Abstract
The microbiome is a term that usually refers to the community of various microorganisms that inhabit/live inside human/animal bodies or on their skin. It forms a complex ecosystem that includes trillions of commensals, symbiotics, and even pathogenic microorganisms. The external environment, diet, and lifestyle are the major determinants influencing the microbiome’s composition and vitality. Recent studies have indicated the tremendous influence of the microbiome on health and disease. Their number, constitution, variation, and viability are dynamic. All these elements are responsible for the induction, development, and treatment of many health disorders. Serious diseases such as cancer, metabolic disorders, cardiovascular diseases, and even psychological disorders such as schizophrenia are influenced directly or indirectly by microbiota. In addition, in the last few weeks, accumulating data about the link between COVID-19 and the microbiota were published. In the present work, the role of the microbiome in health and disease is discussed. A deep understanding of the exact role of microbiota in disease induction enables the prevention of diseases and the development of new therapeutic concepts for most diseases through the correction of diet and lifestyle. The present review brings together evidence from the most recent works and discusses suggested nutraceutical approaches for the management of COVID-19 pandemic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The microbiome consists of various bacteria, archaea, fungi, protozoa, and even viruses. At least 800 different bacterial species could be isolated from healthy humans’ gut (muniesa and jofre 2014; el-sayed et al. 2021). In addition, members of the viral pool inhabitant in the gut (virome) have been recently identified in the gut. The virome consists mainly of bacteriophages, plant-derived viruses, and various giant viruses. In contrast to the gut’s bacterial inhabitants, most of the virome members have not been previously recognized due to technical difficulties in the detection, identification, and isolation of the saprophytic gut viruses. On average, one gram of stool is estimated to contain at least 109 virus-like particles (VLPs) and 109 bacterial cells (Kim et al. 2011). However, the microbiota composition is dynamic and undergoes continuous changes in response to external factors, including nutritional status, environmental factors, lifestyle, health status, and diseases, medications such as antibiotics (Table 2) (Scarpellini et al. 2015; El-Sayed et al. 2021). For example, the administration of high doses of antibiotics for 3 weeks leads to a dramatic reduction in the amounts of gut microbiome and fecal DNA content (Pak et al. 2019). The impact of infectious diseases and different strategies for their treatment and control on microbial dysbiosis are illustrated in Figure 1.
The influence of microbiota on our health and disease status is exerted in many directions. They can induce/modify metabolic-, auto-immunogenic, and neurodegenerative diseases. In addition, they regulate the host immune response and modulate drug interactions (Grice and Segre 2012). Their effect on our health can only be understood if we know that human origin cells constitute only 10% of the total number of living cells in human bodies. In comparison, the microbiota represents the remaining 90% of the cells. Similarly, only 1% of the total unique genes in our bodies have a human origin, and the remaining 99% belong to the gut and the oral cavity’s microbiota. Therefore, the human microbiome is considered our second genome (Tremlett et al. 2017; Scarpellini et al. 2015; Grice and Segre 2012).
Beside the gut, the microbiota can also colonize the oral cavity, nares, vagina, breast, and the skin (Table 1). The oral microbiota consists mainly of streptococci and to a lesser extent other species as Veillonella, Gamella, Rothia, Fusobacterium, and Neisseria. Although the oral microbiota is essential for the maintenance of oral health, they may induce various diseases such as caries (by Streptococcus mutans, Veillonella, Lactobacillus, Bifidobacterium, and Propionibacterium) and periodontitis (by Porphyromonas gingivalis, Actinobacillus actinomycetemcomitans, Treponema denticola, and Tannerella forsythia) (Bik et al. 2010; Aas et al. 2008; Belda-Ferre et al. 2011).
Beside the previously listed bacterial species, viruses (particularly bacteriophages) are also represented in the community of oral microbiota (known as salivary viromes) and may also play a significant role in the induction of oral diseases (Pride et al. 2012). It is worth mentioning that the oral microbiota’s harmful effect extends beyond the induction of dental diseases to induce systemic diseases. One of the best examples is the connection between periodontitis and the induction of diabetes and cardiovascular diseases (e.g., atherosclerosis and stroke) where the cholesterol portion narrowing the arteriosclerotic artery walls is synthesized by oral microbiota and does not have a dietary origin as believed (Koren et al. 2011; Leishman et al. 2010; Patrakka et al. 2019).
Competition among different bacterial species has been traditionally used to counteract the harmful effects of invading pathogens. This therapeutic approach is mostly applied for the treatment of gastrointestinal diseases, such as the use of Saccharomyces boulardii for the treatment of post-antibiotic diarrhoea (Dinleyici et al. 2012), Lactobacillus GG for the treatment of diarrhoea caused by salmonella (Aggarwal et al. 2014), Weissella cibaria JW15 (JW15) to inhibit pathogens like Listeria monocytogenes, Salmonella Typhimurium, S. enteritidis, and Escherichia coli (Yu et al. 2019). The E. coli strain A0 34/86 can also be used to prevent the colonization of pathogenic E. coli in the gut of infants (Micenková et al. 2019), while Escherichia coli Nissle 1917 is administrated for the treatment of leaky gut (Ukena et al. 2007). Similarly, bacteriophages have been also used to treat antibiotic-resistant MRSA strains (Botka et al. 2019) and infections caused by Mycobacterium abscessus (Dedrick et al. 2019). Recently, genetically modified bacteriophages were used for the treatment of pathogens in the gut, even intracellular pathogens (Møller-Olsen et al. 2018).
Alteration in gut microbiota with aging
The microbial content of the GIT tract of infants has a simple structured composition dominated by bifidobacteria. The composition and the prevalence of various bacterial species are influenced by many factors such as the maternal feed during pregnancy, the mode of delivery (normal birth vs. caesarean sections), and the type of feeding (breastfeeding vs. milk replacers). However, the composition of the microbiome changes continuously with age. Comparison of the microbiome composition in infants and in 4-year-old children revealed the reduction in the population of phyla Proteobacteria and Actinobacteria accompanied by the increase in phyla Firmicutes and Bacteroidetes in the gut of the older children (Guo et al. 2020).
In general, as the microbiome functionality differs between infants and adults, therefore the microbial profiles are adapted in response, e.g., while the microbiota involved in amino acid and vitamin metabolism dominates in infants gut, these microbes will be slowly replaced by microbiota participated in fat metabolism with age (Guo et al. 2020).
The microbial structure varies even among elderly populations from different countries in response to age, diet, lifestyle, metabolic diseases like obesity, and environmental influencing factors (Claesson et al. 2011). Therefore, with age, the population of disease-associated pathobionts and facultative anaerobes (Clostridia and Bacteroides, Betaproteobacteria, and Deltaproteobacteria) generally increase in conjunction with the reduction of the number of beneficial commensal bacteria (as Actinobacteria, bifidobacterial, Firmicutes, lactobacilli, and bifidobacterial), leading to the disturbance of the Bacteroidetes:Firmicutes ratio (Woodmansey 2007; Pellanda et al. 2021; Odamaki et al. 2016). Although the core microbial profile of elderly persons is clearly distinct from that for younger adults, there is no fixed universal composition of microbiota in the elderly population (Mueller et al. 2006). These changes share to a great extent in age-related physio-pathological changes leading to inflammaging (gut inflammatory status, usually mediated by NF-ĸB transcription factor which induces a state of persistent activation of gut innate immunity) in addition to the reduction of both gut immune response and gut epithelial barrier functionality (Walton et al. 2020; Claesson et al. 2011). The alteration in gut microbiome composition and the resulting physio-pathological changes with age were proposed as possible explanations for the difference in COVID-19 susceptibility between young and elderly populations (Zimmermann and Curtis 2020).
Differences in existing microbiota at the geographical level
The composition of microbiota was also found to be influenced by the geographic location due to the differences in environmental (e.g., environmental temperature and water resources) and cultural factors (e.g., type and availability of diet, sport, lifestyle, and stress) (Redondo-Useros et al. 2020; Mohr et al. 2020).
The effect of the culture is clearly seen when comparing the Mediterranean versus Western diet. Evidence linking consumption of the Mediterranean diet and the modulation of the gut microbiome are accumulating. This modulation includes the increase in the population of Lachnospira and Prevotella and the reduction of the population of Ruminococcus, which reflects positively on the health status. The protective role of the Mediterranean dietary pattern against diabetes, cardiovascular disease, Crohn’s disease, and cancer was reported (Shively et al. 2018).
The shift to Mediterranean diet plan reduces insulin resistance and protects against diabetes by restoring the normal population of P. distasonis, B. thetaiotaomicron, F. prausnitzii, B. adolescentis, Butyricimonas, Desulfovibrio, Oscillospira, and B. longum (Jin et al. 2019). Similarly, the profiles of gut microbiome varied clearly between African and European children. In African kids, the intestinal flora is more variable, had more Prevotella than Bacteroides, and produced more butyrate. This is mainly attributed to the high-fiber low-protein diet consumption in African countries (de Filippo et al. 2010; Yatsunenko et al. 2012; Lin et al. 2013). A direct effect of climatic temperature on microbiota was documented. In high/cold geographical locations, the participation of two phyla changes in response to external temperature. While the prevalence of Firmicutes increases with latitude, the Bacteroidetes population sinks with the latitude. Both phyla are essential for increasing the body mass index and body fats (Suzuki and Worobey 2014).
Other environmental factors like floods and drinking water sources influence the gut microbiome, especially in warm countries where water-borne pathogens (as salmonella and E. coli infections) are common (Yusof et al. 2017).
Distribution of microbiota
The microbiota is present wherever contact with the external non-sterile environment exists. Therefore, they harbor the gut, oral cavity, nares, vagina, and the outer skin layer. Each location has its characteristic inhabitants. Naturally born babies acquire additional bacterial species during their passage via the vagina, mainly Lactobacillus and Prevotella. In opposite, babies born via caesarean section gain bacterial species similar to those living on the skin, such as Propionibacterium, Staphylococcus, and Corynebacterium (Dominguez-Bello et al. 2010). Similarly, differences were also found in microbiota’s composition the gut of naturally breastfed babies vs. formula-fed (Penders et al. 2006). In addition to the vagina, many studies have been carried out to investigate the presence/absence of bacteria in the gravid uterus. Most of the investigated samples harbored bacterial DNA in the amniotic fluid samples. All mothers and feti involved in these studies were healthy and did not show any signs of infection. The studies referred to the important role of the fatal microbiome for intrauterine development in the immune system (Stinson et al. 2019) (Table 2).
The presence of gut virome was always overseen in human fluid samples as the samples are mostly cultured on bacteriological culture media or tested with PCR using specific bacterial 16S RNA gene probes (Garrido-Cardenas and Manzano-Agugliaro 2017). However, in opposite to the bacterial members of the microbiota, viruses present in the virome (especially bacteriophages) can normally pass through the intestinal wall, circulate in the blood at high concentrations, and cross the physiological barriers as the placenta and the blood-brain barrier (BBB). They, therefore, could be isolated from the cerebrospinal fluid (CSF), which is always thought to be sterile (Tetz and Tetz 2018; Srivastava et al. 2019; Srivastava et al. 2004). It was estimated that daily, on average, about 31 billion bacteriophages can be transcytosed across the gut epithelium in the human body and spread via blood and lymph to all body organs, including the brain (Nguyen et al. 2017).
Clinical potential of understanding the role of microbiota in disease induction
The changes in the structure and composition of the microbiome have a great influence on the individuum’s health and disease status. Understanding the microbiota-gut-liver-brain axis provides a better understanding of microbiota’s role in the induction of serious infectious (e.g., COVID-19 and neoplasm) and non-infectious diseases (e.g., diabetes and CNS disorders). Such data are important for (1) the prevention and control of the diseases, (2) for the development of non-pharmaceutical therapeutic approaches (e.g., via probiotics and nutraceuticals), and (3) as disease markers for the early prediction of diseases via monitoring the changes in microbial composition in the gut (e.g., prediction of metabolic diseases) (Ding et al. 2020).
Role of gut microbiota in disease induction
The intestinal mucosal barrier is one of the major body defense barriers, consisting of physical, chemical, microbial, and immunological components. It protects the body against bacterial invasion, the passage of foreign antigens and toxins to the circulation, and against the loss of water and nutrients. It regulates molecular exchange and the coexistence/colonization of gut microbiota. Therefore, the disruption of this balanced equilibrium or the occurrence of leaky gut has serious consequences (Bischoff et al. 2014). The intestinal barrier functionality is supported by the administration of probiotics such as Streptococcus thermophilus, Lactobacillus acidophilus, and Escherichia coli Nissle 1917, which not only improves the efficiency of the intestinal barrier but also prevents invasion of gut pathogens (Ukena et al. 2007). Even minimal changes in gut microbiota composition have great consequences and influence the future development of serious diseases such as neoplasms (Bongers et al. 2014) as shown in Fig. 2.
Coronavirus infection (COVID-19)
Recently, the presence of a strong gut- lung axis was suggested by many authors. This was confirmed during the present COVID-19 pandemic where a clear correlation between infection with COVID-19 and the alteration in the composition of fecal microbiome could be reported. Based on that, modulation of the gut microbiome showed significant alleviation of the symptoms. According to Tursi and his team (2020), the administration of oral probiotic formulation of Streptococcus thermophilus, L. acidophilus, L. helveticus, L. paracasei, L. plantarum, L. brevis, and B. lactis could relieve the COVID-19-induced diarrhoea and the risk of respiratory failure and decreases the mortality rates in COVID-19-hospitalized patients (Tursi and Papa 2020; Janda et al. 2020). These effects are attributed mainly to the direct modulatory effect of diet on gut microbiota or through the alleviation of chronic gut inflammation which result from prolonged consumption of red meat and alcohol (Rishi et al. 2020; Ferreira et al. 2020).
Moreover, the gut microbiota was observed to upregulate the production of ACE2 in the gut of laboratory animals. By so doing, the microbiota can influence the ability of the virus to invade the gut epithelium (Walton et al. 2020). The effect of diet on gut microbiota and the prevalence of COVID-19 were clearly noticed during the lockdown, where healthy home-cooked food partially replaced fast food. The accumulating evidence surrounding the interaction between gut microbiota and COVID-19 drew the attention towards the benefits of (nutraceutical approaches for COVID-19 therapy). The recommended protocol involves administering probiotics, vitamins (mainly vitamins C, D, and E), and trace elements (mainly selenium and zinc). It is also recommended the reduction of caloric intake (carbohydrates and trans-fat or saturated fats) and to replace red meat with fish (omega-3 polyunsaturated fatty acids) and high-fiber plant-based food (Ferreira et al. 2020; Belančić 2020; Tursi and Papa 2020). Moreover, the involvement of nutraceuticals such as polyphenols (flavonoids, phenolic acids, stilbenes, lignans) and spices like curcumin in the diet plan plays a potential role in countering COVID-19 pathways (Infusino et al. 2020).
Cancer
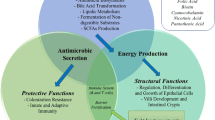
The composition and source of diet are major factors that influence the vitality and composition of microbiota. The worldwide increase in the consumption of sweets, soft drinks, and junk foods leads to changes in the composition of gut microbiota. The consumption of high-carbohydrate low amino acid diet leads to the disturbance of Firmicutes-to-Bacteroidetes ratio. Restricted protein intake was also shown to change the gene expression pattern in the liver and reduce hepatic FXR-FGF15 signaling (Pak et al. 2019; Holmes et al. 2017; El-Sayed et al. 2021). On the other hand, the consumption of low-fiber/high red meat diet alters the intestinal microbiota to favor tumor formation; diet rich in NSP enhances bacterial fermentation/utilization of ingested fibers and the production of short-chain fatty acids (SCFA) such as butyrate, acetate, and propionate. Butyrate and SCFA (in general) are used for energy production by colon epithelium; they exhibit powerful anti-inflammatory effects, required for cell regeneration and the maintenance of a healthy gut barrier (Mutlu et al. 2012; David et al. 2014; Kim et al. 2013; Suzuki et al. 2008). Daily consumption of red meat at high amounts may induce colonic cancer. The microbial lipoperoxidation of heme iron plays a role in carcinogenesis (Martin et al. 2018).
Red meat consumption increases the population of bacterial species involved in the metabolism of amino acids such as Fusobacteria and Alistipes, accompanied by a decrease in the prevalence of those that limit neoplasm induction (SCFA producing species). Supplying the patients with Lactobacillus and Bifidobacterium species improves the anti-inflammatory anti-carcinogenicity properties (Borges-Canha et al. 2015; Damman et al. 2012; Sokol et al. 2008; Tirandaz and Mohammadi 2013; Veerappan et al. 2012). The role of every species in the enhancement/prevention of cancer development has been studied by many researchers (Moore and Moore 1995; Scanlan et al. 2009).
In addition to red meat, the consumption of processed meat, particularly that fried at very high temperatures, enhances cancer formation due to the production of various mutagenic carcinogenic heterocyclic amines, N-nitroso compounds, heterocyclic amines, and polycyclic aromatic hydrocarbons (Wu et al. 2006). Meanwhile, the consumption of white meat was seen to be less harmful to red meat. This finding is attributed to the maintenance of microbiota balance in white meat consumers. The beneficial genus Lactobacillus was higher in the white meat than in the red meat consumers or even non-meat protein feeders (Zhu et al. 2015).
In addition to red meat, the consumption of fats, especially essential unsaturated fatty acids (n-6 polyunsaturated fatty acids (PUFAs)), plays a critical role in the induction of gastric and colon cancer. The ingested fat enhances cancer by influencing gut flora to metabolize fats to carcinogenic by-products (Lee et al. 2018). It is also believed that a fat-rich diet can stimulate the secretion of high amounts of bile. The bile is then dehydrogenated by anaerobic gut bacteria to produce carcinogenic by-products (Harris 2016).
However, the effect of fat consumption on health status depends on the type of fat consumed as the consumption of good fats like fish oils and omega-3 polyunsaturated fatty acids can protect against colorectal cancer (CRC) (Volpato and Hull 2018). Beside fish oil, other nutritional elements have been found to provide a protective effect against CRC, such as fiber-rich food and high doses of vitamin D or calcium (Thun et al. 2018; Morales-Oyarvide et al. 2016; Harris 2016).
The above-described lifestyle, known as westernized lifestyle (consumption of high meat, high energy, high-fat, and poor-fiber diet) besides smoking and consuming alcohol together, are responsible for the high prevalence of cancer in the Western world (Pelucchi et al. 2011; Marley and Nan 2016; Harris 2016; Bishehsari et al. 2014).
Alcohol consumption is also considered to be a predisposing factor for colon cancer. Alcohol is metabolized in the gut by microflora to produce carcinogenic by-products such as acetaldehyde. Produced acetaldehyde in the gut is responsible for the degradation of folate needed for DNA repair. The permanent deficiency of folate may lead to severe chromosomal damage and neoplasms development (Homann et al. 2000; Duthie 1999).
Besides the role of diet in cancer development, many cancer types may arise following infections or as a result of direct disturbance in the microbiota (e.g., following infection with Fusobacterium H. pylori, Leptotrichia, and Campylobacter species) (Warren et al. 2013) or viral infection (e.g., human papillomavirus (HPV), hepatitis B virus (HBV) and hepatitis C virus (HCV), Kaposi’s sarcoma-associated herpesvirus (KSHV), Merkel cell polyomavirus (MCV)) (Mui et al. 2017; Lunn et al. 2017).
The induction of cancer may also be induced by disturbances in the prevalence/constitution of gut flora. Therefore, the prevalence of CRC is much higher in the large intestine than in the small intestine. This is attributed to the higher microbial population in the large intestine, especially the gut virome and bacteriophages (Dahiya and Renuka 2017; Sun and Kato 2016). An example of the relationship between present microflora and the type of resulting cancer is infections with Streptococcus bovis (SB), which includes Strep. gallolyticus (SB type I and II/2) and Strep. infantarius (SB type II/1). While the Strep. gallolyticus is linked to CRC, Strep. infantarius was found to be related to other types of cancer, such as pancreatic and biliary tract tumors (Corredoira et al. 2008a; Corredoira et al. 2008b). SB is a normal inhabitant in the digestive tract of ruminants and can tolerate up to 40% bile concentration. It can also be detected in human fecal samples. Its presence in humans is usually related to serious diseases such as colon carcinoma/ adenoma, inflammatory bowel disease (IBD), endocarditis, or bacteremia (Potter et al. 1998; Dubrow et al. 1991; Nielsen et al. 2007; Sharara et al. 2013; Teitelbaum and Triantafyllopoulou 2006).
Examples of infection-induced neoplasms
Fusobacterium (F) nucleatum is one of the oral microbiota that can be isolated from stool. Its presence in fecal samples is usually bound to colorectal carcinoma (Tahara et al. 2014; Castellarin et al. 2012).
Although E. coli is a normal commensal inhabitant in the human intestinal tract, some isolates acquired mobile genetic elements that harbor various virulence genes and become human pathogens (El Sayed 2000). One of these mobile genetic elements is the pathogenicity island polyketide synthases (pks), which encode colibactin, which is linked to the induction of IBD, Crohn’s disease, and even CRC (Buc et al. 2013; Bonnet et al. 2014; Martin et al. 2004).
Even some colonic symbiotes (e.g., Bacteroides fragilis), which represent one of the GIT microbiota, produces a zinc-dependent metalloprotease toxin that induces epithelial inflammation, activates cell proliferation, and probably damages the host cell DNA (Goodwin et al. 2011; Wu et al. 2009; Boleij et al. 2015).
Helicobacter pylori (H. pylori) is listed as a group 1 carcinogen in the list according to the International Agency for Research on Cancer (IARC) as it induces chronic gastritis, which may result in gastric adenocarcinoma or lymphoid tissue lymphoma. Furthermore, the pathogen’s extension to the intestine may also result in CRC (Parkin 2006; Wu et al. 2013; Konturek et al. 2009).
Salmonella infections usually result from consumption of contaminated foodstuffs of animal sources, especially eggs. The infection may turn chronic, and the carrier sheds the bacteria in the feces (Nagaraja and Eslick 2014; Kato et al. 2013). Chronic infection with Salmonella results in persistent inflammation in the GIT, which may develop inflammatory bowel syndrome (IBS), arthritis, or even colorectal and gall bladder cancer. In parallel, as many members of the clostridial family are incriminated in tumor induction, antibiotic therapy and transplantation of microbiota from healthy individuals have been shown to prevent the development of cancer in tumor- susceptible mice as an animal model (Kumar et al. 2006; Lu et al. 2014; Bongers et al. 2014).
Metabolic diseases
Gut microbiota was found to influence the prevalence of metabolic disorders and general health conditions by mediating the effects of drugs and diet. A low-protein diet improves glycemic control, and metabolic health, increases energy expenditure, and decreases the prevalence of obesity, insulin resistance, and type 2 diabetes. This was attributed to the alteration in gut microbiota at the phylum level and therefore affects liver metabolism (Pak et al. 2019). Gut microbiota may also influence metabolic disorders in a second pathway. Various species of microbiota catalyze the bile acids differently. Bile acids are now known to play a role in fat digestion and as activators of the nuclear hormone receptors. Therefore, the variation in the composition of microbiota consequently leads to variation in the processing of bile acids in the gut, reflecting diverse body activities (Kuipers et al. 2014; Sayin et al. 2013).
An apparent variation in the composition of the microbiota and phenotype of diabetes has been reported in animal models. The alteration in microbiota composition in some instances can enhance insulin resistance and thereby induce type II diabetes. Therefore, recent studies have shown that microbiota transplantation results in a change in susceptibility to metabolic disorders. The tendency for metabolic disorders increases in host harboring microbiota, which can better process dietary sugar and generate hydrophobic bile acids (Kreznar et al. 2017). The main players that determine the occurrence of metabolic disorders among various microbial metabolites in the gut are the SCFA. Insulin sensitivity was positively related to concentration of butyrate.
Similarly, acetate stimulates insulin secretion from the pancreas. However, when SCFA increases, the host will suffer from obesity (Hartstra et al. 2015; Priyadarshini et al. 2015). Examination of microbiota present in the gut of patients suffering from insulin resistance/type 2 diabetes showed a decrease in butyrate-producing bacteria and an increase in bacteria inducing sulfate reduction and oxidative stress resistance (Qin et al. 2012). Although metformin has been used for about 60 years to treat type 2 diabetes, the mechanism of action of metformin could only be understood recently. Metformin was found to alter the gut microbiota in a way that improves glucose tolerance in patients. These findings were confirmed by studying the level of glucose tolerance in germ-free mice that received microbiota obtained from metformin-treated patients (Wu et al. 2017). In addition to their role in the induction of type 2 diabetes, a possible role of microbiota in the induction of type 1 diabetes has also been reported (Greiner et al. 2014). Based on this, new approaches were developed to treat obesity and diabetes by transplanting microbiota from the gut of healthy individuals into the gut of diabetic people, which leads to the potential sinking of blood glucose levels (Greiner et al. 2014; Zhang et al. 2013).
Obesity
Obesity is defined as excessive fat accumulation to the degree that impairs health. It is primarily, but not exclusively, caused by an energy imbalance between calorie intake and calories expended in the presence of predisposing genetic factors. It may also result from other factors, such as hormonal imbalance (Hill et al. 2012) or disturbance in microbiome composition (mainly the increase in the proportion of Firmicutes accompanied by the decrease in the proportion of Bacteroidetes) (Suzuki and Worobey 2014). Therefore, certain individuals can keep their weight constant despite their high-calorie feed. Present obesity control programs depend mainly on the reduction of energy intake and/or increasing energy expended. This concept ignores the role of other factors, such as insulin resistance and hormonal disturbances (Camacho and Ruppel 2017), and ignores the source of energy in the diet. Weight control programs that do not consider the source of calories usually fail to maintain weight loss. This is attributed to the high demand for energy (175 kCal/day) for the digestion of macronutrients (catabolism), which will not be spent when eating low-fiber high-calorie industrial products (Heymsfield et al. 2012; Feinman and Fine 2004; Jéquier 2002).
According to the WHO, a global obesity epidemic occurred due to the explosive consumption of soft drinks and fast foods among young people, especially between 70th and the 90th (Jaacks et al. 2019). Sugar consumption similarly leads to addiction to drugs do as it leads to the release of dopamine and delays the release of acetylcholine. Repeated and excessive consumption of sugar-rich diets as soft drinks induces behavioral and mental changes similar to those generated in junkies suffering from drug abuse (Hoebel et al. 2009; Avena et al. 2008). Meanwhile, high-calorie diet consumption such as sugar-/fat-rich foodstuffs (ideally sugar-fat combinations such as cookies) has behavioral/neurological and physiological influences such as bulimia and anorexia. This is due to the sharing of behavioral impacts on neural pathways such as the mesolimbic dopamine (DA) system. The release of dopamine represents a rewarding effect similar to addictive behavior (Avena et al. 2009; Davis and Claridge 1998; Volkow et al. 2008; Marrazzi and Luby 1986). Symptoms indicating sugar-addictive potential in animal models are common to drug dependency. They included “Bingeing,” “withdrawal signs,” “craving,” and cross-sensitization, which are attributed to neurochemical changes in the CNS (Avena et al. 2008; Rada et al. 2005). These findings are supported by brain tomography scans, which revealed similar images in both sugar and drug-addicted persons (Wang et al. 2004).
Due to sugar addiction, the global consumption of sugar has increased rapidly in connection with the increase in overweight problems worldwide. Overweight is not only a predisposing factor for heart diseases, metabolic diseases, and joint and backbone diseases, but it has also been found to be linked to at least 13 various types of cancer, including endometrial cancer (Dougan et al. 2015), esophageal adenocarcinoma (Hoyo et al. 2012), gastric cardia cancer, (Chen et al. 2013), liver cancer (Chen et al. 2012), kidney cancer (Sanfilippo et al. 2014; Wang and Xu 2014), multiple myeloma (Wallin and Larsson 2011), meningioma (Niedermaier et al. 2015), pancreatic cancer (Genkinger et al. 2011), CRC, (Ma et al. 2013), gall bladder cancer (Li et al. 2016), breast cancer (Munsell et al. 2014), ovarian cancer (127), and thyroid cancer (Kitahara et al. 2016).
Long-term consumption of a high-protein diet is linked to insulin resistance, diabetes, and cardiovascular disease and enhances the occurrence of certain types of cancer such as colonic cancer. In patients suffering from obesity, shifting their diet to a low-protein diet leads to weight loss, improved insulin sensitivity, and restoration of metabolic health (Pak et al. 2019). Alternatively, the transplantation of microbiota from the gut of healthy people may help solve the problem of obesity (Jéquier 2002).
Liver diseases
Recent studies allied various liver diseases, including non-alcoholic steatohepatitis, liver fibrosis/cirrhosis, and hepatic cancer (hepatic cell carcinoma) with the composition of gut microbiota (Friedman 2013; Guarner and Soriano 2005).
Neural diseases
The interaction between gut microbiota and the development of the nervous system has been investigated by many researchers. It is now clear that the microbiota exerts a long-term influence on the nervous system, starting from birth until aging.
Recent publications have hypothesized that neurodegenerative diseases as multiple sclerosis, amyotrophic lateral sclerosis, Parkinson’s disease, Alzheimer’s disease, and even certain psychiatric and neurological diseases as autism and depression could be attributed to functional disorders of the gastrointestinal tract due to alterations and reduction in the diversity of gut microbiota with age (Pellegrini et al. 2018; Hsiao et al. 2013). Additionally, people with limited microbial diversity are found to suffer from poor memory/cognition functions (Verdi et al. 2018) and other neuroinflammatory/neurodegenerative diseases such as traumatic brain injury and acute ischemia. This is attributed to the breakdown of the gut barrier, allowing pro-inflammatory cytokines and mediators produced by the bacteria to cross the BBB accompanied by immune cells infiltration from the circulation into the CNS, which induces neuronal inflammation, demyelination, and finally damage (Janakiraman and Krishnamoorthy 2018; Kowalski and Mulak 2019; Calvani et al. 2018).
Other diseases
The commensal oral anaerobic bacteria Fusobacterium nucleatum has invasive pro-inflammatory properties, which enable them to induce acute and chronic infections all over the digestive tract (Signat et al. 2011). Although these bacteria are normally commensal and have no pathogenic life pattern in the oral cavity, they are associated with cases of Crohn’s disease, IBD, and appendicitis (Swidsinski et al. 2011; Strauss et al. 2011).
Role of virome in health and diseases
More than 1200 viral genotypes can be detected in fecal samples of healthy individuals (about 108–109 VLPs/g feces)(Breitbart et al. 2003; Kim et al. 2011). Virome present in the human gut contains primarily non-pathogenic viruses (Breitbart et al. 2003). This includes plant-derived viruses such as Pepper mild mottle v., Oat blue dwarf v. and Oat chlorotic stunt v., Tobacco mosaic v., Grapevine asteroid mosaic-associated v., Panicum mosaic v., and the Maize chlorotic mottle v. Virome also involves many DNA giant viruses (> 300 kb) such as members of Poxviridae, Mimiviridae, Asfarviridae, Mamaviridae, Marseilleviridae, Phycodnaviridae, Iridoviridae, and finally the Ascoviridae. Additional groups present in the virome include the Microviridae family represented by the members: Microvirus, Gokushovirinae, Alpavirinae, and Pichovirinae (Scarpellini et al. 2015). In addition to the previously mentioned families, the gut contains another category of viruses that play an essential role in bacterial/pathogen evolution, namely, the bacteriophages (the phagobiota). The phagobiota represents the most abundant members of the human gut microbiome (Duerkop 2018; Dalmasso et al. 2014). Phages are mobile genetic elements that play a major role in transferring virulence genes among the bacterial population. The large genetic diversity and heterogeneity of microbiota and phages enable the phages’ continuous adaptation to new usually non-susceptible bacterial hosts. This, in turn, promotes diversification and leads to the continuous evolution of new genotypes and even new pathogens. This phage/bacterial interaction maintains the bacteriophage and microbial diversity in the GIT and may even play a vital role in the induction of dysbiosis following treatment with antibiotics or certain gut inflammatory diseases (Sordi et al. 2017).
They play a significant role in regulating the bacterial community in the gut and can translocate via the gut mucosa to lymph/internal organs. Therefore, they can play an additional immunoregulatory role. Some can share in disease induction; others possibly have a protective role in the gut (Łusiak-Szelachowska et al. 2017).
In the very early life of infants, the variety of gut bacteriophages is usually great with a clear dominance of the DNA phages from the order Caudovirales. Later on, the composition shifts and the variety decreases where the members of the family Microviridae begin to dominate. The alteration in virobiota composition and the speed of these shifts depends mainly on the feed and will have great consequences on the microbiota and, consequently, on human health later in life (Lim et al. 2016).
Generally, bacteriophages can be grouped into two major groups according to their lifestyle, lytic phages that kill the bacteria after infection to produce virions. The second type is known as lysogenic phages that integrate their DNA in the bacterial genome and replicate passively with the bacteria. As bacteriophages are host-specific, they selectively infect and kill only susceptible bacterial species (El Sayed 2000). This may lead to a decrease in bacterial diversity and, therefore, indirectly induce certain human diseases such as IBD and even certain oral diseases (Norman et al. 2015; Ly et al. 2014).
Phage induction can be induced in many ways, which subject the bacteria to stress conditions such as administering certain antibiotics that stimulate the SOS signals in lysogenic bacteriophages. As a result, the transcription of late genes starts and results in increased phage integrase DNA within the gut phage community. Phage induction leads to the death of hosting bacteria and the release of billions of bacteriophages simultaneously. This, in turn, has consequences like disturbances in the bacterial equilibrium among symbionts to pathobionts in the gut, transmission of mobile genetic elements among the related bacterial species, evolution of new pathogens, and possibly intestinal dysbiosis (Johnson et al. 2017; Ghaisas et al. 2016; Allen et al. 2011).
In general, phage induction can be influenced by nutrient availability. The effect of diet on bacteriophage populations can be clearly seen when shifting from a low-fat diet to a high-fat diet (Howe et al. 2016). Bacterial lysis also leads to local inflammation in the intestinal epithelium due to the release of bacterial proteins, lipids, and DNA (Manrique et al. 2016). The released bacteriophages may also penetrate the gut mucosa and migrate via blood/lymphatics towards the immune tissues (Górski et al. 2006). Under certain circumstances, the mucin covering the mucous membranes of the GIT may trap bacteriophages through a mechanism known as bacteriophage adherence to mucin (BAM), where capsids of certain phages contain immunoglobulin (Ig)-like domains, which can adhere to the host mucin. Their intestinal mucosa position allows them to act as a guard offering protection against invading bacteria (Barr et al. 2013; Minot et al. 2012). The interaction between phages and mammalian cells is not limited to their adhesion to mucus membranes. The phages may be endocytosed by the epithelial lining and again exit from them. The necked phage DNA may be present uncoated inside the cells (Nguyen et al. 2017; Lehti et al. 2017).
The composition of the phages population in the gut is affected by many factors, particularly those affecting gut microflora composition, such as the host diet. For example, the availability of amino acids promotes the induction of ϕV1/7 bacteriophage in E. faecalis, which seems to be a mechanism by which E. faecalis dominates over other related bacteria in the GIT (Duerkop et al. 2012).
Disease induction by virome
The presence/absence or the alteration of the phage community in the gut may be linked to disease conditions. The increase in their population or change in their composition leads to inflammation of the gut and induction of inflammatory bowel diseases. Alteration in their composition may also lead to type 1 diabetes in children (Zhao et al. 2017; Wagner et al. 2013).
Bacteriophages have been shown to induce IBD through the reduction of bacterial diversity in the gut. A direct role in disease induction was also suspected (Lucas López et al. 2017; Dalmasso et al. 2014). Beside IBD, phages have also incriminated in the induction of other bowel diseases such as Crohn’s disease (Wagner et al. 2013; Norman et al. 2015) or even metabolic diseases besides their role as immune modulators (Burcelin 2016).
Phages were also found to influence human health by increasing gut permeability (leaky gut) and lead to the impairment of the intestinal barrier (Tetz and Tetz 2018; Tetz and Tetz 2016; Lawrence et al. 2019). This, in turn, leads to the escape of toxins into circulation and inflammatory mediators’ production (Bischoff et al. 2014; Fang 2016). Bacteriophages can also directly interfere with the immune system and, by so doing, enhance bacterial infections (Sweere et al. 2019). In addition, the presence of prion-like domains in bacteriophages is thought to play a role in their virulence (Hasegawa et al. 2017; Tetz and Tetz 2017).
The ingestion of bacteriophages with the diet leads to mass destruction of bacteria in the gut. This, in turn, leads to the release of high amounts of bacterial DNA in the circulation and elevates the pathogen-associated molecular pattern (PAMP). Beside the bacterial DNA, free bacteriophage particles, fractions of bacterial LP, amyloid, and other bacterial antigens may enter the circulation. All these elements increase the gut permeability (leaky gut) and irritate the immune system, resulting in the induction of autoimmune diseases, neurodegenerative diseases, and other serious diseases (Aas et al. 2008; Sursal et al. 2013; Wang et al. 2015; de Paepe et al. 2014; Anunobi et al. 2018).
Bacteriophages in the CSF have been reported to induce neurodegenerative such as multiple sclerosis (MS) and autoimmune diseases. This link between the presence of phages in CSF and neurodegenerative diseases is supported by the fact that MS patients suffer additionally from leaky gut and a disrupted BBB, which facilitates phage transmission from the gut to the brain (Tetz and Tetz 2018; McManus and Heneka 2017; Zhou et al. 2013).
The possible role of lytic phages in the induction of neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases or amyotrophic lateral sclerosis was documented. Patients who have Parkinson’s disease showed 10-fold higher concentrations of Lactococcus lytic phages with a severe reduction in the lactococci population in the gut. This is attributed to the killing effect of available c2-like and 936 groups of phages on the Lactococci. The virulent (Abi-escape phage mutants) were provided through dairy products (Tetz et al. 2018; Bäuerl et al. 2018; Braak et al. 2003; Scheperjans 2016).
Medical uses of bacteriophages
It was surprising that short-term oral administration of narrow-spectrum antibiotics did not influence the composition of phages/viruses in the gut. However, it affected the composition of the available bacteria (Ly et al. 2016). Although the treatment of patients with certain antibiotics increases the number of free Clostridium difficile and P. aeruginosa bacteriophages in human feces and sputum, respectively, according to some reports (Fothergill et al. 2011; Thomas 1966), due to the growing challenge of antibiotic resistance among pathogenic bacteria, alternative therapeutic possibilities via phage therapy are being investigated. The phages can infect and lyse the bacteria; in addition, they collaborate with the immune system to clear the invading bacteria. Additional applications of phages in gene therapy are under development at the time (Roach et al. 2017).
Conclusion
The influence of microbiota on our health/disease status is underestimated. Various infectious and non-infectious diseases can be induced by disturbances in the composition, prevalence, and vitality of the microbiota. Manipulation of external factors such as the nature of diet and lifestyle can re-establish the balance among microbiota populations in order to prevent or even treat such diseases. The role played by microbiota in health and disease was highlighted during the COVID-19 pandemic. Nutraceutical non-pharmacological therapeutic and prophylactic approaches could reduce the mortality rate, the diseases severity, and serious complications. The use of probiotics, dietary supplements, and microbiota transplantation from healthy individuals are promising future therapeutic trends for several infectious and non-infectious diseases.
References
Aas JA, Griffen AL, Dardis SR, Lee AM, Olsen I, Dewhirst FE, Leys EJ, Paster BJ (2008) Bacteria of dental caries in primary and permanent teeth in children and young adults▿. J Clin Microbiol 46(4):1407–1417. https://doi.org/10.1128/JCM.01410-07
Aggarwal S, Upadhyay A, Shah D, Teotia N, Agarwal A, Jaiswal V (2014) Lactobacillus GG for treatment of acute childhood diarrhoea: an open labelled, randomized controlled trial. Indian J Med Res 139(3):379–385
Allen HK, Looft T, Bayles DO, Humphrey S, Levine UY, Alt D, Stanton TB (2011) Antibiotics in feed induce prophages in swine fecal microbiomes mBio:2(6). https://doi.org/10.1128/mBio.00260-11
Anunobi R, Boone BA, Cheh N, Tang D, Kang R, Loux T, Lotze MT, Zeh HJ (2018) Extracellular DNA promotes colorectal tumor cell survival after cytotoxic chemotherapy. J Surg Res 226:181–191. https://doi.org/10.1016/j.jss.2018.02.042
Avena NM, Rada P, Hoebel BG (2008) Evidence for sugar addiction: behavioral and neurochemical effects of intermittent, excessive sugar intake. Neurosci Biobehav Rev 32(1):20–39. https://doi.org/10.1016/j.neubiorev.2007.04.019
Avena NM, Rada P, Hoebel BG (2009) Sugar and fat bingeing have notable differences in addictive-like behavior. J Nutr 139(3):623–628. https://doi.org/10.3945/jn.108.097584
Barr JJ, Auro R, Furlan M, Whiteson KL, Erb ML, Pogliano J, Stotland A, Wolkowicz R, Cutting AS, Doran KS, Salamon P, Youle M, Rohwer F (2013) Bacteriophage adhering to mucus provide a non-host-derived immunity. Proc Natl Acad Sci U S A 110(26):10771–10776. https://doi.org/10.1073/pnas.1305923110
Bäuerl C, Collado MC, Diaz Cuevas A, Viña J, Pérez Martínez G (2018) Shifts in gut microbiota composition in an APP/PSS1 transgenic mouse model of Alzheimer's disease during lifespan. Lett Appl Microbiol 66(6):464–471. https://doi.org/10.1111/lam.12882
Belančić A (2020) Gut microbiome dysbiosis and endotoxemia - Additional pathophysiological explanation for increased COVID-19 severity in obesity. Obes Med 20:100302. https://doi.org/10.1016/j.obmed.2020.100302
Belda-Ferre P, Alcaraz LD, Cabrera-Rubio R, Romero H, Simón-Soro A, Pignatelli M, Mira A (2011) The oral metagenome in health and disease. The ISME journal 6(1):46–56. https://doi.org/10.1038/ismej.2011.85
Bik EM, Long CD, Armitage GC, Loomer P, Emerson J, Mongodin EF, Nelson KE, Gill SR, Fraser-Liggett CM, Relman DA (2010) Bacterial diversity in the oral cavity of ten healthy individuals. The ISME journal 4(8):962–974. https://doi.org/10.1038/ismej.2010.30
Bischoff SC, Barbara G, Buurman W, Ockhuizen T, Schulzke J-D, Serino M, Tilg H, Watson A, Wells JM (2014) Intestinal permeability--a new target for disease prevention and therapy. BMC Gastroenterol 14:189. https://doi.org/10.1186/s12876-014-0189-7
Bishehsari F, Mahdavinia M, Vacca M, Malekzadeh R, Mariani-Costantini R (2014) Epidemiological transition of colorectal cancer in developing countries: environmental factors, molecular pathways, and opportunities for prevention. World J Gastroenterol: WJG 20(20):6055–6072. https://doi.org/10.3748/wjg.v20.i20.6055
Boleij A, Hechenbleikner EM, Goodwin AC, Badani R, Stein EM, Lazarev MG, Ellis B, Carroll KC, Albesiano E, Wick EC, Platz EA, Pardoll DM, Sears CL (2015) The Bacteroides fragilis toxin gene is prevalent in the colon mucosa of colorectal cancer patients. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America 60(2):208–215. https://doi.org/10.1093/cid/ciu787
Bongers G, Pacer ME, Geraldino TH, Chen L, He Z, Hashimoto D, Furtado GC, Ochando J, Kelley KA, Clemente JC, Merad M, van Bakel H, Lira SA (2014) Interplay of host microbiota, genetic perturbations, and inflammation promotes local development of intestinal neoplasms in mice. J Exp Med 211(3):457–472. https://doi.org/10.1084/jem.20131587
Bonnet M, Buc E, Sauvanet P, Darcha C, Dubois D, Pereira B, Déchelotte P, Bonnet R, Pezet D, Darfeuille-Michaud A (2014) Colonization of the human gut by E. coli and colorectal cancer risk. Clinical cancer research : an official journal of the American Association for Cancer Research 20(4):859–867. https://doi.org/10.1158/1078-0432.CCR-13-1343
Borges-Canha M, Portela-Cidade JP, Dinis-Ribeiro M, Leite-Moreira AF, Pimentel-Nunes P (2015) Role of colonic microbiota in colorectal carcinogenesis: a systematic review. Revista espanola de enfermedades digestivas. Organo oficial de la Sociedad Espanola de Patologia Digestiva 107(11):659–671. https://doi.org/10.17235/reed.2015.3830/2015
Botka T, Pantůček R, Mašlaňová I, Benešík M, Petráš P, Růžičková V, Havlíčková P, Varga M, Žemličková H, Koláčková I, Florianová M, Jakubů V, Karpíšková R, Doškař J (2019) Lytic and genomic properties of spontaneous host-range Kayvirus mutants prove their suitability for upgrading phage therapeutics against staphylococci. Sci Rep 9(1):5475. https://doi.org/10.1038/s41598-019-41868-w
Braak H, Rüb U, Gai WP, Del Tredici K (2003) Idiopathic Parkinson's disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. Journal of neural transmission (Vienna, Austria : 1996) 110(5):517–536. https://doi.org/10.1007/s00702-002-0808-2
Breitbart M, Hewson I, Felts B, Mahaffy JM, Nulton J, Salamon P, Rohwer F (2003) Metagenomic analyses of an uncultured viral community from human feces. J Bacteriol 185(20):6220–6223. https://doi.org/10.1128/jb.185.20.6220-6223.2003
Buc E, Dubois D, Sauvanet P, Raisch J, Delmas J, Darfeuille-Michaud A, Pezet D, Bonnet R (2013) High prevalence of mucosa-associated E. coli producing cyclomodulin and genotoxin in colon cancer. PLoS One 8 (2):e56964. https://doi.org/10.1371/journal.pone.0056964
Burcelin R (2016) Gut microbiota and immune crosstalk in metabolic disease. Molecular Metabolism 5(9):771–781. https://doi.org/10.1016/j.molmet.2016.05.016
Calvani R, Picca A, Lo Monaco MR, Landi F, Bernabei R, Marzetti E (2018) Of microbes and minds: a narrative review on the second brain aging. Frontiers in medicine 5:53. https://doi.org/10.3389/fmed.2018.00053
Camacho S, Ruppel A (2017) Is the calorie concept a real solution to the obesity epidemic? Glob Health Action 10(1):1289650. https://doi.org/10.1080/16549716.2017.1289650
Castellarin M, Warren RL, Freeman JD, Dreolini L, Krzywinski M, Strauss J, Barnes R, Watson P, Allen-Vercoe E, Moore RA, Holt RA (2012) Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res 22(2):299–306. https://doi.org/10.1101/gr.126516.111
Chen Y, Wang X, Wang J, Yan Z, Luo J (2012) Excess body weight and the risk of primary liver cancer: an updated meta-analysis of prospective studies. European journal of cancer (Oxford, England : 1990) 48(14):2137–2145. https://doi.org/10.1016/j.ejca.2012.02.063
Chen Y, Liu L, Wang X, Wang J, Yan Z, Cheng J, Gong G, Li G (2013) Body mass index and risk of gastric cancer: a meta-analysis of a population with more than ten million from 24 prospective studies. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 22(8):1395–1408. https://doi.org/10.1158/1055-9965.EPI-13-0042
Claesson MJ, Cusack S, O'Sullivan O, Greene-Diniz R, de Weerd H, Flannery E, Marchesi JR, Falush D, Dinan T, Fitzgerald G, Stanton C, van Sinderen D, O'Connor M, Harnedy N, O'Connor K, Henry C, O'Mahony D, Fitzgerald AP, Shanahan F, Twomey C, Hill C, Ross RP, O'Toole PW (2011) Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proceedings of the National Academy of Sciences of the United States of America 108 Suppl 1(Suppl 1):4586–4591. https://doi.org/10.1073/pnas.1000097107
Corredoira J, Alonso MP, Coira A, Casariego E, Arias C, Alonso D, Pita J, Rodriguez A, López MJ, Varela J (2008a) Characteristics of Streptococcus bovis endocarditis and its differences with Streptococcus viridans endocarditis. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology 27(4):285–291. https://doi.org/10.1007/s10096-007-0441-y
Corredoira J, Alonso MP, Coira A, Varela J (2008b) Association between Streptococcus infantarius (formerly S. bovis II/1) bacteremia and noncolonic cancer. J Clin Microbiol 46(4):1570. https://doi.org/10.1128/JCM.00129-08
Dahiya DK, Renuka (2017) The gut virome: a neglected actor in colon cancer pathogenesis. Future Microbiol 12:1345–1348. https://doi.org/10.2217/fmb-2017-0159
Dalmasso M, Hill C, Ross RP (2014) Exploiting gut bacteriophages for human health. Trends Microbiol 22(7):399–405. https://doi.org/10.1016/j.tim.2014.02.010
Damman CJ, Miller SI, Surawicz CM, Zisman TL (2012) The microbiome and inflammatory bowel disease: is there a therapeutic role for fecal microbiota transplantation? Am J Gastroenterol 107(10):1452–1459. https://doi.org/10.1038/ajg.2012.93
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, Ling AV, Devlin AS, Varma Y, Fischbach MA, Biddinger SB, Dutton RJ, Turnbaugh PJ (2014) Diet rapidly and reproducibly alters the human gut microbiome. Nature 505(7484):559–563. https://doi.org/10.1038/nature12820
Davis C, Claridge G (1998) The eating disorders as addiction: a psychobiological perspective. Addict Behav 23(4):463–475
de Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, Collini S, Pieraccini G, Lionetti P (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A 107(33):14691–14696. https://doi.org/10.1073/pnas.1005963107
de Paepe M, Leclerc M, Tinsley CR, Petit M-A (2014) Bacteriophages: an underestimated role in human and animal health? Front Cell Infect Microbiol 4:39. https://doi.org/10.3389/fcimb.2014.00039
Dedrick RM, Guerrero-Bustamante CA, Garlena RA, Russell DA, Ford K, Harris K, Gilmour KC, Soothill J, Jacobs-Sera D, Schooley RT, Hatfull GF, Spencer H (2019) Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat Med 25(5):730–733. https://doi.org/10.1038/s41591-019-0437-z
Ding J-H, Jin Z, Yang X-X, Lou J, Shan W-X, Hu Y-X, Du Q, Liao Q-S, Xie R, Xu J-Y (2020) Role of gut microbiota via the gut-liver-brain axis in digestive diseases. World J Gastroenterol: WJG 26(40):6141–6162. https://doi.org/10.3748/wjg.v26.i40.6141
Dinleyici EC, Eren M, Ozen M, Yargic ZA, Vandenplas Y (2012) Effectiveness and safety of Saccharomyces boulardii for acute infectious diarrhea. Expert Opin Biol Ther 12(4):395–410. https://doi.org/10.1517/14712598.2012.664129
Dominguez-Bello MG, Costello EK, Contreras M, Magris M, Hidalgo G, Fierer N, Knight R (2010) Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci U S A 107(26):11971–11975. https://doi.org/10.1073/pnas.1002601107
Dougan MM, Hankinson SE, de Vivo I, Tworoger SS, Glynn RJ, Michels KB (2015) Prospective study of body size throughout the life-course and the incidence of endometrial cancer among premenopausal and postmenopausal women. Int J Cancer 137(3):625–637. https://doi.org/10.1002/ijc.29427
Dubrow R, Edberg S, Wikfors E, Callan D, Troncale F, Vender R, Brand M, Yapp R (1991) Fecal carriage of Streptococcus bovis and colorectal adenomas. Gastroenterology 101(3):721–725. https://doi.org/10.1016/0016-5085(91)90531-O
Duerkop BA (2018) Bacteriophages shift the focus of the mammalian microbiota. PLoS Pathog 14(10). https://doi.org/10.1371/journal.ppat.1007310
Duerkop BA, Clements CV, Rollins D, Rodrigues JLM, Hooper LV (2012) A composite bacteriophage alters colonization by an intestinal commensal bacterium. Proc Natl Acad Sci U S A 109(43):17621–17626. https://doi.org/10.1073/pnas.1206136109
Duthie SJ (1999) Folic acid deficiency and cancer: mechanisms of DNA instability. Br Med Bull 55(3):578–592. https://doi.org/10.1258/0007142991902646
El Sayed AAA (2000) Molekularbiologische Charakterisierung von zwei Shigatoxin-kodierenden Bakteriophagen enterhämorrhagischer Escherichia coli-Stämme. Uitgever niet vastgesteld
El-Sayed A, Aleya L, Kamel M (2021) The link among microbiota, epigenetics, and disease development. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-13862-1
Fang X (2016) Potential role of gut microbiota and tissue barriers in Parkinson's disease and amyotrophic lateral sclerosis. The International journal of neuroscience 126(9):771–776. https://doi.org/10.3109/00207454.2015.1096271
Feinman RD, Fine EJ (2004) "A calorie is a calorie" violates the second law of thermodynamics. Nutr J 3:9. https://doi.org/10.1186/1475-2891-3-9
Ferreira C, Viana SD, Reis F (2020) Gut microbiota dysbiosis-Immune hyperresponse-inflammation triad in coronavirus disease 2019 (COVID-19): impact of pharmacological and nutraceutical approaches. Microorganisms 8(10):1514. https://doi.org/10.3390/microorganisms8101514
Fothergill JL, Mowat E, Walshaw MJ, Ledson MJ, James CE, Winstanley C (2011) Effect of antibiotic treatment on bacteriophage production by a cystic fibrosis epidemic strain of Pseudomonas aeruginosa. Antimicrob Agents Chemother 55(1):426–428. https://doi.org/10.1128/AAC.01257-10
Friedman SL (2013) Liver fibrosis in 2012: convergent pathways that cause hepatic fibrosis in NASH. Nature reviews. Gastroenterol Hepatol 10(2):71–72. https://doi.org/10.1038/nrgastro.2012.256
Garrido-Cardenas JA, Manzano-Agugliaro F (2017) The metagenomics worldwide research. Curr Genet 63(5):819–829. https://doi.org/10.1007/s00294-017-0693-8
Genkinger JM, Spiegelman D, Anderson KE, Bernstein L, van den Brandt PA, Calle EE, English DR, Folsom AR, Freudenheim JL, Fuchs CS, Giles GG, Giovannucci E, Horn-Ross PL, Larsson SC, Leitzmann M, Männistö S, Marshall JR, Miller AB, Patel AV, Rohan TE, Stolzenberg-Solomon RZ, Verhage BAJ, Virtamo J, Willcox BJ, Wolk A, Ziegler RG, Smith-Warner SA (2011) A pooled analysis of 14 cohort studies of anthropometric factors and pancreatic cancer risk. Int J Cancer 129(7):1708–1717. https://doi.org/10.1002/ijc.25794
Ghaisas S, Maher J, Kanthasamy A (2016) Gut microbiome in health and disease: linking the microbiome-gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases. Pharmacol Ther 158:52–62. https://doi.org/10.1016/j.pharmthera.2015.11.012
Goodwin AC, Destefano Shields CE, Wu S, Huso DL, Wu X, Murray-Stewart TR, Hacker-Prietz A, Rabizadeh S, Woster PM, Sears CL, Casero RA (2011) Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc Natl Acad Sci U S A 108(37):15354–15359. https://doi.org/10.1073/pnas.1010203108
Górski A, Wazna E, Dabrowska B-W, Dabrowska K, Switała-Jeleń K, Miedzybrodzki R (2006) Bacteriophage translocation. FEMS Immunol Med Microbiol 46(3):313–319. https://doi.org/10.1111/j.1574-695X.2006.00044.x
Greiner TU, Hyötyläinen T, Knip M, Bäckhed F, Orešič M (2014) The gut microbiota modulates glycaemic control and serum metabolite profiles in non-obese diabetic mice. PLoS One 9(11):e110359. https://doi.org/10.1371/journal.pone.0110359
Grice EA, Segre JA (2012) The human microbiome: our second genome. Annu Rev Genomics Hum Genet 13:151–170. https://doi.org/10.1146/annurev-genom-090711-163814
Guarner C, Soriano G (2005) Bacterial translocation and its consequences in patients with cirrhosis. Eur J Gastroenterol Hepatol 17(1):27–31
Guo M, Miao M, Wang Y, Duan M, Yang F, Chen Y, Yuan W, Zheng H (2020) Developmental differences in the intestinal microbiota of Chinese 1-year-old infants and 4-year-old children. Sci Rep 10(1):19470. https://doi.org/10.1038/s41598-020-76591-4
Harris RE (2016) Global epidemiology of cancer. Jones & Bartlett Learning, Burlington, MA
Hartstra AV, Bouter KEC, Bäckhed F, Nieuwdorp M (2015) Insights into the role of the microbiome in obesity and type 2 diabetes. Diabetes Care 38(1):159–165. https://doi.org/10.2337/dc14-0769
Hasegawa M, Nonaka T, Masuda-Suzukake M (2017) Prion-like mechanisms and potential therapeutic targets in neurodegenerative disorders. Pharmacol Ther 172:22–33. https://doi.org/10.1016/j.pharmthera.2016.11.010
Heymsfield SB, Thomas D, Martin CK, Redman LM, Strauss B, Bosy-Westphal A, Müller MJ, Shen W, Martin Nguyen A (2012) Energy content of weight loss: kinetic features during voluntary caloric restriction. Metab Clin Exp 61(7):937–943. https://doi.org/10.1016/j.metabol.2011.11.012
Hill JO, Wyatt HR, Peters JC (2012) Energy balance and obesity. Circulation 126(1):126–132. https://doi.org/10.1161/CIRCULATIONAHA.111.087213
Hoebel BG, Avena NM, Bocarsly ME, Rada P (2009) Natural addiction: a behavioral and circuit model based on sugar addiction in rats. J Addict Med 3(1):33–41. https://doi.org/10.1097/ADM.0b013e31819aa621
Holmes AJ, Chew YV, Colakoglu F, Cliff JB, Klaassens E, Read MN, Solon-Biet SM, McMahon AC, Cogger VC, Ruohonen K, Raubenheimer D, Le Couteur DG, Simpson SJ (2017) Diet-microbiome interactions in health are controlled by intestinal nitrogen source constraints. Cell Metab 25(1):140–151. https://doi.org/10.1016/j.cmet.2016.10.021
Homann N, Tillonen J, Salaspuro M (2000) Microbially produced acetaldehyde from ethanol may increase the risk of colon cancer via folate deficiency. Int J Cancer 86(2):169–173. https://doi.org/10.1002/(sici)1097-0215(20000415)86:2<169:aid-ijc4>3.0.co;2-3
Howe A, Ringus DL, Williams RJ, Choo Z-N, Greenwald SM, Owens SM, Coleman ML, Meyer F, Chang EB (2016) Divergent responses of viral and bacterial communities in the gut microbiome to dietary disturbances in mice. The ISME journal 10(5):1217–1227. https://doi.org/10.1038/ismej.2015.183
Hoyo C, Cook MB, Kamangar F, Freedman ND, Whiteman DC, Bernstein L, Brown LM, Risch HA, Ye W, Sharp L, Wu AH, Ward MH, Casson AG, Murray LJ, Corley DA, Nyrén O, Pandeya N, Vaughan TL, Chow W-H, Gammon MD (2012) Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: a pooled analysis from the International BEACON Consortium. Int J Epidemiol 41(6):1706–1718. https://doi.org/10.1093/ije/dys176
Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T, Codelli JA, Chow J, Reisman SE, Petrosino JF, Patterson PH, Mazmanian SK (2013) Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 155(7):1451–1463. https://doi.org/10.1016/j.cell.2013.11.024
Infusino F, Marazzato M, Mancone M, Fedele F, Mastroianni CM, Severino P, Ceccarelli G, Santinelli L, Cavarretta E, Marullo AGM, Miraldi F, Carnevale R, Nocella C, Biondi-Zoccai G, Pagnini C, Schiavon S, Pugliese F, Frati G, d'Ettorre G (2020) Diet supplementation, probiotics, and nutraceuticals in SARS-CoV-2 infection: a scoping review. Nutrients 12(6):1718. https://doi.org/10.3390/nu12061718
Jaacks LM, Vandevijvere S, Pan A, CJ MG, Wallace C, Imamura F, Mozaffarian D, Swinburn B, Ezzati M (2019) The obesity transition: stages of the global epidemic. The Lancet Diabetes & Endocrinology 7(3):231–240. https://doi.org/10.1016/S2213-8587(19)30026-9
Janakiraman M, Krishnamoorthy G (2018) Emerging role of diet and microbiota interactions in neuroinflammation. Front Immunol 9:2067. https://doi.org/10.3389/fimmu.2018.02067
Janda L, Mihalčin M, Šťastná M (2020) Is a healthy microbiome responsible for lower mortality in COVID-19? Biologia (Bratisl):1–11. https://doi.org/10.2478/s11756-020-00614-8
Jéquier E (2002) Pathways to obesity. International journal of obesity and related metabolic disorders : journal of the International Association for the Study of Obesity 26(Suppl 2):S12–S17. https://doi.org/10.1038/sj.ijo.0802123
Jin Q, Black A, Kales SN, Vattem D, Ruiz-Canela M, Sotos-Prieto M (2019) Metabolomics and microbiomes as potential tools to evaluate the effects of the Mediterranean diet. Nutrients 11(1):207. https://doi.org/10.3390/nu11010207
Johnson TA, Looft T, Severin AJ, Bayles DO, Nasko DJ, Wommack KE, Howe A, Allen HK (2017) The In-feed antibiotic Carbadox induces phage gene transcription in the swine gut microbiome. mBio:8(4). https://doi.org/10.1128/mBio.00709-17
Kato I, Boleij A, Kortman GAM, Roelofs R, Djuric Z, Severson RK, Tjalsma H (2013) Partial associations of dietary iron, smoking and intestinal bacteria with colorectal cancer risk. Nutr Cancer 65(2):169–177. https://doi.org/10.1080/01635581.2013.748922
Kim M-S, Park E-J, Roh SW, Bae J-W (2011) Diversity and abundance of single-stranded DNA viruses in human feces. Appl Environ Microbiol 77(22):8062–8070. https://doi.org/10.1128/AEM.06331-11
Kim M-S, Hwang S-S, Park E-J, Bae J-W (2013) Strict vegetarian diet improves the risk factors associated with metabolic diseases by modulating gut microbiota and reducing intestinal inflammation. Environ Microbiol Rep 5(5):765–775. https://doi.org/10.1111/1758-2229.12079
Kitahara CM, McCullough ML, Franceschi S, Rinaldi S, Wolk A, Neta G, Olov Adami H, Anderson K, Andreotti G, Beane Freeman LE, Bernstein L, Buring JE, Clavel-Chapelon F, de Roo LA, Gao Y-T, Gaziano JM, Giles GG, Håkansson N, Horn-Ross PL, Kirsh VA, Linet MS, MacInnis RJ, Orsini N, Park Y, Patel AV, Purdue MP, Riboli E, Robien K, Rohan T, Sandler DP, Schairer C, Schneider AB, Sesso HD, Shu X-O, Singh PN, van den Brandt PA, Ward E, Weiderpass E, White E, Xiang Y-B, Zeleniuch-Jacquotte A, Zheng W, Hartge P, Berrington de González A (2016) Anthropometric factors and thyroid cancer risk by histological subtype: pooled analysis of 22 prospective studies. Thyroid : official journal of the American Thyroid Association 26(2):306–318. https://doi.org/10.1089/thy.2015.0319
Konturek PC, Konturek SJ, Brzozowski T (2009) Helicobacter pylori infection in gastric cancerogenesis. Journal of physiology and pharmacology : an official journal of the Polish Physiological Society 60(3):3–21
Koren O, Spor A, Felin J, Fåk F, Stombaugh J, Tremaroli V, Behre CJ, Knight R, Fagerberg B, Ley RE, Bäckhed F (2011) Human oral, gut, and plaque microbiota in patients with atherosclerosis. Proc Natl Acad Sci U S A 108(Suppl 1):4592–4598. https://doi.org/10.1073/pnas.1011383107
Kowalski K, Mulak A (2019) Brain-gut-microbiota axis in Alzheimer's disease. Journal of neurogastroenterology and motility 25(1):48–60. https://doi.org/10.5056/jnm18087
Kreznar JH, Keller MP, Traeger LL, Rabaglia ME, Schueler KL, Stapleton DS, Zhao W, Vivas EI, Yandell BS, Broman AT, Hagenbuch B, Attie AD, Rey FE (2017) Host genotype and gut microbiome modulate insulin secretion and diet-induced metabolic phenotypes. Cell Rep 18(7):1739–1750. https://doi.org/10.1016/j.celrep.2017.01.062
Kuipers F, Bloks VW, Groen AK (2014) Beyond intestinal soap--bile acids in metabolic control. Nature reviews. Endocrinology 10(8):488–498. https://doi.org/10.1038/nrendo.2014.60
Kumar S, Kumar S, Kumar S (2006) Infection as a risk factor for gallbladder cancer. J Surg Oncol 93(8):633–639. https://doi.org/10.1002/jso.20530
Lawrence D, Baldridge MT, Handley SA (2019) Phages and human health: more than idle hitchhikers. Viruses 11
Lee S, Lee J, Choi IJ, Kim Y-W, Ryu KW, Kim Y-I, Kim J (2018) Dietary n-3 and n-6 polyunsaturated fatty acids, the FADS gene, and the risk of gastric cancer in a Korean population. Sci Rep 8(1):3823. https://doi.org/10.1038/s41598-018-21960-3
Lehti TA, Pajunen MI, Skog MS, Finne J (2017) Internalization of a polysialic acid-binding Escherichia coli bacteriophage into eukaryotic neuroblastoma cells. Nat Commun 8(1):1915. https://doi.org/10.1038/s41467-017-02057-3
Leishman SJ, Do HL, Ford PJ (2010) Cardiovascular disease and the role of oral bacteria. Journal of Oral Microbiology 2. https://doi.org/10.3402/jom.v2i0.5781
Li L, Gan Y, Li W, Wu C, Lu Z (2016) Overweight, obesity and the risk of gallbladder and extrahepatic bile duct cancers: a meta-analysis of observational studies. Obesity (Silver Spring, Md) 24(8):1786–1802. https://doi.org/10.1002/oby.21505
Lim ES, Wang D, Holtz LR (2016) The bacterial microbiome and virome milestones of infant development. Trends Microbiol 24(10):801–810. https://doi.org/10.1016/j.tim.2016.06.001
Lin A, Bik EM, Costello EK, Dethlefsen L, Haque R, Relman DA, Singh U (2013) Distinct distal gut microbiome diversity and composition in healthy children from Bangladesh and the United States. PLoS One 8(1):e53838–e53838. https://doi.org/10.1371/journal.pone.0053838
Lu R, Wu S, Zhang Y-G, Xia Y, Liu X, Zheng Y, Chen H, Schaefer KL, Zhou Z, Bissonnette M, Li L, Sun J (2014) Enteric bacterial protein AvrA promotes colonic tumorigenesis and activates colonic beta-catenin signaling pathway. Oncogenesis 3:e105. https://doi.org/10.1038/oncsis.2014.20
Lucas López R, Grande Burgos MJ, Gálvez A, Pérez Pulido R (2017) The human gastrointestinal tract and oral microbiota in inflammatory bowel disease: a state of the science review. APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 125(1):3–10. https://doi.org/10.1111/apm.12609
Lunn RM, Jahnke GD, Rabkin CS (2017) Tumour virus epidemiology. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 372(1732). https://doi.org/10.1098/rstb.2016.0266
Łusiak-Szelachowska M, Weber-Dąbrowska B, Jończyk-Matysiak E, Wojciechowska R, Górski A (2017) Bacteriophages in the gastrointestinal tract and their implications. Gut pathogens 9:44. https://doi.org/10.1186/s13099-017-0196-7
Ly M, Abeles SR, Boehm TK, Robles-Sikisaka R, Naidu M, Santiago-Rodriguez T, Pride DT (2014) Altered oral viral ecology in association with periodontal disease. mBio 5(3):e01133–e01114. https://doi.org/10.1128/mBio.01133-14
Ly M, Jones MB, Abeles SR, Santiago-Rodriguez TM, Gao J, Chan IC, Ghose C, Pride DT (2016) Transmission of viruses via our microbiomes. Microbiome 4(1):64. https://doi.org/10.1186/s40168-016-0212-z
Ma Y, Yang Y, Wang F, Zhang P, Shi C, Zou Y, Qin H (2013) Obesity and risk of colorectal cancer: a systematic review of prospective studies. PLoS One 8(1):e53916. https://doi.org/10.1371/journal.pone.0053916
Manrique P, Bolduc B, Walk ST, van der Oost J, de Vos WM, Young MJ (2016) Healthy human gut phageome. Proc Natl Acad Sci U S A 113(37):10400–10405. https://doi.org/10.1073/pnas.1601060113
Marley AR, Nan H (2016) Epidemiology of colorectal cancer. International Journal of Molecular Epidemiology and Genetics 7(3):105–114
Marrazzi MA, Luby ED (1986) An auto-addiction opioid model of chronic anorexia nervosa. Int J Eat Disord 5(2):191–208
Martin HM, Campbell BJ, Hart CA, Mpofu C, Nayar M, Singh R, Englyst H, Williams HF, Rhodes JM (2004) Enhanced Escherichia coli adherence and invasion in Crohn's disease and colon cancer. Gastroenterology 127(1):80–93. https://doi.org/10.1053/j.gastro.2004.03.054
Martin OCB, Naud N, Taché S, Debrauwer L, Chevolleau S, Dupuy J, Chantelauze C, Durand D, Pujos-Guillot E, Blas-Y-Estrada F, Urbano C, Kuhnle GGC, Santé-Lhoutellier V, Sayd T, Viala D, Blot A, Meunier N, Schlich P, Attaix D, Guéraud F, Scislowski V, Corpet DE, Pierre FHF (2018) Targeting colon luminal lipid peroxidation limits colon carcinogenesis associated with red meat consumption. Cancer Prev Res (Phila) 11(9):569–580. https://doi.org/10.1158/1940-6207.CAPR-17-0361
McManus RM, Heneka MT (2017) Role of neuroinflammation in neurodegeneration: new insights. Alzheimers Res Ther 9(1):14. https://doi.org/10.1186/s13195-017-0241-2
Micenková L, Bosák J, Smatana S, Novotný A, Budinská E, Šmajs D (2019) Administration of the probiotic Escherichia coli strain A0 34/86 resulted in a stable colonization of the human intestine during the first year of life. Probiotics and antimicrobial proteins. https://doi.org/10.1007/s12602-019-09548-3
Minot S, Grunberg S, Wu GD, Lewis JD, Bushman FD (2012) Hypervariable loci in the human gut virome. Proc Natl Acad Sci U S A 109(10):3962–3966. https://doi.org/10.1073/pnas.1119061109
Mohr AE, Jäger R, Carpenter KC, Kerksick CM, Purpura M, Townsend JR, West NP, Black K, Gleeson M, Pyne DB, Wells SD, Arent SM, Kreider RB, Campbell BI, Bannock L, Scheiman J, Wissent CJ, Pane M, Kalman DS, Pugh JN, Ortega-Santos CP, Ter Haar JA, Arciero PJ, Antonio J (2020) The athletic gut microbiota. J Int Soc Sports Nutr 17(1):24. https://doi.org/10.1186/s12970-020-00353-w
Møller-Olsen C, Ho SFS, Shukla RD, Feher T, Sagona AP (2018) Engineered K1F bacteriophages kill intracellular Escherichia coli K1 in human epithelial cells. Sci Rep 8(1):17559. https://doi.org/10.1038/s41598-018-35859-6
Moore WE, Moore LH (1995) Intestinal floras of populations that have a high risk of colon cancer. Appl Environ Microbiol 61(9):3202–3207
Morales-Oyarvide V, Meyerhardt JA, Ng K (2016) Vitamin D and physical activity in patients with colorectal cancer: epidemiological evidence and therapeutic implications. Cancer journal (Sudbury, Mass) 22(3):223–231. https://doi.org/10.1097/PPO.0000000000000197
Mueller S, Saunier K, Hanisch C, Norin E, Alm L, Midtvedt T, Cresci A, Silvi S, Orpianesi C, Verdenelli MC, Clavel T, Koebnick C, Zunft H-JF, Doré J, Blaut M (2006) Differences in fecal microbiota in different European study populations in relation to age, gender, and country: a cross-sectional study. Appl Environ Microbiol 72(2):1027–1033. https://doi.org/10.1128/AEM.72.2.1027-1033.2006
Mui UN, Haley CT, Tyring SK (2017) Viral oncology: molecular biology and pathogenesis. J Clin Med 6(12). https://doi.org/10.3390/jcm6120111
Muniesa M, Jofre J (2014) Identifying and analyzing bacteriophages in humMuniesa, Maite; Jofre, Juanan fecal samples: what could we discover? Future Microbiol 9(7):879–886. https://doi.org/10.2217/fmb.14.47
Munsell MF, Sprague BL, Berry DA, Chisholm G, Trentham-Dietz A (2014) Body mass index and breast cancer risk according to postmenopausal estrogen-progestin use and hormone receptor status. Epidemiol Rev 36:114–136. https://doi.org/10.1093/epirev/mxt010
Mutlu EA, Gillevet PM, Rangwala H, Sikaroodi M, Naqvi A, Engen PA, Kwasny M, Lau CK, Keshavarzian A (2012) Colonic microbiome is altered in alcoholism. American journal of physiology. Gastrointestinal and liver physiology 302(9):G966–G978. https://doi.org/10.1152/ajpgi.00380.2011
Nagaraja V, Eslick GD (2014) Systematic review with meta-analysis: the relationship between chronic Salmonella typhi carrier status and gall-bladder cancer. Aliment Pharmacol Ther 39(8):745–750. https://doi.org/10.1111/apt.12655
Nguyen S, Baker K, Padman BS, Patwa R, Dunstan RA, Weston TA, Schlosser K, Bailey B, Lithgow T, Lazarou M, Luque A, Rohwer F, Blumberg RS, Barr JJ (2017) Bacteriophage transcytosis provides a mechanism to cross epithelial cell layers. mBio:8(6). https://doi.org/10.1128/mBio.01874-17
Niedermaier T, Behrens G, Schmid D, Schlecht I, Fischer B, Leitzmann MF (2015) Body mass index, physical activity, and risk of adult meningioma and glioma: a meta-analysis. Neurology 85(15):1342–1350. https://doi.org/10.1212/WNL.0000000000002020
Nielsen SD, Christensen JJE, Laerkeborg A, Haunsø S, Knudsen JD (2007) Molekylaerbiologisk metodik anvendt til påvisning af colonrelateret Streptococcus bovis-endokarditis (Molecular-biological methods of diagnosing colon-related Streptococcus bovis endocarditis). Ugeskr Laeger 169(7):610–611
Norman JM, Handley SA, Baldridge MT, Droit L, Liu CY, Keller BC, Kambal A, Monaco CL, Zhao G, Fleshner P, Stappenbeck TS, McGovern DPB, Keshavarzian A, Mutlu EA, Sauk J, Gevers D, Xavier RJ, Wang D, Parkes M, Virgin HW (2015) Disease-specific alterations in the enteric virome in inflammatory bowel disease. Cell 160(3):447–460. https://doi.org/10.1016/j.cell.2015.01.002
Odamaki T, Kato K, Sugahara H, Hashikura N, Takahashi S, Xiao J-Z, Abe F, Osawa R (2016) Age-related changes in gut microbiota composition from newborn to centenarian: a cross-sectional study. BMC Microbiol 16:90. https://doi.org/10.1186/s12866-016-0708-5
Ovarian cancer and body size (n.d.) Iindividual participant meta-analysis including 25,157 women with ovarian cancer from 47 epidemiological studies. PLoS Med 9(4):e1001200. https://doi.org/10.1371/journal.pmed.1001200
Pak HH, Cummings NE, Green CL, Brinkman JA, Yu D, Tomasiewicz JL, Yang SE, Boyle C, Konon EN, Ong IM, Lamming DW (2019) The metabolic response to a low amino acid diet is independent of diet-induced shifts in the composition of the gut microbiome. Sci Rep 9(1):67. https://doi.org/10.1038/s41598-018-37177-3
Parkin DM (2006) The global health burden of infection-associated cancers in the year 2002. Int J Cancer 118(12):3030–3044. https://doi.org/10.1002/ijc.21731
Patrakka O, Pienimäki J-P, Tuomisto S, Ollikainen J, Lehtimäki T, Karhunen PJ, Martiskainen M (2019) Oral bacterial signatures in cerebral thrombi of patients with acute ischemic stroke treated with thrombectomy. J Am Heart Assoc 8(11):e012330. https://doi.org/10.1161/JAHA.119.012330
Pellanda P, Ghosh TS, O’Toole PW (2021) Understanding the impact of age-related changes in the gut microbiome on chronic diseases and the prospect of elderly-specific dietary interventions. Curr Opin Biotechnol 70:48–55. https://doi.org/10.1016/j.copbio.2020.11.001
Pellegrini C, Antonioli L, Colucci R, Blandizzi C, Fornai M (2018) Interplay among gut microbiota, intestinal mucosal barrier and enteric neuro-immune system: a common path to neurodegenerative diseases? Acta Neuropathol 136(3):345–361. https://doi.org/10.1007/s00401-018-1856-5
Pelucchi C, Tramacere I, Boffetta P, Negri E, La Vecchia C (2011) Alcohol consumption and cancer risk. Nutr Cancer 63(7):983–990. https://doi.org/10.1080/01635581.2011.596642
Penders J, Thijs C, Vink C, Stelma FF, Snijders B, Kummeling I, van den Brandt PA, Stobberingh EE (2006) Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 118(2):511–521. https://doi.org/10.1542/peds.2005-2824
Potter MA, Cunliffe NA, Smith M, Miles RS, Flapan AD, Dunlop MG (1998) A prospective controlled study of the association of Streptococcus bovis with colorectal carcinoma. J Clin Pathol 51(6):473–474. https://doi.org/10.1136/jcp.51.6.473
Pride DT, Salzman J, Haynes M, Rohwer F, Davis-Long C, White RA, Loomer P, Armitage GC, Relman DA (2012) Evidence of a robust resident bacteriophage population revealed through analysis of the human salivary virome. The ISME journal 6(5):915–926. https://doi.org/10.1038/ismej.2011.169
Priyadarshini M, Villa SR, Fuller M, Wicksteed B, Mackay CR, Alquier T, Poitout V, Mancebo H, Mirmira RG, Gilchrist A, Layden BT (2015) An acetate-specific GPCR, FFAR2, regulates insulin secretion. Molecular endocrinology (Baltimore, Md) 29(7):1055–1066. https://doi.org/10.1210/me.2015-1007
Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, Peng Y, Zhang D, Jie Z, Wu W, Qin Y, Xue W, Li J, Han L, Lu D, Wu P, Dai Y, Sun X, Li Z, Tang A, Zhong S, Li X, Chen W, Xu R, Wang M, Feng Q, Gong M, Yu J, Zhang Y, Zhang M, Hansen T, Sanchez G, Raes J, Falony G, Okuda S, Almeida M, LeChatelier E, Renault P, Pons N, Batto J-M, Zhang Z, Chen H, Yang R, Zheng W, Li S, Yang H, Wang J, Ehrlich SD, Nielsen R, Pedersen O, Kristiansen K, Wang J (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490(7418):55–60. https://doi.org/10.1038/nature11450
Rada P, Avena NM, Hoebel BG (2005) Daily bingeing on sugar repeatedly releases dopamine in the accumbens shell. Neuroscience 134(3):737–744. https://doi.org/10.1016/j.neuroscience.2005.04.043
Redondo-Useros N, Nova E, González-Zancada N, Díaz LE, Gómez-Martínez S, Marcos A (2020) Microbiota and lifestyle: a special focus on diet. Nutrients 12(6):1776. https://doi.org/10.3390/nu12061776
Rishi P, Thakur K, Vij S, Rishi L, Singh A, Kaur IP, Patel SKS, Lee J-K, Kalia VC (2020) Diet, gut microbiota and COVID-19. Indian J Microbiol 60(4):1–10. https://doi.org/10.1007/s12088-020-00908-0
Roach DR, Leung CY, Henry M, Morello E, Singh D, Di Santo JP, Weitz JS, Debarbieux L (2017) Synergy between the host immune system and bacteriophage is essential for successful phage therapy against an acute respiratory pathogen. Cell Host Microbe 22(1):38–47.e4. https://doi.org/10.1016/j.chom.2017.06.018
Sanfilippo KM, McTigue KM, Fidler CJ, Neaton JD, Chang Y, Fried LF, Liu S, Kuller LH (2014) Hypertension and obesity and the risk of kidney cancer in 2 large cohorts of US men and women. Hypertension (Dallas, Tex : 1979) 63(5):934–941. https://doi.org/10.1161/HYPERTENSIONAHA.113.02953
Sayin SI, Wahlström A, Felin J, Jäntti S, Marschall H-U, Bamberg K, Angelin B, Hyötyläinen T, Orešič M, Bäckhed F (2013) Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab 17(2):225–235. https://doi.org/10.1016/j.cmet.2013.01.003
Scanlan PD, Shanahan F, Marchesi JR (2009) Culture-independent analysis of desulfovibrios in the human distal colon of healthy, colorectal cancer and polypectomized individuals. FEMS Microbiol Ecol 69(2):213–221. https://doi.org/10.1111/j.1574-6941.2009.00709.x
Scarpellini E, Ianiro G, Attili F, Bassanelli C, de Santis A, Gasbarrini A (2015) The human gut microbiota and virome: potential therapeutic implications. Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver 47(12):1007–1012. https://doi.org/10.1016/j.dld.2015.07.008
Scheperjans F (2016) Gut microbiota, 1013 new pieces in the Parkinson's disease puzzle. Curr Opin Neurol 29(6):773–780. https://doi.org/10.1097/WCO.0000000000000389
Sharara AI, Abou Hamdan T, Malli A, El-Halabi MM, Hashash JG, Ghaith OA, Kanj SS (2013) Association of Streptococcus bovis endocarditis and advanced colorectal neoplasia: a case-control study. J Dig Dis 14(7):382–387. https://doi.org/10.1111/1751-2980.12059
Shively CA, Register TC, Appt SE, Clarkson TB, Uberseder B, Clear KYJ, Wilson AS, Chiba A, Tooze JA, Cook KL (2018) Consumption of Mediterranean versus Western diet Leads to distinct mammary gland microbiome populations. Cell Rep 25(1):47–56.e3. https://doi.org/10.1016/j.celrep.2018.08.078
Signat B, Roques C, Poulet P, Duffaut D (2011) Fusobacterium nucleatum in periodontal health and disease. Current issues in molecular biology 13(2):25–36
Sokol H, Pigneur B, Watterlot L, Lakhdari O, Bermúdez-Humarán LG, Gratadoux J-J, Blugeon S, Bridonneau C, Furet J-P, Corthier G, Grangette C, Vasquez N, Pochart P, Trugnan G, Thomas G, Blottière HM, Doré J, Marteau P, Seksik P, Langella P (2008) Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci U S A 105(43):16731–16736. https://doi.org/10.1073/pnas.0804812105
Sordi L, de Khanna V, Debarbieux L (2017) The gut microbiota facilitates drifts in the genetic diversity and infectivity of bacterial viruses. Cell Host Microbe 22(6):801–808.e3. https://doi.org/10.1016/j.chom.2017.10.010
Srivastava AS, Chauhan DP, Carrier E (2004) In utero detection of T7 phage after systemic administration to pregnant mice. BioTechniques 37(1):81–83. https://doi.org/10.2144/04371ST04
Stinson LF, Boyce MC, Payne MS, Keelan JA (2019) The not-so-sterile womb: evidence that the human fetus is exposed to bacteria prior to birth. Front Microbiol:10. https://doi.org/10.3389/fmicb.2019.01124
Strauss J, Kaplan GG, Beck PL, Rioux K, Panaccione R, Devinney R, Lynch T, Allen-Vercoe E (2011) Invasive potential of gut mucosa-derived Fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm Bowel Dis 17(9):1971–1978. https://doi.org/10.1002/ibd.21606
Sun J, Kato I (2016) Gut microbiota, inflammation and colorectal cancer. Genes & Diseases 3(2):130–143. https://doi.org/10.1016/j.gendis.2016.03.004
Sursal T, Stearns-Kurosawa DJ, Itagaki K, Oh S-Y, Sun S, Kurosawa S, Hauser CJ (2013) Plasma bacterial and mitochondrial DNA distinguish bacterial sepsis from sterile SIRS and quantify inflammatory tissue injury in nonhuman primates. Shock (Augusta, Ga) 39(1):55–62. https://doi.org/10.1097/SHK.0b013e318276f4ca
Suzuki TA, Worobey M (2014) Geographical variation of human gut microbial composition. Biol Lett 10(2):20131037. https://doi.org/10.1098/rsbl.2013.1037
Suzuki T, Yoshida S, Hara H (2008) Physiological concentrations of short-chain fatty acids immediately suppress colonic epithelial permeability. Br J Nutr 100(2):297–305. https://doi.org/10.1017/S0007114508888733
Sweere JM, van Belleghem JD, Ishak H, Bach MS, Popescu M, Sunkari V, Kaber G, Manasherob R, Suh GA, Cao X, Vries CR, de Lam DN, Marshall PL, Birukova M, Katznelson E, Lazzareschi DV, Balaji S, Keswani SG, Hawn TR, Secor PR, Bollyky PL (2019) Bacteriophage trigger antiviral immunity and prevent clearance of bacterial infection. Science (New York, NY) 363(6434). https://doi.org/10.1126/science.aat9691
Swidsinski A, Dörffel Y, Loening-Baucke V, Theissig F, Rückert JC, Ismail M, Rau WA, Gaschler D, Weizenegger M, Kühn S, Schilling J, Dörffel WV (2011) Acute appendicitis is characterised by local invasion with Fusobacterium nucleatum/necrophorum. Gut 60(1):34–40. https://doi.org/10.1136/gut.2009.191320
Tahara T, Yamamoto E, Suzuki H, Maruyama R, Chung W, Garriga J, Jelinek J, Yamano H, Sugai T, An B, Shureiqi I, Toyota M, Kondo Y, Estécio MRH, Issa J-PJ (2014) Fusobacterium in colonic flora and molecular features of colorectal carcinoma. Cancer Res 74(5):1311–1318. https://doi.org/10.1158/0008-5472.CAN-13-1865
Teitelbaum JE, Triantafyllopoulou M (2006) Inflammatory bowel disease and Streptococcus bovis. Dig Dis Sci 51(8):1439–1442. https://doi.org/10.1007/s10620-005-9053-5
Tetz G, Tetz V (2016) Bacteriophage infections of microbiota can lead to leaky gut in an experimental rodent model. Gut Pathog 8(1):33. https://doi.org/10.1186/s13099-016-0109-1
Tetz G, Tetz V (2017) Prion-like domains in phagobiota. Front Microbiol 8:2239. https://doi.org/10.3389/fmicb.2017.02239
Tetz G, Tetz V (2018) Bacteriophages as new human viral pathogens. Microorganisms 6(2). https://doi.org/10.3390/microorganisms6020054
Tetz G, Brown S, Hao Y, Tetz V (2018) Parkinson’s disease and bacteriophages as its overlooked contributors
Thomas R (1966) Control of development in temperate bacteriophages: I. Induction of prophage genes following hetero-immune super-infection. J Mol Biol 22(1):79–95. https://doi.org/10.1016/0022-2836(66)90181-1
Thun MJ, Linet MS, Cerhan JR, Haiman CA, Schottenfeld D (2018) Schottenfeld and Fraumeni cancer epidemiology and prevention, Fourth edition. / edited by Michael Thun, Martha S. Linet, James R. Cerhan, Christopher A. Haiman and David Schottenfeld. Oxford University Press, New York, NY
Tirandaz H, Mohammadi E (2013) Efficient tumor targeting by anaerobic butyrate-producing bacteria. Med Hypotheses 80(5):675–678. https://doi.org/10.1016/j.mehy.2013.01.024
Tremlett H, Bauer KC, Appel-Cresswell S, Finlay BB, Waubant E (2017) The gut microbiome in human neurological disease: a review. Ann Neurol 81(3):369–382. https://doi.org/10.1002/ana.24901
Tursi A, Papa A (2020) Intestinal microbiome modulation during COVID-19: another chance to manage the disease? Gastroenterology:S0016–5085(20)35155-6. https://doi.org/10.1053/j.gastro.2020.08.056
Ukena SN, Singh A, Dringenberg U, Engelhardt R, Seidler U, Hansen W, Bleich A, Bruder D, Franzke A, Rogler G, Suerbaum S, Buer J, Gunzer F, Westendorf AM (2007) Probiotic Escherichia coli Nissle 1917 inhibits leaky gut by enhancing mucosal integrity. PLoS One 2(12). https://doi.org/10.1371/journal.pone.0001308
Veerappan GR, Betteridge J, Young PE (2012) Probiotics for the treatment of inflammatory bowel disease. Current gastroenterology reports 14(4):324–333. https://doi.org/10.1007/s11894-012-0265-5
Verdi S, Jackson MA, Beaumont M, Bowyer RCE, Bell JT, Spector TD, Steves CJ (2018) An investigation into physical frailty as a link between the gut microbiome and cognitive health. Front Aging Neurosci 10:398. https://doi.org/10.3389/fnagi.2018.00398
Volkow ND, Wang G-J, Fowler JS, Telang F (2008) Overlapping neuronal circuits in addiction and obesity: evidence of systems pathology. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 363(1507):3191–3200. https://doi.org/10.1098/rstb.2008.0107
Volpato M, Hull MA (2018) Omega-3 polyunsaturated fatty acids as adjuvant therapy of colorectal cancer. Cancer Metastasis Rev 37(2-3):545–555. https://doi.org/10.1007/s10555-018-9744-y
Wagner J, Maksimovic J, Farries G, Sim WH, Bishop RF, Cameron DJ, Catto-Smith AG, Kirkwood CD (2013) Bacteriophages in gut samples from pediatric Crohn's disease patients: metagenomic analysis using 454 pyrosequencing. Inflamm Bowel Dis 19(8):1598–1608. https://doi.org/10.1097/MIB.0b013e318292477c
Wallin A, Larsson SC (2011) Body mass index and risk of multiple myeloma: a meta-analysis of prospective studies. European journal of cancer (Oxford, England : 1990) 47(11):1606–1615. https://doi.org/10.1016/j.ejca.2011.01.020
Walton GE, Gibson GR, Hunter KA (2020) Mechanisms linking the human gut microbiome to prophylactic and treatment strategies for COVID-19. Br J Nutr:1–9. https://doi.org/10.1017/S0007114520003980
Wang F, Xu Y (2014) Body mass index and risk of renal cell cancer: a dose-response meta-analysis of published cohort studies. Int J Cancer 135(7):1673–1686. https://doi.org/10.1002/ijc.28813
Wang G-J, Volkow ND, Thanos PK, Fowler JS (2004) Similarity between obesity and drug addiction as assessed by neurofunctional imaging: a concept review. J Addict Dis 23(3):39–53. https://doi.org/10.1300/J069v23n03_04
Wang L, Llorente C, Hartmann P, Yang A-M, Chen P, Schnabl B (2015) Methods to determine intestinal permeability and bacterial translocation during liver disease. J Immunol Methods 421:44–53. https://doi.org/10.1016/j.jim.2014.12.015
Warren RL, Freeman DJ, Pleasance S, Watson P, Moore RA, Cochrane K, Allen-Vercoe E, Holt RA (2013) Co-occurrence of anaerobic bacteria in colorectal carcinomas. Microbiome 1(1):16. https://doi.org/10.1186/2049-2618-1-16
Woodmansey EJ (2007) Intestinal bacteria and ageing. J Appl Microbiol 102(5):1178–1186. https://doi.org/10.1111/j.1365-2672.2007.03400.x
Wu K, Giovannucci E, Byrne C, Platz EA, Fuchs C, Willett WC, Sinha R (2006) Meat mutagens and risk of distal colon adenoma in a cohort of U.S. men. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 15(6):1120–1125. https://doi.org/10.1158/1055-9965.EPI-05-0782
Wu S, Rhee K-J, Albesiano E, Rabizadeh S, Wu X, Yen H-R, Huso DL, Brancati FL, Wick E, McAllister F, Housseau F, Pardoll DM, Sears CL (2009) A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat Med 15(9):1016–1022. https://doi.org/10.1038/nm.2015
Wu Q, Yang Z-P, Xu P, Gao L-C, Fan D-M (2013) Association between Helicobacter pylori infection and the risk of colorectal neoplasia: a systematic review and meta-analysis. Colorectal disease : the official journal of the Association of Coloproctology of Great Britain and Ireland 15(7):e352–e364. https://doi.org/10.1111/codi.12284
Wu H, Esteve E, Tremaroli V, Khan MT, Caesar R, Mannerås-Holm L, Ståhlman M, Olsson LM, Serino M, Planas-Fèlix M, Xifra G, Mercader JM, Torrents D, Burcelin R, Ricart W, Perkins R, Fernàndez-Real JM, Bäckhed F (2017) Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat Med 23(7):850–858. https://doi.org/10.1038/nm.4345
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI (2012) Human gut microbiome viewed across age and geography. Nature 486(7402):222–227. https://doi.org/10.1038/nature11053
Yu H-S, Lee N-K, Choi A-J, Choe J-S, Bae CH, Paik H-D (2019) Antagonistic and antioxidant effect of probiotic Weissella cibaria JW15. Food Sci Biotechnol 28(3):851–855. https://doi.org/10.1007/s10068-018-0519-6
Yusof N, Hamid N, Ma ZF, Lawenko RM, Wan M, Zahiruddin WM, Collins DA, Liong MT, Odamaki T, Xiao J, Lee YY (2017) Exposure to environmental microbiota explains persistent abdominal pain and irritable bowel syndrome after a major flood. Gut pathogens 9:75. https://doi.org/10.1186/s13099-017-0224-7
Zhang X, Shen D, Fang Z, Jie Z, Qiu X, Zhang C, Chen Y, Ji L (2013) Human gut microbiota changes reveal the progression of glucose intolerance. PLoS One 8(8):e71108. https://doi.org/10.1371/journal.pone.0071108
Zhao G, Vatanen T, Droit L, Park A, Kostic AD, Poon TW, Vlamakis H, Siljander H, Härkönen T, Hämäläinen A-M, Peet A, Tillmann V, Ilonen J, Wang D, Knip M, Xavier RJ, Virgin HW (2017) Intestinal virome changes precede autoimmunity in type I diabetes-susceptible children. Proc Natl Acad Sci U S A 114(30):E6166–E6175. https://doi.org/10.1073/pnas.1706359114
Zhou L, Miranda-Saksena M, Saksena NK (2013) Viruses and neurodegeneration. Virol J 10:172. https://doi.org/10.1186/1743-422X-10-172
Zhu Y, Lin X, Zhao F, Shi X, Li H, Li Y, Zhu W, Xu X, Li C, Zhou G (2015) Meat, dairy and plant proteins alter bacterial composition of rat gut bacteria. Sci Rep 5:15220. https://doi.org/10.1038/srep15220
Zimmermann P, Curtis N (2020) Why is COVID-19 less severe in children? A review of the proposed mechanisms underlying the age-related difference in severity of SARS-CoV-2 infections. Archives of Disease in Childhood:archdischild-2020-320338. https://doi.org/10.1136/archdischild-2020-320338
Availability of data and materials
Not applicable
Author information
Authors and Affiliations
Contributions
All authors had contributed, read, critically revised, and finally approved the final version.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Sayed, A., Aleya, L. & Kamel, M. Microbiota’s role in health and diseases. Environ Sci Pollut Res 28, 36967–36983 (2021). https://doi.org/10.1007/s11356-021-14593-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-14593-z