Abstract
Background
Serum estradiol levels in severe acute injury are correlated with in hospital mortality. In acute pancreatitis, serum estradiol levels are strong predictors of disease severity. Studies of whether changes in estradiol levels play a causative role in acute pancreatitis severity are limited. The ovariectomized mouse model has been used to study the effects of estradiol in health and disease.
Aims
We assessed whether the ovariectomized mouse model could be used to assess the effects of estradiol on pancreatitis severity.
Methods
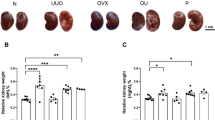
C57BL/6 mice with their ovaries removed were used to simulate low circulating estradiol conditions. Ovariectomized mice were treated with six hourly injections of cerulein to induce mild acute pancreatitis and compared to ovariectomized mice pre-treated with subcutaneous estradiol injections.
Results
Findings suggest ovariectomized model is a problematic preparation to study pancreatitis. At baseline, ovariectomy leads to prominent acinar cell ultrastructure changes as well as changes in other select morphologic and biomarkers of pancreatitis. In addition, ovariectomy changed select acute pancreatitis responses that were only partially rescued by estradiol pre-treatment.
Conclusions
These findings suggest that the ovariectomized mouse as a model of estradiol depletion should be used with caution in pancreatic studies. Future studies should explore whether derangements in other female hormones produced by the ovaries can lead to changes in pancreatic studies.







Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this published article.
References
Dossett LA, Swenson BR, Evans HL, Bonatti H, Sawyer RG, May AK. Serum estradiol concentration as a predictor of death in critically ill and injured adults. Surg Infect. 2008;9:41–48.
Kauffmann RM, Norris PR, Jenkins JM, Dupont WD, Torres RE, Blume JD et al. Trends in estradiol during critical illness are associated with mortality independent of admission estradiol. J Am Coll Surg. 2011;212:703–712.
Kumar RG, DiSanto D, Awan N, Vaughan LE, Levochkina MS, Weppner JL, et al. Temporal acute serum estradiol and tumor necrosis factor-α associations and risk of death after severe traumatic brain injury. J Neurotrauma. 2020;37:2198–2210.
Pappa T, Vemmos K, Mantzou E, Savvari P, Stamatelopoulos K, Alevizaki M. Estradiol levels predict short-term adverse health outcomes in postmenopausal acute stroke women. Eur J Neurol. 2012;19:1300–1304.
Mauvais-Jarvis F, Klein SL, Levin ER. Estradiol, Progesterone, Immunomodulation, and COVID-19 Outcomes. Endocrinology. 2020;161:bqaa127.
Lu C-W, Liu L-C, Hsieh Y-C, Yang L-H, Chen R-J, Hsieh C-H. Increased admission serum estradiol level is correlated with high mortality in patients with severe acute pancreatitis. J Gastroenterol. 2013;48:374–381.
Staubli SM, Oertli D, Nebiker CA. Laboratory markers predicting severity of acute pancreatitis. Crit Rev Clin Lab Sci. 2015;52:273–283.
Castro MR, Nguyen TT, O'Brien T. Clomiphene-induced severe hypertriglyceridemia and pancreatitis. In Mayo Clinic Proceedings. Vol. 74. Elsevier; 1999, pp. 1125-1128.
Rao K, Eagon P, Okamura K, Van Thiel D, Gavaler J, Kelly R et al. Acute hemorrhagic pancreatic necrosis in mice. Induction in male mice treated with estradiol. Am J Pathol. 1982;109:8.
Camporez JPG, Jornayvaz FR, Lee H-Y, Kanda S, Guigni BA, Kahn M et al. Cellular mechanism by which estradiol protects female ovariectomized mice from high-fat diet-induced hepatic and muscle insulin resistance. Endocrinology. 2013;154:1021–1028.
Hou Y, Wei H, Luo Y, Liu G. Modulating expression of brain heat shock proteins by estrogen in ovariectomized mice model of aging. Exp Gerontol. 2010;45:323–330.
Ohlsson C, Engdahl C, Fåk F, Andersson A, Windahl SH, Farman HH et al. Probiotics protect mice from ovariectomy-induced cortical bone loss. PloS One. 2014;9:e92368.
Sniekers Y, Weinans H, Bierma-Zeinstra S, Van Leeuwen J, Van Osch G. Animal models for osteoarthritis: the effect of ovariectomy and estrogen treatment–a systematic approach. Osteoarthr Cartil. 2008;16:533–541.
Turner RT, Maran A, Lotinun S, Hefferan T, Evans GL, Zhang M et al. Animal models for osteoporosis. Rev Endocr Metab Disord. 2001;2:117.
Kolodecik TR, Reed AM, Date K, Shugrue CA, Patel V, Chung S-L et al. The serum protein renalase reduces injury in experimental pancreatitis. J Biol Chem. 2017;292:21047–21059.
Wu L, Cai B, Zheng S, Liu X, Cai H, Li H. Effect of emodin on endoplasmic reticulum stress in rats with severe acute pancreatitis. Inflammation. 2013;36:1020–1029.
García ME, Borelli MI, Dumm CG, Gagliardino J, de Gagliardino EE. Functional and ultrastructural changes induced by short term ovariectomy on pancreatic islets. Hormone Metab Res. 1983;15:76–81.
Bhatia M. Apoptosis versus necrosis in acute pancreatitis. Am J Physiology-Gastrointest Liver Physiol. 2004;286:G189–G196.
Jie L, Liu X, Min W, Guo X, Zhan X. Changes of zymophagy during cell model of caerulein-induced experimental acute pancreatitis. Chin J Digest. 2014;34:752–755.
Sah RP, Saluja A. Molecular mechanisms of pancreatic injury. Curr Opin Gastroenterol. 2011;27:444.
Grossman A, Boctor AM, Band P, Lane B. Role of steroids in secretion—modulating effect of triamcinolone and estradiol on protein synthesis and secretion from the rat exocrine pancreas. J Steroid Biochem. 1983;19:1069–1081.
Xiang J, Liu X, Ren J, Chen K, Wang H-L, Miao Y-Y et al. How does estrogen work on autophagy? Autophagy. 2019;15:197–211.
Yang Y-H, Chen K, Li B, Chen J-W, Zheng X-F, Wang Y-R et al. Estradiol inhibits osteoblast apoptosis via promotion of autophagy through the ER–ERK–mTOR pathway. Apoptosis. 2013;18:1363–1375.
Yang Y, Zheng X, Li B, Jiang S, Jiang L. Increased activity of osteocyte autophagy in ovariectomized rats and its correlation with oxidative stress status and bone loss. Biochem Biophys Res Commun. 2014;451:86–92.
Choi S, Shin H, Song H, Lim HJ. Suppression of autophagic activation in the mouse uterus by estrogen and progesterone. J Endocrinol. 2014;221:39–50.
Wise PM, Ratner A. Effect of ovariectomy on plasma LH, FSH, estradiol, and progesterone and medial basal hypothalamic LHRH concentrations in old and young rats. Neuroendocrinology. 1980;30:15–19.
Blenck CL, Harvey PA, Reckelhoff JF, Leinwand LA. The Importance of biological sex and estrogen in rodent models of cardiovascular health and disease. Circ Res. 2016;118:1294–1312.
Acknowledgments
We would like to thank Thomas Pointer for his assistance in study design.
Funding
This study was funded by NIH Medical Student Fellowship to MW (DK007017) and a Veterans Administration Merit Award to FG (BX-003250).
Author information
Authors and Affiliations
Contributions
MW performed data acquisition, data interpretation and statistical analysis. MW and FG performed study concept, design, writing, review, and paper revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to report.
Consent to publish
No human participants were included in this study, and consent to publish was not required.
Ethics approval
All animals and procedures within this study were in compliance by the Animal Welfare Act, the Public Health Service Policy, and the Guide for the Care and Use of Laboratory Animals. This research protocol was approved by the Veterans Health Administration Institutional Animal Care and Use Committee (FG009, West Haven, CT, USA).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, M., Gorelick, F. Ovariectomy Affects Acute Pancreatitis in Mice. Dig Dis Sci 67, 2971–2980 (2022). https://doi.org/10.1007/s10620-021-07116-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-07116-w