Summary
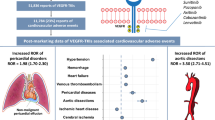
Purpose Renal toxicities are common with angiogenesis multikinase inhibitors (AMKI), and can be limiting in phase I trials. Factors associated with such toxicities are poorly known. The aims of this exploratory study were to describe renovascular toxicities associated with AMKI, impact on drug development and to identify baseline parameters associated with the occurrence of renal toxicities in phase I trials. Methods Consecutive patients treated with AMKI in Gustave Roussy phase I unit between October 2005 and August 2013 were included. We retrospectively collected baseline characteristics and renovascular side effects. Associations were assessed in univariate and multivariate analyses. Results Overall, 168 patients were included: male 53.0 %, mean age 55.5 years old, history of hypertension 26.8 %, diabetes 6.0 %, atherosclerosis 13.6 %, stage 3 Chronic Kidney Disease (CKD, NKF-KDOQI) 17.2 %. Incidences of reno-vascular side effects were: hypertension 47.6 %, proteinuria 19.0 %, renal failure 11.9 % and thrombotic microangiopathy 10.1 %. Eighty percent of dose limiting toxicities (DLTs) were related to a renal toxicity. Multivariate analysis showed that onset of renal failure was associated with history of hypertension (p = 0.0003) and stage 3 CKD (p = 0.032). Conclusions A majority of the DLTs associated with AMKI in phase 1 trials are renal toxicities. Baseline hypertension and stage 3 CKD (NKF-KDOQI) might help to better identify patients at risk of AMKI-related renal toxicities.
Similar content being viewed by others
References
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285(21):1182–1186
Folkman J (2002) Role of angiogenesis in tumor growth and metastasis. Semin Oncol 29(6 Suppl 16):15–18
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol Off J Am Soc Clin Oncol 23(5):1011–1027
Ikeda N et al (1999) Prognostic significance of angiogenesis in human pancreatic cancer. Br J Cancer 79(9–10):1553–1563
Manders P et al (2002) The prognostic value of vascular endothelial growth factor in 574 node-negative breast cancer patients who did not receive adjuvant systemic therapy. Br J Cancer 87(7):772–778
George DJ et al (2001) Prognostic significance of plasma vascular endothelial growth factor levels in patients with hormone-refractory prostate cancer treated on Cancer and Leukemia Group B 9480. Clin Cancer Res Off J Am Assoc Cancer Res 7(7):1932–1936
Fontanini G et al (1997) Angiogenesis as a prognostic indicator of survival in non-small-cell lung carcinoma: a prospective study. J Natl Cancer Inst 89(12):881–886
Gorski DH, Leal AD, Goydos JS (2003) Differential expression of vascular endothelial growth factor-A isoforms at different stages of melanoma progression. J Am Coll Surg 197(3):408–418
Jayson GC, Kerbel R, Ellis LM, Harris AL (2016) Antiangiogenic therapy in oncology: current status and future directions. Lancet Lond. Engl
Bair SM, Choueiri TK, Moslehi J (2013) Cardiovascular complications associated with novel angiogenesis inhibitors: emerging evidence and evolving perspectives. Trends Cardiovasc Med 23(4):104–113
Launay-Vacher V et al (2015) Renal effects of molecular targeted therapies in oncology: a review by the Cancer and the Kidney International Network (C-KIN). Ann Oncol Off J Eur Soc Med Oncol ESMO 26(8):1677–1684
Zhu X, Wu S, Dahut WL, Parikh CR (2007) Risks of proteinuria and hypertension with bevacizumab, an antibody against vascular endothelial growth factor: systematic review and meta-analysis. Am J Kidney Dis Off J Natl Kidney Found 49(2):186–193
Levy A et al (2012) Reappraisal of treatment-induced renal dysfunction in patients receiving antiangiogenic agents in phase I trials. Invest New Drugs 30(3):1116–1120
Inker LA et al (2014) KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD. Am J Kidney Dis 63(5):713–735
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130(6):461–470
Barbour T, Johnson S, Cohney S, Hughes P (2012) Thrombotic microangiopathy and associated renal disorders. Nephrol Dial Transpl Off Publ Eur Dial Transpl Assoc - Eur Ren Assoc 27(7):2673–2685
Schrijvers BF, Flyvbjerg A, De Vriese AS (2004) The role of vascular endothelial growth factor (VEGF) in renal pathophysiology. Kidney Int 65(6):2003–2017
Eremina V et al (2003) Glomerular-specific alterations of VEGF-A expression lead to distinct congenital and acquired renal diseases. J Clin Invest 111(5):707–716
Eremina V et al (2008) VEGF inhibition and renal thrombotic microangiopathy. N Engl J Med 358(11):1129–1136
Halimi J-M et al (2008) Vascular and renal effects of anti-angiogenic therapy. Néphrol Thér 4(7):602–615
Author contributions
BR, EB, OM designed the study.
BR, EB, OM performed the data collection.
BR performed the statistical analysis.
BR, EB, OM, AH wrote the manuscript.
All authors performed data interpretation and final review of the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Emilie Boissier declares that she has no conflict of interest with respect to this manuscript.
Dr. Mir has acted as a consultant for Astra-Zeneca, Amgen, Bayer, BMS, GSK, Roche, Servier, Pfizer, PharmaMar, Novartis.
Antoine Hollebecque declares that he has no conflict of interest with respect to this manuscript.
Hassan Izzedine declares that he has no conflict of interest with respect to this manuscript.
Stéphane Ederhy declares that he has no conflict of interest with respect to this manuscript.
Anas Gazzah declares that he has no conflict of interest with respect to this manuscript.
Rastislav Bahleda declares that he has no conflict of interest with respect to this manuscript.
Christophe Massard has acted as consultant and advisory board for Astellas, Astra Zeneca, Bayer, Celgene, Genentech, Ipsen, Jansen, Lilly, Novartis, Pfizer, Roche, Sanofi, Orion, MedImmune, New Oncology, DebioPharm.
Isabelle Macquin-Mavier declares that he has no conflict of interest with respect to this manuscript.
Christophe Tournigand declares that he has no conflict of interest with respect to this manuscript.
Jean-Philippe Spano declares that he has no conflict of interest with respect to this manuscript.
Jean-Charles Soria declares that he has no conflict of interest with respect to this manuscript.
Benoît Rousseau received travel accommodation from Amgen, Novartis.
Funding
This work did not receive any financial support.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the national research committee and with the 1964 Helsinki declaration and its later amendments.
Human and animals rights and informed consent
This article does not contain any studies with animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Additional information
Emilie Boissier and Olivier Mir participated equally to the work
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 21 kb)
Rights and permissions
About this article
Cite this article
Boissier, E., Mir, O., Hollebecque, A. et al. Predictive factors of renal toxicities related to anti-VEGFR multikinase inhibitors in phase 1 trials. Invest New Drugs 35, 79–86 (2017). https://doi.org/10.1007/s10637-016-0402-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-016-0402-3