Abstract
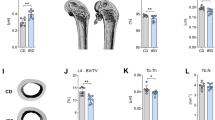
This study reports the effects of dietary iron (Fe) deficiency and recovery on bone mineral composition and strength in anemic rats submitted to a hemoglobin (Hb) repletion assay. Weanling male Wistar rats were fed a low-Fe diet (12 mg/kg) for 15 days followed by 2 weeks of Fe repletion with diets providing 35 mg Fe/kg as either ferrous sulfate (n = 8) or ferric pyrophosphate (FP; n = 12). At final day of each period (depletion and repletion), Fe-adequate animals were also euthanized. Iron status (blood Hb, Hb Fe pool, Hb regeneration efficiency), tibia mineral concentrations (Ca, Mg, Fe, Cu, and Zn) and biomechanical properties were evaluated. Iron-deficient rats had lower tibia Fe and Mg levels and bone strength when compared to controls. Yield load and resilience were positively related to tibia Mg levels (r = 0.47, P = 0.02 and r = 0.56, P = 0.004, respectively). Iron repletion did not recover tibia Mg concentrations impaired by Fe deficiency. Moreover, bone elastic properties were negatively affected by FP consumption. In conclusion, bone mineral composition and strength were affected by Fe deficiency, whereas dietary Fe source influenced tibia Mg and resistance in the period during which rats were recovering from anemia.


Similar content being viewed by others
Abbreviations
- FS:
-
Ferrous sulfate
- FP:
-
Ferric pyrophosphate
- HRE:
-
Hemoglobin regeneration efficiency
References
Nieves JW (2005) Osteoporosis: the role of micronutrients. Am J Clin Nutr 81:1232S–1239S
Medeiros DM, Plattner A, Jennings D et al (2002) Bone morphology, strength and density are compromised in iron-deficient rats and exacerbated by calcium restriction. J Nutr 132:3135–3141
Medeiros DM, Stoecker B, Plattner A et al (2004) Iron deficiency negatively affects vertebrae and femurs of rats independently of energy intake and body weight. J Nutr 134:3061–3067
Katsumata S-I, Katsumata-Tsuboi R, Uehara M, Suzuki K (2009) Severe iron deficiency decreases both bone formation and bone resorption in rats. J Nutr 139:1–6
Tuderman L, Myllyla R, Kivirikko KI (1977) Mechanism of the prolyl hydroxylase reaction. 1. Role of co-substrates. Eur J Biochem 80:341–348
DeLuca HF (1976) Metabolism of vitamin D: current status. Am J Clin Nutr 29:1258–1270
Hoenderop JGJ, Nilius B, Bindels RJM (2005) Calcium absorption across epithelia. Physiol Rev 85:373–422
Vaz RTC, Lobo AR, Cocato ML et al (2010) Effects of inulin-type fructans consumption on mineral intestinal absorption and balance in rats fed control and iron-deficient diets. Braz J Food Nutr 21:5–11
Lobo AR, Cocato ML, Jorgetti V et al (2009) Changes in bone mass, biomechanical properties, and microarchitecture of calcium- and iron-deficient rats fed diets supplemented with inulin-type fructans. Nutr Res 29:873–881
Association of Official Analytical Chemists (2006) Official methods of analysis of Association of Official Analytical Chemists, 18th edn. AOAC International, Maryland, pp 80–81, Current through Revision 1, chapter 45
Reeves PG, Nielsen FH, Fahey GC Jr (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on reformulation of the AIN-76A rodent diet. J Nutr 123:1939–1951
Cocato ML, Ré MI, Trindade Neto MA et al (2007) Avaliação por métodos in vitro e in vivo da biodisponibilidade de sulfato ferroso microencapsulado. Braz J Nutr 20:239–247
Sedlin ED, Hirsch C (1966) Factors affecting the determination of the physical properties of femoral cortical bone. Acta Orthop Scand 37:29–48
Drabkin DL, Austin JH (1935) Spectrophotometric studies II. Preparations from washed blood cells: nitric oxide hemoglobin and sulphemoglobin. J Biol Chem 112:51–65
Mahoney AW, Van Orden CC, Hendricks DG (1974) Efficiency of converting food iron into haemoglobin by the anemic rat. Nutr Metab 17:223–230
Fritz JC, Pla GW, Harrison BN et al (1974) Collaborative study of the rat hemoglobin repletion test for biovailability of iron. J AOAC Int 57:513–517
Forbes AL, Adams CE, Arnaud MJ et al (1989) Comparison of in vitro, animal, and clinical determinations of iron bioavailability: International Nutritional Anemia Consultative Group Task Force report on iron bioavailability. Am J Clin Nutr 49:225–238
Wienk KJH, Marx JJM, Beynen AC (1999) The concept of iron bioavailabilty and its assessment. Eur J Nutr 38:51–75
Fritz JC, Pla GW, Harrison BN et al (1978) Measurement of the bioavailability of iron, using the rat hemoglobin repletion test. J AOAC Int 61:709–714
Hurrel RF (2002) How to ensure adequate iron absorption from iron-fortified food. Nutr Rev 60:S7–S15
Heaton FW, Tongyai S, Motta C et al (1987) Changes in the erythrocyte membrane during magnesium deficiency. Nutr Res 7:655–663
De Franceschi L, Brugnara C, Beuzard Y (1997) Dietary magnesium supplementation ameliorates anemia in a mouse model of β-thalassemia. Blood 90:1283–1290
Shimano MM, Shimano AC, Volpon JB (2002) Histerese de fêmures de ratas submetidos a ensaio de flexão, em diferentes faixas etárias. Rev Bras Eng Biomed 18:88–97
Knott L, Bailey AJ (1998) Collagen cross-links in mineralizing tissues: a review of their chemistry, function, and clinical relevance. Bone 22:181–187
Burr DB (2002) The contribution of the organic matrix to bone’s material properties. Bone 31:8–11
Kobayashi M, Hara K, Akyiama Y (2004) Effects of vitamin K2 (menatetrenone) and alendronate on bone mineral density and bone strength in rats fed a low-magnesium diet. Bone 35:1136–1143
Amizuka N, Li M, Kobayashi M et al (2008) Vitamin k2, a γ-carboxylating factor of gla-proteins, normalizes the bone crystal nucleation impaired by Mg-insufficiency. Histol Histopathol 23:1353–1366
Acknowledgments
The authors thank Dr. José Alfredo Gomes Arêas and Dr. Luiz Antonio Gioielli for their collaboration in the bone biomechanical assays; Ms. Tatiana Garofalo Quintal and Maura Sayuri de Andrade for technical assistance; Fundação de Amparo à Pesquisa do Estado de São Paulo (research project 2006/01735-0) for supporting the research; and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the fellowships awarded to Alexandre R. Lobo. This study was also supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). We also thank Álvaro Augusto Feitosa Pereira for reviewing the manuscript.
Conflicts of Interest
There are no financial, professional, or personal conflicts of interests for any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lobo, A.R., Gaievski, E.H.S. & Colli, C. Hemoglobin Regeneration Efficiency in Anemic Rats: Effects on Bone Mineral Composition and Biomechanical Properties. Biol Trace Elem Res 143, 403–411 (2011). https://doi.org/10.1007/s12011-010-8871-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8871-2