Abstract
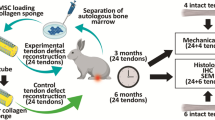
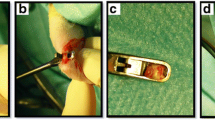
In order to investigate cell-based tendon regeneration, a tendon rupture was simulated by utilizing a critical full-size model in female rat achilles tendons. For bridging the defect, polyglycol acid (PGA) and collagen type I scaffolds were used and fixed with a frame suture to ensure postoperatively a functional continuity. Scaffolds were seeded with mesenchymal stem cells (MSC) or tenocytes derived from male animals, while control groups were left without cells. After a healing period of 16 weeks, biomechanical, PCR, histologic, and electron microscopic analyses of the regenerates were performed. Genomic PCR for male-specific gene was used to detect transplanted cells in the regenerates. After 16 weeks, central ossification and tendon-like tissue in the superficial tendon layers were observed in all study groups. Biomechanical test showed that samples loaded with tenocytes had significantly better failure strength/cross-section ratio (P < 0.01) compared to MSC and the control groups whereas maximum failure strength was similar in all groups. Thus, we concluded that the application of tenocytes improves the outcome in this model concerning the grade of ossification and the mechanical properties in comparison to the use of MSC or just scaffold materials.






Similar content being viewed by others
References
Lichtenberg S, Liem D, Magosch P, Habermeyer P. Influence of tendon healing after arthroscopic rotator cuff repair on clinical outcome using single-row Mason–Allen suture technique: a prospective, MRI controlled study. Knee Surg Sports Traumatol Arthrosc. 2006;14:1200–6.
Anders JO, Venbrocks RA, Weinberg M. Proprioceptive skills and functional outcome after anterior cruciate ligament reconstruction with a bone–tendon–bone graft. Int Orthop. 2007;32:627–33.
Costa ML, MacMillan K, Halliday D, Chester R, Shepstone L, Robinson AH, Donell ST. Randomized controlled trials of immediate weight-bearing mobilization for rupture of the tendo Achillis. J Bone Joint Surg Br. 2006;88:69–77.
Favard L, Bacle G, Berhouet J. Rotator cuff repair. Joint Bone Spine. 2007;74:551–7.
Kasten P, Loew M. How to treat massive rotator cuff tears. Orthopade. 2007;36:855–61.
Moser M, Jablonski MV, Horodyski M, Wright TW. Functional outcome of surgically treated massive rotator cuff tears: a comparison of complete repair, partial repair, and debridement. Orthopedics. 2007;30:479–82.
Boes MT, McCann PD, Dines DM. Diagnosis and management of massive rotator cuff tears: the surgeon’s dilemma. Instr Course Lect. 2006;55:45–57.
Boehm TD, Ilg A, Werner A, Kirschner S, Gohlke F. Long-term results (5–13 years) after surgical local repair and deltoid muscle flap for massive rotator cuff tears. Z Orthop Ihre Grenzgeb. 2004;142:228–34.
Ho JY, Miller SL. Allografts in the treatment of athletic injuries of the shoulder. Sports Med Arthrosc. 2007;15:149–57.
Kryger GS, Chong AK, Costa M, Pham H, Bates SJ, Chang J. A comparison of tenocytes and mesenchymal stem cells for use in flexor tendon tissue engineering. J Hand Surg Am. 2007;32:597–605.
MacGillivray JD, Fealy S, Terry MA, Koh JL, Nixon AJ, Warren RF. Biomechanical evaluation of a rotator cuff defect model augmented with a bioresorbable scaffold in goats. J Shoulder Elbow Surg. 2006;15:639–44.
Suckow MA, Hodde JP, Wolter WR, Hiles MC. Repair of experimental achilles tenotomy with porcine renal capsule material in a rat model. J Mater Sci Mater Med. 2007;18:1105–10.
Zalavras CG, Gardocki R, Huang E, Stevanovic M, Hedman T, Tibone J. Reconstruction of large rotator cuff tendon defects with porcine small intestinal submucosa in an animal model. J Shoulder Elbow Surg. 2006;15:224–31.
Nicholson GP, Breur GJ, Van Sickle D, Yao JQ, Kim J, Blanchard CR. Evaluation of a cross-linked acellular porcine dermal patch for rotator cuff repair augmentation in an ovine model. J Shoulder Elbow Surg. 2007;16:S184–90.
Chen JM, Willers C, Xu J, Wang A, Zheng MH. Autologous tenocyte therapy using porcine-derived bioscaffolds for massive rotator cuff defect in rabbits. Tissue Eng. 2007;13:1479–91.
Walton JR, Bowman NK, Khatib Y, Linklater J, Murrell GA. Restore orthobiologic implant: not recommended for augmentation of rotator cuff repairs. J Bone Joint Surg Am. 2007;89:786–91.
Iannotti JP, Codsi MJ, Kwon YW, Derwin K, Ciccone J, Brems JJ. Porcine small intestine submucosa augmentation of surgical repair of chronic two-tendon rotator cuff tears. A randomized, controlled trial. J Bone Joint Surg Am. 2006;88:1238–44.
Moore DR, Cain EL, Schwartz ML, Clancy WG Jr. Allograft reconstruction for massive, irreparable rotator cuff tears. Am J Sports Med. 2006;34:392–6.
Porada CD, Zanjani ED, Almeida-Porad G. Adult mesenchymal stem cells: a pluripotent population with multiple applications. Curr Stem Cell Res Ther. 2006;1:365–9.
Docheva D, Popov C, Mutschler W, Schieker M. Human mesenchymal stem cells in contact with their environment: surface characteristics and the integrin system. J Cell Mol Med. 2007;11:21–38.
Tilley JM, Chaudhury S, Hakimi O, Carr AJ, Czernuszka JT. Tenocyte proliferation on collagen scaffolds protects against degradation and improves scaffold properties. J Mater Sci Mater Med. 2011;23:823–33.
Cao Y, Liu Y, Liu W, Shan Q, Buonocore SD, Cui L. Bridging tendon defects using autologous tenocyte engineered tendon in a hen model. Plast Reconstr Surg. 2002;110:1280–9.
Muller PE, Schrimpf F, Milz S, Kircher J, Durr HR, Wegener B, Pellengahr C, Jansson V. Repair of osteochondral defects in the knee by resorbable bioimplants in a rabbit model. Acta Orthop. 2006;77:981–5.
Jansson V, Muller PE, Thal S, Arnholz C, Milz S, Koch KU, Refior HJ. A new resorbable bone-cartilage replacement transplant. Results of an animal experiment study. Orthopade. 2000;29:151–7.
Benjamin M, Hillen B. Mechanical influences on cells, tissues and organs — ‘Mechanical Morphogenesis’. Eur J Morphol. 2003;41:3–7.
Pauwels F. A new theory on the influence of mechanical stimuli on the differentiation of supporting tissue. The tenth contribution to the functional anatomy and causal morphology of the supporting structure. Z Anat Entwicklungsgesch. 1960;121:478–515.
Awad HA, Boivin GP, Dressler MR, Smith FN, Young RG, Butler DL. Repair of patellar tendon injuries using a cell–collagen composite. J Orthop Res. 2003;21:420–31.
Butler DL, Gooch C, Kinneberg KR, Boivin GP, Galloway MT, Nirmalanandhan VS, Shearn JT, Dyment NA, Juncosa-Melvin N. The use of mesenchymal stem cells in collagen-based scaffolds for tissue-engineered repair of tendons. Nat Protoc. 2010;5:849–63.
Chong AK, Ang AD, Goh JC, Hui JH, Lim AY, Lee EH, Lim BH. Bone marrow-derived mesenchymal stem cells influence early tendon-healing in a rabbit achilles tendon model. J Bone Joint Surg Am. 2007;89:74–81.
Okamoto N, Kushida T, Oe K, Umeda M, Ikehara S, Iida H. Treating achilles tendon rupture in rats with bone-marrow-cell transplantation therapy. J Bone Joint Surg Am. 2010;92:2776–84.
Drosse I, Volkmer E, Seitz S, Seitz H, Penzkofer R, Zahn K, Matis U, Mutschler W, Augat P, Schieker M. Validation of a femoral critical size defect model for orthotopic evaluation of bone healing: a biomechanical, veterinary and trauma surgical perspective. Tissue Eng Part C Methods. 2008;14:79–88.
Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ, Krause DS, Keating A. Clarification of the nomenclature for MSC: the International Society for Cellular Therapy position statement. Cytotherapy. 2005;7:393–5.
Colter DC, Class R, DiGirolamo CM, Prockop DJ. Rapid expansion of recycling stem cells in cultures of plastic-adherent cells from human bone marrow. Proc Natl Acad Sci USA. 2000;97:3213–8.
Bocker W, Rossmann O, Docheva D, Malterer G, Mutschler W, Schieker M. Quantitative polymerase chain reaction as a reliable method to determine functional lentiviral titer after ex vivo gene transfer in human mesenchymal stem cells. J Gene Med. 2007;9:585–95.
Slack C, Bradley G, Beaumont B, Poole A, Flint M. Changes in the morphology and synthetic activity of cultured rat tail tendon. Cell Tissue Res. 1986;245:359–68.
Wegener B, Schrimpf FM, Bergschmidt P, Pietschmann MF, Utzschneider S, Milz S, Jansson V, Muller PE. Cartilage regeneration by bone marrow cells-seeded scaffolds. J Biomed Mater Res A. 2010;95:735–40.
Wegener B, Schrimpf FM, Pietschmann MF, Milz S, Berger-Lohr M, Bergschmidt P, Jansson V, Muller PE. Matrix-guided cartilage regeneration in chondral defects. Biotechnol Appl Biochem. 2009;53:63–70.
Shakibaei M, Zimmermann B, Merker HJ. Changes in integrin expression during chondrogenesis in vitro: an immunomorphological study. J Histochem Cytochem. 1995;43:1061–9.
Schulze-Tanzil G, Mobasheri A, Clegg PD, Sendzik J, John T, Shakibaei M. Cultivation of human tenocytes in high-density culture. Histochem Cell Biol. 2004;122:219–28.
Kuddus RH, Lee YH, Valdivia LA. A semiquantitative PCR technique for detecting chimerism in hamster-to-rat bone marrow xenotransplantation. J Immunol Methods. 2004;285:245–51.
Forslund C, Aspenberg P. CDMP-2 induces bone or tendon-like tissue depending on mechanical stimulation. J Orthop Res. 2002;20:1170–4.
Harris MT, Butler DL, Boivin GP, Florer JB, Schantz EJ, Wenstrup RJ. Mesenchymal stem cells used for rabbit tendon repair can form ectopic bone and express alkaline phosphatase activity in constructs. J Orthop Res. 2004;22:998–1003.
Dressler MR, Butler DL, Boivin GP. Effects of age on the repair ability of mesenchymal stem cells in rabbit tendon. J Orthop Res. 2005;23:287–93.
Henderson JH, Carter DR. Mechanical induction in limb morphogenesis: the role of growth-generated strains and pressures. Bone. 2002;31:645–53.
Liu L, Yuan W, Wang J. Mechanisms for osteogenic differentiation of human mesenchymal stem cells induced by fluid shear stress. Biomech Model Mechanobiol. 2010;9:659–70.
Potier E, Ferreira E, Andriamanalijaona R, Pujol JP, Oudina K, Logeart-Avramoglou D, Petite H. Hypoxia affects mesenchymal stromal cell osteogenic differentiation and angiogenic factor expression. Bone. 2007;40:1078–87.
Cao Y, Rodriguez A, Vacanti M, Ibarra C, Arevalo C, Vacanti CA. Comparative study of the use of poly(glycolic acid), calcium alginate and pluronics in the engineering of autologous porcine cartilage. J Biomater Sci Polym Ed. 1998;9:475–87.
Heino A, Naukkarinen A, Pietilainen T, Merasto ME, Tormala P, Pohjonen T, Makela EA. Application of a self-reinforced polyglycolic acid (SR-PGA) membrane to the closure of an abdominal fascial defect in rats. J Biomed Mater Res. 1999;48:596–601.
Pihlajamaki H, Tynninen O, Karjalainen P, Rokkanen P. Enveloping bioabsorbable polyglycolide membrane and immobilization in achilles tendon repair: a comparative experimental study on rabbits. J Orthop Res. 2008;26:264–70.
Acknowledgments
We thank Prof. Ulrich Welsch for support in electron microscopy. The histologic consultation was kindly performed by the anatomist Prof. Mehdi Shakibaei. The Medical School Research Fund of the Ludwig Maximilians University in Munich (grant: 419-2005) supported this study. Dr. Denitsa Docheva and Prof. Matthias Schieker acknowledge the support of the AO Research Fund of the AO Foundation (grants S-07-18D and S-10-74D). We also thank Martina Burggraf and Krystyna Ern for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors M.F. Pietschmann and B. Frankewycz contributed evenly to this study.
Rights and permissions
About this article
Cite this article
Pietschmann, M.F., Frankewycz, B., Schmitz, P. et al. Comparison of tenocytes and mesenchymal stem cells seeded on biodegradable scaffolds in a full-size tendon defect model. J Mater Sci: Mater Med 24, 211–220 (2013). https://doi.org/10.1007/s10856-012-4791-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-012-4791-3