ABSTRACT
Purpose
Present study was undertaken to gain insights into the mechanism of cell cycle arrest by ginseng saponin ginsenoside Rh2 (Rh2) using MCF-7 and MDA-MB-231 breast cancer cells.
Methods
Cell viability and cell cycle distribution were determined by trypan blue dye exclusion assay and flow cytometry, respectively. Immunoblotting was performed to determine changes in protein levels. Knockdown of desired protein was achieved by transfection with small interfering RNA (siRNA).
Results
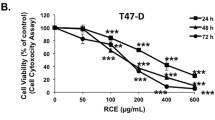
Rh2 treatment significantly inhibited viability of both cells in a concentration-dependent manner, which correlated with G0/G1 phase cell cycle arrest. Rh2-mediated cell cycle arrest was accompanied by down-regulation of cyclin-dependent kinases (Cdk) and cyclins leading to decreased interaction between cyclin D1 and Cdk4/Cdk6 and increased recruitment of p15Ink4B and p27Kip1 to cyclin D1/Cdk4 and cyclin D1/Cdk6 complexes. In addition, Rh2 treatment markedly reduced the levels of phosphorylated retinoblastoma protein (P-Rb) and decreased transcriptional activity of E2F1 in luciferase reporter assay. Rh2-induced cell cycle arrest was significantly attenuated by knockdown of p15Ink4B and/or p27Kip1 proteins.
Conclusions
Rh2-mediated cell cycle arrest in human breast cancer cells is caused by p15Ink4B and p27Kip1-dependent inhibition of kinase activities of G1-S specific Cdks/cyclin complexes.







Similar content being viewed by others
REFERENCES
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006;56:106–30.
Kelsey JL, Gammon MD, John EM. Reproductive factors and breast cancer. Epidemiol Rev. 1993;15:36–47.
Hulka BS, Stark AT. Breast cancer: cause and prevention. Lancet. 1995;346:883–7.
Kelsey JL, Bernstein L. Epidemiology and prevention of breast cancer. Annu Rev Pub Health. 1996;17:47–67.
Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the national surgical adjuvant breast and bowel project P-1 study. J Natl Cancer Inst. 1998;90:1371–88.
Cuzick J, Forbes J, Edwards R, et al. First results from the International Breast Cancer Intervention study (IBIS-I): a randomized prevention trial. Lancet. 2002;360:817–24.
Newman DJ, Cragg GM, Snader KM. Natural products as sources of new drugs over the period 1981–2002. J Nat Prod. 2003;66:1022–37.
Yun TK, Meyer CA. Brief introduction of Panax ginseng. J Korean Med Sci. 2001;16(suppl):S3.
Abe H, Arichi S, Hayashi T, Odashima S. Ultrastructural studies of Morris hepatoma cells reversely transformed by ginsenosides. Experientia. 1979;35:1647–8.
Odashima S, Ohta T, Kohno H, et al. Control of phenotypic expression of cultured B16 melanoma cells by plant glycosides. Cancer Res. 1985;45:2781–4.
Lee KY, Park JA, Chung E, Lee YH, Kim SI, Lee SK. Ginsenoside-Rh2 blocks the cell cycle of SK-HEP-1 cells at the G1/S boundary by selectively inducing the protein expression of p27kip1. Cancer Lett. 1996;110:193–200.
Oh M, Choi YH, Choi SH, et al. Anti-proliferating effects of ginsenoside Rh2 on MCF-7 human breast cancer cells. Int J Oncol. 1999;14:869–75.
Kim YS, Jin SH. Ginsenoside Rh2 induces apoptosis via activation of caspase-1 and -3 and up-regulation of Bax in human neuroblastoma. Arch Pharm Res. 2004;27:834–9.
Kim SH, Lee EH, Ko SR, Choi KJ, Park JH, Im DS. Effects of ginsenosides Rg3 and Rh2 on the proliferation of prostate cancer cells. Arch Pharm Res. 2004;27:429–35.
Cheng CC, Yang SM, Huang CY, Chen JC, Chang WM, Hsu SL. Molecular mechanisms of ginsenoside Rh2-mediated G1 growth arrest and apoptosis in human lung adenocarcinoma A549 cells. Cancer Chemother Pharmacol. 2005;55:531–40.
Kitts DD, Popovich DG, Hu C. Characterizing the mechanism for ginsenoside-induced cytotoxicity in cultured leukemia (THP-1) cells. Can J Physiol Pharmacol. 2007;85:1173–83.
Huang J, Tang XH, Ikejima T, et al. A new triterpenoid from Panax ginseng exhibits cytotoxicity through p53 and the caspase signaling pathway in the HepG2 cell line. Arch Pharm Res. 2008;31:323–9.
Kim SY, Kim DH, Han SJ, Hyun JW, Kim HS. Repression of matrix metalloproteinase gene expression by ginsenoside Rh2 in human astroglioma cells. Biochem Pharmacol. 2007;74:1642–51.
Xie X, Eberding A, Madera C, et al. Rh2 synergistically enhances paclitaxel or mitoxantrone in prostate cancer models. J Urol. 2006;175:1926–31.
Wang Z, Zheng Q, Liu K, Li G, Zheng R. Ginsenoside Rh2 enhances antitumor activity and decreases genotoxic effect of cyclophosphamide. Basic Clin Pharmacol Toxicol. 2006;98:411–5.
Xiao D, Choi S, Johnson DE, et al. Diallyl trisulfide-induced apoptosis in human prostate cancer cells involves c-Jun N-terminal kinase and extracellular-signal regulated kinase-mediated phosphorylation of Bcl-2. Oncogene. 2004;23:5594–606.
Herman-Antosiewicz A, Singh SV. Checkpoint kinase 1 regulates diallyl trisulfide-induced mitotic arrest in human prostate cancer cells. J Biol Chem. 2005;280:28519–28.
Xiao D, Srivastava SK, Lew KL, et al. Allyl isothiocyanate, a constituent of cruciferous vegetables, inhibits proliferation of human prostate cancer cells by causing G2/M arrest and inducing apoptosis. Carcinogenesis. 2003;24:891–7.
Deep G, Singh RP, Agarwal C, Kroll DJ, Agarwal R. Silymarin and silibinin cause G1 and G2-M cell cycle arrest via distinct circuitries in human prostate cancer PC3 cells: a comparison of flavanone silibinin with flavanolignan mixture silymarin. Oncogene. 2006;25:1053–69.
Hahm ER, Singh SV. Honokiol causes G0–G1 phase cell cycle arrest in human prostate cancer cells in association with suppression of retinoblastoma protein level/phosphorylation and inhibition of E2F1 transcriptional activity. Mol Cancer Ther. 2007;6:2686–95.
Kim WY, Sharpless NE. The regulation of INK4/ARF in cancer and aging. Cell. 2006;127:265–75.
Brown L, Boswell S, Raj L, Lee SW. Transcriptional targets of p53 that regulate cellular proliferation. Crit Rev Eukaryot Gene Expr. 2007;17:73–85.
Molinari M. Cell cycle checkpoints and their inactivation in human cancer. Cell Prolif. 2000;33:261–74.
Schafer KA. The cell cycle: a review. Vet Pathol. 1998;35:461–78.
Taylor WR, Stark GR. Regulation of the G2/M transition by p53. Oncogene. 2001;20:1803–15.
Harper JW, Elledge SJ, Keyomarsi K, et al. Inhibition of cyclin-dependent kinases by p21. Mol Biol Cell. 1995;6:387–400.
Kato J, Matsushime H, Hiebert SW, Ewen ME, Sherr CJ. Direct binding of cyclin-D to the retinoblastoma gene product (pRB) and pRb phosphorylation by the cyclin D-dependent kinase cdk4. Genes Dev. 1993;7:331–42.
Arroyo M, Raychaudhuri P. Retinoblastoma-repression of E2F-dependent transcription depends on the ability of the retinoblastoma protein to interact with E2F and is abrogated by the adenovirus E1A oncoprotein. Nucleic Acids Res. 1992;20:5947–54.
Weinberg RA. The retinoblastoma protein and cell cycle control. Cell. 1995;81:323–30.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Grant Support: This investigation was supported in part by the Kangwon Bio-Nuri grant, Korea Research Foundation, Korea, and US PHS grant CA129347, awarded by the National Cancer Institute.
Requests for reprints: S. Choi, Ph.D., Dept. of Life Science, Hallym University, 39 Hallymdaehak-gil, Chuncheon, Gangwon-do, 200-702, Korea. Phone: 82-33-248-2097; E-mail: sachoi@hallym.ac.kr or Shivendra V. Singh, 2.32A Hillman Cancer Center Research Pavilion, 5117 Centre Avenue, Pittsburgh, PA 15213. Phone: 412-623-3263; Fax: 412-623-7828. Email: singhs@upmc.edu
Rights and permissions
About this article
Cite this article
Choi, S., Kim, T. & Singh, S.V. Ginsenoside Rh2-mediated G1 Phase Cell Cycle Arrest in Human Breast Cancer Cells Is Caused by p15Ink4B and p27Kip1-dependent Inhibition of Cyclin-dependent Kinases. Pharm Res 26, 2280–2288 (2009). https://doi.org/10.1007/s11095-009-9944-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-009-9944-9