, Cookson MR (2009) Leucine-rich repeat kinase 2 mutations and Parkinson’s disease: three questions. ASN Neurol 1:e00002
Guo L, Gandhi PN, Wang W, Petersen RB, Wilson-Delfosse AL, Chen SG (2007) The Parkinson’s disease associated protein, leucine-rich repeat kinase 2 (LRRK2), is an authentic GTPase that stimulates kinase activity. Exp Cell Res 313:3658–3670
Hakimi M, Selvanantham T, Swinton E, Padmore RF, Tong Y, Kabbach G, Venderova K, Girardin SE, Bulman DE, Scherzer CR, LaVoie MJ, Gris D, Park DS, Angel JB, Shen J, Philpott DJ, Schlossmacher MG (2011) Parkinson’s disease-linked LRRK2 is expressed in circulating and tissue immune cells and upregulated following recognition of microbial structures. J Neural Transm 118:795–808
Healy DG et al (2008) Phenotype, genotype, and worldwide genetic penetrance of LRRK2-associated Parkinson’s disease: a case–control study. Lancet Neurol 7:583–590
Kortekaas R, Lenders KL, van Oostrom JCH et al (2005) Blood-brain barrier dysfunction in parkinsonian midbrain in vivo. Ann Neurol 57:176–179
Martin I et al (2011) Recent advances in the genetics of Parkinson’s disease. Annu Rev Genomics Hum Genet 12:301–325
Moehle MS, Webber PJ, Tse T, Sukar N, Standaert DG, DeSilva TM, Cowell RM, West AB (2012) LRRK2 inhibition attenuates microglial inflammatory responses. J Neurosci 32(5):1602–1611
Mogi M, Harada M, Narabayashi H, Inagaki H, Minami M, Nagatsu T (1996) Interleukin (IL)-1 beta, IL-2, IL-4, IL-6 and transforming growth factor-alpha levels are elevated in ventricular cerebrospinal fluid in juvenile Parkinsonism and Parkinson’s disease. Neuroscience 211:13–16
Mogi M, Kondo T, Mizuno Y, Nagatsu T (2007) p53 protein, interferon-c, and NF-kB levels are elevated in the parkinsonian brain. Neurosci Lett 414:94–97
Nagatsu T, Sawada M (2005) Inflammatory process in Parkinson’s disease: role for cytokines. Curr Pharm Des 11:999–1016
Sheng B, Wang X, Su B, Lee HG, Casadesus G et al (2012) Impaired mitochondrial biogenesis contributes to mitochondrial dysfunction in Alzheimer’s disease. J Neurochem 120(3):419–429
Shi H, Sheng B, Zhang F (2013) Kruppel-like factor 2 protects against ischemic stroke by regulating endothelial blood brain barrier function. Am J Physiol Heart Circ Physiol 304(6):H796–H805
Smith WW, Pei Z, Jiang H, Dawson VL, Dawson TM, Ross CA (2006) Kinase activity of mutant LRRK2 mediates neuronal toxicity. Nat Neurosci 9:1231–1233
Springer TA (1994) Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 76:301–314
Thevenet J, Pescini Gobert R, Hooft van Huijsduijnen R, Wiessner C, Sagot YJ (2011) Regulation of LRRK2 expression points to a functional role in human monocyte maturation. PLoS One 6:e21519
West AB, Moore DJ, Biskup S, Bugayenko A, Smith WW, Ross CA, Dawson VL, Dawson TM (2005) Parkinson’s disease-associated mutations in leucine-rich repeat kinase 2 augment kinase activity. Proc Natl Acad Sci U S A 102(46):16842–16847
Zhou G, Hamik A, Nayak L et al (2012) Endothelial Kruppel-like factor 4 protects against atherothrombosis in mice. J Clin Invest 122(12):4727–4731
Zimprich A, Biskup S, Leitner P, Lichtner P, Farrer M, Lincoln S, Kachergus J, Hulihan M, Uitti RJ, Calne DB, Stoessl AJ, Pfeiffer RF, Patenge N, Carbajal IC, Vieregge P, Asmus F, Müller-Myhsok B, Dickson DW, Meitinger T, Strom TM, Wszolek ZK, Gasser T (2004a) Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron 44(4):601–607
Zimprich A et al (2004b) Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron 44:601–607
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hongge, L., Kexin, G., Xiaojie, M. et al. The Role of LRRK2 in the Regulation of Monocyte Adhesion to Endothelial Cells. J Mol Neurosci 55, 233–239 (2015). https://doi.org/10.1007/s12031-014-0312-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-014-0312-9