Abstract
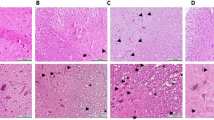
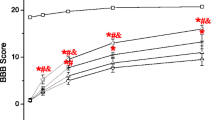
Spinal cord injury (SCI) caused by trauma mainly occurs in two mechanisms as primary and secondary injury. Secondary injury following the primary impact includes various pathophysiological and biochemical events. Methylprednisolone is the only pharmacological agent having clinically proven beneficial effects on SCI. Citicoline has been shown to have clinical and experimental beneficial effects on brain ischemia. This study aims to investigate the neuroprotective effect of citicoline in an experimental SCI model in rats. Sixty adult Wistar albino rats were randomized into five groups. SCI was performed by the weight-drop model. Group 1 underwent laminectomy alone. The Group 2 underwent laminectomy followed by SCI and received no medication. Group3, Group 4 and Group 5 underwent laminectomy followed by SCI and received medication. Group 3 and Group 5 received citicoline and Group 4 and Group 5 received methylprednisolone. The rats were divided into two subgroups for biochemical analysis (sacrificed at 24 h after surgery) and neurobehavioral and histopathological evaluation (sacrificed at 6 weeks after surgery). Malonildialdehyde levels, nitric oxide levels and trauma size ratios were lower and reduced glutathione levels were higher in Group 3, Group 4 and Group 5 as compared to Group 2. Posttraumatic neurological recovery after surgery was significantly better in Group 3, Group 4 and Group 5 compared to Group 2. In conclusion, this study demonstrates that citicoline is as effective as methylprednisolone. The efficacy of citicoline combined with methylprednisolone is not superior to either citicoline or methylprednisolone alone.







Similar content being viewed by others
References
Tator CH, Fehlings MG (1991) Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 75:15–26
Blight AR, Young W (1989) Central axons in injured cat spinal cord recover electrophysiological function following remyelination by Schwann cells. J Neurol Sci 91:15–34
Basso DM, Beattie MS, Bresnahan JC (1996) Graded histological and locomotor outcomes after spinal cord contusion using the NYU weight-drop device versus transection. Exp Neurol 139:244–256
Bracken MB, Shepard MJ, Collins WF, Holford TR, Young W, Baskin DS (1990) Eisenberg HM, Flamm E, Leo-Summers L, Maroon J, et al, A randomized, controlled trial of MP or naloxone in the treatment of acute spinal-cord injury, Result of the Second National Acute Spinal Cord Injury Study. N Engl J Med 322:1405–1411
Braughler JM, Hall ED (1982) Correlation of methylprednisolone levels in cat spinal cord with its effects on (Na+K)-ATP-ase, lipid peroxidation, and alpha motor neuron function. J Neurosurg 56:838–844
Young W, Flamm ES (1982) Effects of high-dose corticosteroid therapy on blood flow, evoked potentials and extracellular calcium in experimental spinal cord injury. J Neurosurg 57:667–673
Schabitz WR, Li F, Irie K, Sandage BW, Locke KW, Fisher M (1999) Synergistic effects of a combination of low dose basic fibroblast growth factor and citicoline after temporary experimental focal ischemia. Stroke 30:427–432
Adibhatla RM, Hatcher JF (2002) Citicoline mechanisms and clinical efficacy in cerebral ischemia. Journal of Neurosci Res 70:133–139
Secades JJ, Frontera G (1995) CDP-choline: pharmacological and clinical review. Meth Find Exp Clin Pharmacol 17:2–54
Baskaya MK, Dogan A, Rao AM, Dempsey RJ (2000) Neuroprotective effects of citicoline on brain edema and blood-brain barrier breakdown after traumatic brain injury. J Neurosurg 92:448–452
Shuaib A, Yang Y, Li Q (2000) Evaluating the efficacy of citicoline in embolic ischemic stroke in rats: neuroprotective effects when used alone or in combination with urokinase. Exp Neurol 161:733–739
Allen AR (1911) Surgery of experimental lesion of spinal cord equivalent to crush injury of fracture dislocation of spinal column. A preliminary report. JAMA 57:878–880
Mihara S, Uchiyama M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Elman GL (1959) Tissue sulphydryl groups. Arch Biochem Biophys 82:70–77
Ozbek E, Turkoz Y, Gokdeniz R, Davarcı M, Ozugurlu F (2000) Increased nitric oxide production in the spermatic vein of patients with varicocele. Eur Urol 37:172–175
Farooque M, Isaksson J, Jackson D, Olsson Y (1999) Clomethiazole improves hind limb function and reduces neuronal damage after severe spinal cord injury in rat. Acta Neuropathol 98:22–30
Gale K, Kerasidis H, Wrathall JR (1985) Spinal cord contusions in the rat: behavioral analysis of functional neurologic impairment. Exp Neurol 88:123–134
Rivlin AS, Tator CH (1977) Objective clinical assessment of motor function after experimental spinal cord injury in the rat. J Neurosurg 47:577–581
Huang SS, Tsai MC, Chih CL, Hung LM, Tsai SK (2001) Resveratrol reduction of infarct size in Long-Evans rats subjected to focal cerebral ischemia. Life Sci 69:1057–1065
Hall ED (1992) The neuroprotective pharmacology of methylprednisolone. J Neurosurg 76:13–22
Hall ED (1993) Lipid peroxidants in acute central nervous system injury. Ann Emerg Med 22:1022–1027
Anderson DK, Hall ED (1993) Pathophysiology of spinal cord trauma. Ann Emerg Med 22:987–992
Fercakova A, Halat G, Marsala M, Lukacova N, Marsala J (1992) Graded postischemic reoxigenation reduces lipid peroxidation and reperfusion injury in the rabbit spinal cord. Brain Res 593:159–167
Rao AM, Hatcher JF, Dempsey RJ (2000) Lipid alterations in transient forebrain ischemia: possible new mechanisms of CDP-choline neuroprotection. J Neurochem 75:2528–2535
Rao AM, Hatcher JF, Dempsey RJ (1999) CDP-choline; neuroprotection in transient forebrain ischemia of gerbils. J Neurosci Res 58:697–705
Constantini S, Young W (1994) The effects of methylprednisolone and the ganglioside GM1 on acute spinal cord injury in rats. J Neurosurg 80:97–111
Braughler JM, Hall ED (1985) Current application of “high dose” steroid therapy for CNS injury. A pharmacological perspective. J Neurosurg 62:806–810
Hall ED, Braughler JM (1982) Effects of intravenous methylprednisolone on spinal cord lipid peroxidation and (Na+-K+ ) ATPase activity. Dose-response analysis during the 1st hour after contusion injury in the cat. J Neurosurg 57:247–253
Young W (1992) Medical treatments of acute spinal cord injury. J Neurol Neurosurg Psychiatry 55:635–639
Amar AP, Levy ML (1999) Pathogenesis and pharmacological strategies for mitigating secondary damage in acute spinal cord injury. Neurosurgery 44:1027–1039
Demediuk P, Saunders RD, Clendenon NR, Means ED, Anderson DK, Horrocks LA (1985) Changes in lipid metabolism in traumatized spinal cord. Prog Brain Res 63:1–16
Rao AM, Hatcher JF, Dempsey RJ (2001) Does CDP-choline modulate phospholipase activities after transient forebrain ischemia? Brain Res 893:268–272
Adibhatla RM, Hatcher JF, Dempsey RJ (2003) Phospholipase A2, hydroxyl radicals and lipid peroxidation in transient cerebral ischemia. Antioxid Redox Signal 5:647–654
Adibhatla RM, Hatcher JF, Dempsey RJ (2001) Effects of citicoline on phospholilipid and glutathione levels in transient cerebral ischemia. Stroke 32:2376–2381
Barut S, Canbolat A, Bilge T, Aydin Y, Cokneseli B, Kaya U (1993) Lipid peroxidation in experimental spinal cord injury: time–level relationship. Neurosurg Rev 16:53–59
Cakir E, Ulusul H, Peksoylu B, Sayin OC, Alver A, Topbas M, Baykal S, Kuzeyli K (2005) Effects of citicoline on experimental spinal cord injury. J Clin Neurosci 12:923–926
Schabitz WR, Weber J, Tokano K, Sandage BW, Locke KW, Fisher M (1996) The effects of prolonged treatment with citicoline in temporary experimental focal ischemia. J Neurol Sci 138:21–25
Moncada S, Higgs A (1993) The L-arginine-nitric oxide pathway. N Engl J Med 329:2002–2012
Peunova N, Enikolopov G (1993) Amplification of calcium-induced gene transcription by nitric oxide in neuronal cells. Nature 364:450–453
Ignarro LJ, Buga GM, Wood KS, Byrns RE (1987) Chaudhuri G, Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc Natl Acad Sci USA 84:9265–9269
Goldberger ME, Bregman BS, Vierck CJ Jr, Brown M (1990) Criteria for assesing recovery of function after spinal cord injury: behavioral methods. Exp Neurol 107:113–117
Von Euler M, Akesson E, Samuelsson EB, Seiger A, Sundström E (1996) Motor performance score: a new algorithm for accurate behavioral testing of spinal cord injury in rats. Exp Neurol 137:242–245
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yücel, N., Çaylı, S.R., Ateş, Ö. et al. Evaluation of the Neuroprotective Effects of Citicoline after Experimental Spinal Cord Injury: Improved Behavioral and Neuroanatomical Recovery. Neurochem Res 31, 767–775 (2006). https://doi.org/10.1007/s11064-006-9075-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-006-9075-1