Abstract
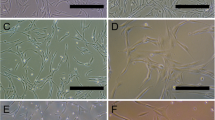
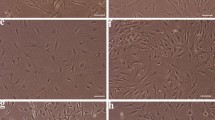
The aim of this study was to compare the neural differentiation potential and the expression of neurotrophic factors (NTFs) in differentiated adipose-derived stem cells (ADSCs) using three established induction protocols, serum free (Protocol 1), chemical reagents (Protocol 2), and spontaneous (Protocol 3) protocols. Protocol 1 produced the highest percentage of mature neural-like cells (MAP2ab+). Protocol 2 showed the highest percentage of immature neural-like cells (β-tubulin III+), but the neural-like state was transient and reversible. Protocol 3 caused ADSCs to differentiate spontaneously into immature neural-like cells, but not into mature neural cell types. The neural-like cells produced by Protocol 1 lived the longest in culture with little cell death, but Protocol 2 and 3 led to the significant cell death. Therefore, Protocol 1 is the most efficient among these protocols. Additionally, soon after differentiation, the mRNA levels of nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) in dADSCs were sharply decreased by Protocol 1 and 2 (acute induction protocol), but not by Protocol 3 (chronic induction protocol). The results indicate that NTFs played an important role in neural differentiation via acute responses to NGF and BDNF, but not chronically during the transdifferentiation process.



Similar content being viewed by others
References
Kingham PJ, Kalbermatten DF, Mahay D, Armstrong SJ, Wiberg M, Terenghi G (2007) Adipose-derived stem cells differentiate into a Schwann cell phenotype and promote neurite outgrowth in vitro. Exp Neurol 207(2):267–274
Gimble JM, Guilak F (2003) Differentiation potential of adipose derived adult stem (ADAS) cells. Curr Top Dev Biol 58:137–160
Strem BM, Hicok KC, Zhu M, Wulur I, Alfonso Z, Schreiber RE et al (2005) Multipotential differentiation of adipose tissue-derived stem cells. Keio J Med 54(3):132–141
Ning H, Lin G, Lue TF, Lin CS (2006) Neuron-like differentiation of adipose tissue-derived stromal cells and vascular smooth muscle cells. Differentiation 74(9–10):510–518
Huang T, He D, Kleiner G, Kuluz J (2007) Neuron-like differentiation of adipose-derived stem cells from infant piglets in vitro. J Spinal Cord Med 30(Suppl 1):S35–S40
Kang SK, Putnam LA, Ylostalo J, Popescu IR, Dufour J, Belousov A et al (2004) Neurogenesis of Rhesus adipose stromal cells. J Cell Sci 117(Pt 18):4289–4299
Woodbury D, Schwarz EJ, Prockop DJ, Black IB (2000) Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res 61(4):364–370
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR et al (2002) Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418(6893):41–49
Jiang Y, Henderson D, Blackstad M, Chen A, Miller RF, Verfaillie CM (2003) Neuroectodermal differentiation from mouse multipotent adult progenitor cells. Proc Natl Acad Sci USA 100(Suppl 1):11854–11860
Hermann A, Gastl R, Liebau S, Popa MO, Fiedler J, Boehm BO et al (2004) Efficient generation of neural stem cell-like cells from adult human bone marrow stromal cells. J Cell Sci 117(Pt 19):4411–4422
Wislet-Gendebien S, Leprince P, Moonen G, Rogister B (2003) Regulation of neural markers nestin and GFAP expression by cultivated bone marrow stromal cells. J Cell Sci 116(Pt 16):3295–3302
Wislet-Gendebien S, Hans G, Leprince P, Rigo JM, Moonen G, Rogister B (2005) Plasticity of cultured mesenchymal stem cells: switch from nestin-positive to excitable neuron-like phenotype. Stem Cells 23(3):392–402
Sanchez-Ramos J, Song S, Cardozo-Pelaez F, Hazzi C, Stedeford T, Willing A et al (2000) Adult bone marrow stromal cells differentiate into neural cells in vitro. Exp Neurol 164(2):247–256
Kohyama J, Abe H, Shimazaki T, Koizumi A, Nakashima K, Gojo S et al (2001) Brain from bone: efficient “meta-differentiation” of marrow stroma-derived mature osteoblasts to neurons with Noggin or a demethylating agent. Differentiation 68(4–5):235–244
Tseng PY, Chen CJ, Sheu CC, Yu CW, Huang YS (2007) Spontaneous differentiation of adult rat marrow stromal cells in a long-term culture. J Vet Med Sci 69(2):95–102
Wagers AJ, Christensen JL, Weissman IL (2002) Cell fate determination from stem cells. Gene Ther 9(10):606–612
Alvarez-Dolado M, Pardal R, Garcia-Verdugo JM, Fike JR, Lee HO, Pfeffer K et al (2003) Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425(6961):968–973
Weimann JM, Charlton CA, Brazelton TR, Hackman RC, Blau HM (2003) Contribution of transplanted bone marrow cells to Purkinje neurons in human adult brains. Proc Natl Acad Sci USA 100(4):2088–2093
Castro RF, Jackson KA, Goodell MA, Robertson CS, Liu H, Shine HD (2002) Failure of bone marrow cells to transdifferentiate into neural cells in vivo. Science 297(5585):1299
Ono K, Yoshihara K, Suzuki H, Tanaka KF, Takii T, Onozaki K et al (2003) Preservation of hematopoietic properties in transplanted bone marrow cells in the brain. J Neurosci Res 72(4):503–507
Li N, Yang H, Lu L, Duan C, Zhao C, Zhao H (2007) Spontaneous expression of neural phenotype and NGF, TrkA, TrkB genes in marrow stromal cells. Biochem Biophys Res Commun 356(3):561–568
Yaghoobi MM, Mahani MT (2008) NGF and BDNF expression drop off in neurally differentiated bone marrow stromal stem cells. Brain Res 1203:26–31
Levi-Montalcini R, Skaper SD, Dal Toso R, Petrelli L, Leon A (1996) Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci 19(11):514–520
Barbacid M (1995) Neurotrophic factors and their receptors. Curr Opin Cell Biol 7(2):148–155
Zhang HT, Luo J, Sui LS, Ma X, Yan ZJ, Lin JH et al (2009) Effects of differentiated versus undifferentiated adipose tissue-derived stromal cell grafts on functional recovery after spinal cord contusion. Cell Mol Neurobiol
Zhang HT, Cheng HY, Zhang L, Fan J, Chen YZ, Jiang XD et al (2009) Umbilical cord blood cell-derived neurospheres differentiate into Schwann-like cells. Neuroreport 20(4):354–359
Bunnell BA, Ylostalo J, Kang SK (2006) Common transcriptional gene profile in neurospheres-derived from pATSCs, pBMSCs, and pNSCs. Biochem Biophys Res Commun 343(3):762–771
Lu P, Blesch A, Tuszynski MH (2004) Induction of bone marrow stromal cells to neurons: differentiation, transdifferentiation, or artifact? J Neurosci Res 77(2):174–191
Neuhuber B, Gallo G, Howard L, Kostura L, Mackay A, Fischer I (2004) Reevaluation of in vitro differentiation protocols for bone marrow stromal cells: disruption of actin cytoskeleton induces rapid morphological changes and mimics neuronal phenotype. J Neurosci Res 77(2):192–204
Croft AP, Przyborski SA (2006) Formation of neurons by non-neural adult stem cells: potential mechanism implicates an artifact of growth in culture. Stem Cells 24(8):1841–1851
Chen Q, Long Y, Yuan X, Zou L, Sun J, Chen S et al (2005) Protective effects of bone marrow stromal cell transplantation in injured rodent brain: synthesis of neurotrophic factors. J Neurosci Res 80(5):611–619
Lu P, Tuszynski MH (2005) Can bone marrow-derived stem cells differentiate into functional neurons? Exp Neurol 193(2):273–278
Yuen EC, Mobley WC (1996) Therapeutic potential of neurotrophic factors for neurological disorders. Ann Neurol 40(3):346–354
Crigler L, Robey RC, Asawachaicharn A, Gaupp D, Phinney DG (2006) Human mesenchymal stem cell subpopulations express a variety of neuro-regulatory molecules and promote neuronal cell survival and neuritogenesis. Exp Neurol 198(1):54–64
Ye M, Chen S, Wang X, Qi C, Lu G, Liang L et al (2005) Glial cell line-derived neurotrophic factor in bone marrow stromal cells of rat. Neuroreport 16(6):581–584
Yang HJ, Yang XY, Ba YC, Pang JX, Meng BL, Lin N et al (2009) Role of Neurotrophin 3 in spinal neuroplasticity in rats subjected to cord transection. Growth Factors 27(4):237–246
Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm-Grove CJ, Bovenkerk JE et al (2004) Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 109:1292–1298
Qu R, Li Y, Gao Q, Shen L, Zhang J, Liu Z et al (2007) Neurotrophic and growth factor gene expression profiling of mouse bone marrow stromal cells induced by ischemic brain extracts. Neuropathology 27:355–363
Acknowledgments
This work was supported by grants from the Natural Science Foundation of China (No. U0632008), Foundation for Key Sci-Tech Research Projects of Guangdong (No. 2008A030201019, 2007-05/06-7005206) and Guangzhou (No. 09B52120112, 2008A1-E4011-6), Foundation for Medical and Scientific Technology Research of Guangdong (No. A2009293).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Hong-Tian Zhang contributed equally with Dong-Xiang Qian.
Rights and permissions
About this article
Cite this article
Qian, DX., Zhang, HT., Ma, X. et al. Comparison of the Efficiencies of Three Neural Induction Protocols in Human Adipose Stromal Cells. Neurochem Res 35, 572–579 (2010). https://doi.org/10.1007/s11064-009-0101-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-009-0101-y