Abstract
Summary
We performed comorbidity-network analysis to obtain global view of comorbidity related with osteoporosis. We selected 10000-patients with osteoporosis registered in the National-Health-Insurance Service cohort-database. We found 45-significant disease-clusters. Of these, 14-disease-clusters were related to fra, while 10 were related to musculoskeletal diseases. Our findings will serve as basic data for further studies.
Purpose
Osteoporosis causes devastating fractures; however, its exact etiology remains unknown. Elucidating associated comorbidities and their temporal relationships could provide better insights into its pathogenesis. Comorbidity-network analysis was performed to obtain global view of these associations.
Methods
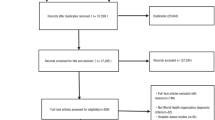
We randomly selected 10000-patients with osteoporosis registered in the National-Health-Insurance Service cohort-database. These patients were identified using ICD-10 codes M81-M82, which represent osteoporosis without pathological fractures. Control group was created through propensity score matching. The comorbidities in each group were grouped into similar classifications to form “disease cluster”; 126 such clusters were identified. To create a comorbidity network, we selected disease clusters with high associations (i.e., odds ratios and relative risks ranked in the upper 50th percentile). To identify the temporal relationships between these clusters and osteoporosis, trajectories of directions were identified.
Results
Finally, we found 45 significant disease clusters. Of these, 14 disease clusters were related to fractures or injuries, while 10 were related to musculoskeletal diseases. Temporal analysis revealed that 15 disease clusters preceded osteoporosis; these included the following three with the strongest associations: “other fracture”, “disorders of bone density and structure (M83–M85)”, and “sequelae of injuries of neck and trunk (T91)”. Thirty disease clusters followed osteoporosis; these included the following three with the strongest associations: “spine fracture,” “spondylopathies (M45–M49)”, and “pelvic region and thigh fracture,”.
Conclusion
We obtained a global view of the osteoporosis comorbidity network, which is otherwise difficult to achieve through study of individual diseases. Our findings will serve as the basic data for further studies.





Similar content being viewed by others
Data Availability
This is not permitted by authority who has the right for data.
References
Divo MJ, Martinez CH, Mannino DM (2014) Ageing and the epidemiology of multimorbidity. Eur Respir J 44:1055–1068
Ben-Shlomo Y, Kuh D (2002) A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol 31:285–293
Aguado A, Moratalla-Navarro F, Lopez-Simarro F, Moreno V (2020) MorbiNet: multimorbidity networks in adult general population. Analysis of type 2 diabetes mellitus comorbidity. Sci Rep 10:2416
Khan A, Uddin S, Srinivasan U (2018) Comorbidity network for chronic disease: A novel approach to understand type 2 diabetes progression. Int J Med Inform 115:1–9
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res 22:465–475
Kim KH, Lee K, Ko YJ, Kim SJ, Oh SI, Durrance DY, Yoo D, Park SM (2012) Prevalence, awareness, and treatment of osteoporosis among Korean women: The Fourth Korea National Health and Nutrition Examination Survey. Bone 50:1039–1047
Wang P, Abdin E, Shafie S, Chong SA, Vaingankar JA, Subramaniam M (2019) Estimation of Prevalence of Osteoporosis Using OSTA and Its Correlation with Sociodemographic Factors, Disability and Comorbidities. Int J Environ Res Public Health 16
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA (1999) Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 353:878–882
Adami G, Saag KG (2019) Osteoporosis Pathophysiology, Epidemiology, and Screening in Rheumatoid Arthritis. Curr Rheumatol Rep 21:34
Crepaldi G, Maggi S (2009) Epidemiologic link between osteoporosis and cardiovascular disease. J Endocrinol Invest 32:2–5
Lopez-Medina C, Molto A (2020) Comorbidity management in spondyloarthritis. RMD Open 6
Austin PC (2011) An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivar Behav Res 46:399–424
Divo MJ, Casanova C, Marin JM et al (2015) COPD comorbidities network. Eur Respir J 46:640–650
Zhu X, Gerstein M, Snyder M (2007) Getting connected: analysis and principles of biological networks. Genes Dev 21:1010–1024
Hanauer DA, Ramakrishnan N (2013) Modeling temporal relationships in large scale clinical associations. J Am Med Inform Assoc 20:332–341
Kim JH, Son KY, Shin DW, Kim SH, Yun JW, Shin JH, Kang MS, Chung EH, Yoo KH, Yun JM (2016) Network analysis of human diseases using Korean nationwide claims data. J Biomed Inform 61:276–282
Sinigaglia L, Nervetti A, Mela Q et al (2000) A multicenter cross sectional study on bone mineral density in rheumatoid arthritis. Italian Study Group on Bone Mass in Rheumatoid Arthritis J Rheumatol 27:2582–2589
Hauser B, Riches PL, Wilson JF, Horne AE, Ralston SH (2014) Prevalence and clinical prediction of osteoporosis in a contemporary cohort of patients with rheumatoid arthritis. Rheumatology (Oxford) 53:1759–1766
Ziegelasch M, Forslind K, Skogh T, Riklund K, Kastbom A, Berglin E (2017) Decrease in bone mineral density during three months after diagnosis of early rheumatoid arthritis measured by digital X-ray radiogrammetry predicts radiographic joint damage after one year. Arthritis Res Ther 19:195
Mori Y, Kuwahara Y, Chiba S, Kogre A, Baba K, Kamimura M, Itoi E (2017) Bone mineral density of postmenopausal women with rheumatoid arthritis depends on disease duration regardless of treatment. J Bone Miner Metab 35:52–57
Vattikuti R, Towler DA (2004) Osteogenic regulation of vascular calcification: an early perspective. Am J Physiol Endocrinol Metab 286:E686–E696
Dhore CR, Cleutjens JP, Lutgens E, Cleutjens KB, Geusens PP, Kitslaar PJ, Tordoir JH, Spronk HM, Vermeer C, Daemen MJ (2001) Differential expression of bone matrix regulatory proteins in human atherosclerotic plaques. Arterioscler Thromb Vasc Biol 21:1998–2003
Schneider DL, Barrett-Connor EL, Morton DJ (1994) Thyroid hormone use and bone mineral density in elderly women. Effects of estrogen. JAMA 271:1245–1249
La Vignera S, Vicari E, Tumino S, Ciotta L, Condorelli R, Vicari LO, Calogero AE (2008) L-thyroxin treatment and post-menopausal osteoporosis: relevance of the risk profile present in clinical history. Minerva Ginecol 60:475–484
Lin SY, Lin CL, Chen HT, Kao CH (2018) Risk of osteoporosis in thyroid cancer patients using levothyroxine: a population-based study. Curr Med Res Opin 34:805–812
Appetecchia M (2005) Effects on bone mineral density by treatment of benign nodular goiter with mildly suppressive doses of L-thyroxine in a cohort women study. Horm Res 64:293–298
Baldini M, Gallazzi M, Orsatti A, Fossati S, Leonardi P, Cantalamessa L (2002) Treatment of benign nodular goitre with mildly suppressive doses of L-thyroxine: effects on bone mineral density and on nodule size. J Intern Med 251:407–414
De Rosa G, Testa A, Maussier ML, Calla C, Astazi P, Albanese C (1995) A slightly suppressive dose of L-thyroxine does not affect bone turnover and bone mineral density in pre- and postmenopausal women with nontoxic goitre. Horm Metab Res 27:503–507
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Puth MT, Klaschik M, Schmid M, Weckbecker K, Munster E (2018) Prevalence and comorbidity of osteoporosis- a cross-sectional analysis on 10,660 adults aged 50 years and older in Germany. BMC Musculoskelet Disord 19:144
Liao CY, Chung CH, Chu P, Wei KY, Feng TM, Lin FH, Tsao CH, Wu CC, Chien WC (2018) Increased risk of osteoporosis in patients with primary biliary cirrhosis. PloS One 13:e0194418
Lygrisse KA, Roof MA, Keitel LN, Callaghan JJ, Schwarzkopf R, Bedard NA (2020) The Inaccuracy of ICD-10 Coding in Revision Total Hip Arthroplasty and Its Implication on Revision Data. J Arthroplasty 35:2960–2965.e3
Acknowledgement
This work was supported by the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (No. NRF-2021R1A4A3023587, 2022R1A2C2005916, 2022R1F1A1074057).
Author information
Authors and Affiliations
Contributions
S.Y.: Data curation; formal analysis; writing original draft
H.L.: Project administration; data curation; formal analysis; writing original draft
J.K.: Project administration; writing original draft
W.A.: Manuscript review and editing
S.L.: Conceptualization; funding acquisition; investigation; methodology; manuscript review and editing
Corresponding author
Ethics declarations
Ethical approval
The study had been approved by the appropriate institutional research ethics committee and has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Conflicts of interest
Competing interest: Hyun Il Lee, Siyeong Yoon, Jin Hwan Kim, Wooyeol Ahn, and Soonchul Lee declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information

ESM 1
Supplementary Fig. 1 Undirected network map of all comorbidities (126 disease cluster). This figure represents the global view of association among total 126 disease-clusters. All links whose p-value is less than 0.05 after Fisher’s exact test, were included. The size of nodes is proportional to the degree (number of link to other disease-clusters). The color of nodes represents the type of ICD-10 (major classification according to initial alphabet character). The thickness of edges indicates the odds ratio between disease clusters. (TIF 1600 kb) (PNG 1118 kb)

ESM 2
Supplementary Fig. 2 The network of disease clusters with high-interconnectivity. The most highly connected 11 disease-clusters were selected to make another network graph. Surprisingly only 3 disease-clusters were omitted in this network graph compared to original graph made by 45 disease-clusters (C73 Malignant neoplasm of thyroid gland; D10-D36 Benign neoplasms; E00-E07 Disorders of thyroid gland) showing the vital role of highly interconnected disease cluster as hub. (PNG 1535 kb)
ESM 3
(DOCX 31.5 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, H.I., Yoon, S., Kim, J.H. et al. Network analysis of osteoporosis provides a global view of associated comorbidities and their temporal relationships. Arch Osteoporos 18, 79 (2023). https://doi.org/10.1007/s11657-023-01290-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-023-01290-2