Abstract
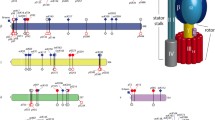
Proteomic analyses of the β subunit of the plastid ATP synthase of barley (Hordeum vulgare L.) revealed that mature protein was not carboxy terminus processed and suggested the correction of the 274 codon (GAT to AAT) in the data bank that was confirmed by DNA sequencing. Six isoforms of the ATP synthase β subunit with pI ranging from 4.95 to 5.14 were resolved by two-dimensional electrophoresis (2-DE). Mass spectrometry analyses indicated that the six isoforms differ in their phosphorylation degree, which was confirmed by the disappearance of more acidic forms after incubation with the protein phosphatase calcineurin. Six Ser and/or Thr were detected as phosphorylated, among them the conserved Thr-179 that is also phosphorylated in the β subunit of human mitochondria. The results are discussed in relation with the proposed regulation of the ATP synthase by phosphorylation and 14-3-3 proteins.
Similar content being viewed by others
Abbreviations
- 2-DE:
-
two-dimensional electrophoresis
- ESI-MS/MS:
-
electro spray ionisation tandem mass spectrometry
- IEF:
-
isoelectric focusing
- MALDI-TOF:
-
matrix-assisted laser desorption/ionisation time-of-flight
References
Abrahams JP, Leslie AGW, Lutter R, Walker JE (1994) Structure at 2.8 Á resolution of F1-ATPase from bovine heart mitochondria. Nature 370:621–628
Berwick DC, Tavaré JM (2004) Identifying protein kinase substrates: hunting for the organ-grinder´s monkey. Trends Biochem Sci 29:227–232
Blom N, Gammeltoft S, Brunak S (1999) Sequence- and structure-based prediction of eukaryotic protein phosphorylation sites. J Mol Biol 294:1351–1362
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Bunney TD, van Walraven HS, de Boer AH (2001) 14–3–3 protein is a regulator of the mitochondrial and chloroplast ATP synthase. Proc Natl Acad Sci USA 98:4249–4554
del Campo EM, Sabater B, Martín M (2002) Post-transcriptional control of chloroplast gene expression: accumulation of stable psaC mRNA is due to downstream cleavages in the ndhD gene. J Biol Chem 277:36457–36464
Casano LM, Lascano HR, Martín M, Sabater B (2004) Topology of the plastid Ndh complex and its NDH-F subunit in thylakoid membranes. Biochem J 382:145–155
Doyle JJ, Doyle L (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Ebbert V, Demmig-Adams B, Adams WW 3rd, Mueth KE, Staehelin LA (2001) Correlation between persistent forms of zeaxanthin-dependent energy dissipation and thylakoid protein phosphorylation. Photosynth Res 67:63–78
Friso G, Giacomelli L, Ytterberg AJ, Peltier J-B, Rudella A, Sun Q, van Wijk KJ (2004) In-depth analysis of the thylakoid membrane proteome of Arabidopsis thaliana chloroplasts: new proteins, new functions, a plastid proteome database. Plant Cell 16:478–499
Groth G, Pohl E (2001) The structure of the chloroplast F1-ATPase at 3.2 Å resolution. J Biol Chem 276:1345–1352
Guex N, Peitsch MC (1997) Swiss-MODEL and the Swiss-Pdb viewer: an environment for comparative protein modelling. Electrophoresis 18:2714–2723
Højlund K, Wrzesinski K, Larsen PM, Fey SJ, Roepstorff P, Handberg A, Dela F, Vinten J, McCormack JG, Reynet C, Beck-Nielsen H (2003) Proteome analysis reveals phosphorylation of ATP synthase β subunit in human skeletal muscle and proteins with potential roles in type 2 diabetes. J Biol Chem 278:10436–10442
Kanekatsu M, Saito H, Motohashi K, Hisabori T (1998) The beta subunit of chloroplast ATP synthase (CF0CF1-ATPase) is phosphorylated by casein kinase II. Biochem Mol Biol Int 46:99–105
Lascano HR, Casano LM, Martín M, Sabater B (2003) The activity of the chloroplastic Ndh complex is regulated by phosphorylation of the NDH-F subunit. Plant Physiol 132:256–262
Laugesen S, Bergoin A, Rossignol M (2004) Deciphering the plant phosphoproteome: tools and strategies for a challenging task. Plant Physiol Biochem 42:929–936
Mackintosh C (2004) Dynamic interactions between 14–3–3 proteins and phosphoproteins regulate diverse cellular processes. Biochem J 381:329–342
Peitsch MC (1995) Protein modelling by E-mail. Bio/Technology 13:658–660
Peltier JB, Friso G, Kalume DE, Roepstorff P, Nilsson F, Adamska I, van Wijk KJ (2000) Proteomic of the chloroplast: systematic identification and targeting analysis of luminal and peripheral thylakoid proteins. Plant Cell 12:319–341
Schwede T, Kopp J, Guex N, Peitsch MC (2003) SWISS-MODEL: an automated protein homology-modelling server. Nucleic Acids Res 31:3381–3385
Sehnke PC, DeLille JM, Ferl RJ (2002) Consumating signal transduction: the role of 14–3–3 proteins in the completion of signal-induced transitions in protein activity. Plant Cell 14:S339–S354
Sehnke PC, Henry R, Cline K, Ferl RJ (2000) Interaction of a plant 14–3–3 protein with the signal peptide of a thylakoid-targeted precursor protein and the presence of 14–3–3 isoforms in the chloroplast stroma. Plant Physiol 122:235–241
Stock D, Leslie AGW, Walker JE (1999) Molecular architecture of the rotary motor in ATP synthase. Science 286:1700–1705
van Wijk KJ (2004) Plastid proteomics. Plant Physiol Biochem 42:963–977
Zhang H, Yan W, Aebersold R (2004) Chemical probes and tandem mass spectrometry: a strategy for the quantitative analyses of proteomes and subproteomes. Curr Op Chem Biol 8:66–75
Acknowledgements
This work was supported by a Grant from the Spanish DGICYT (BMC2003-01261). G. del Riego is recipient of a fellowship of the Spanish MEC. We thank P.H. Serrot for his help in performing DNA sequencing and for English corrections.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
del Riego, G., Casano, L.M., Martín, M. et al. Multiple phosphorylation sites in the β subunit of thylakoid ATP synthase. Photosynth Res 89, 11–18 (2006). https://doi.org/10.1007/s11120-006-9078-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-006-9078-4