Abstract
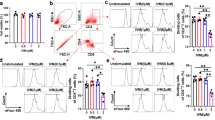
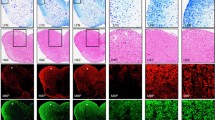
Zinc is an essential trace element with a critical role in normal growth and development and in immune homeostasis. Zinc deficiency impairs both the innate and the adaptive immune system and can be normalized by zinc supplementation. On the other end of the spectrum, high dosages of zinc diminish immune cell functions similar to zinc deficiency. Here, we investigated the influence of zinc aspartate on proliferation and cytokine production of stimulated human T cells and mouse splenocytes in vitro. Furthermore, the effect of zinc aspartate was examined in mice with experimental autoimmune encephalomyelitis (EAE), an animal model of Multiple Sclerosis (MS) with a Th1/Th17 T cell-mediated immunopathogenesis. Zinc aspartate suppressed proliferation as well as IL-2, IL-10 and IL-17 production in stimulated human T cells and mouse splenocytes. Importantly, administration of a medium range dose of 30 μg/day zinc aspartate [1.5 mg/kg body weight (BW)] in a therapeutic manner led to a significant reduction of the clinical severity of the EAE during the first relapse of the disease. A lower zinc aspartate dose (6 μg/day, 0.3 mg/kg BW) had no significant therapeutic effect on the severity of the EAE, while administration of higher zinc aspartate amounts (120 μg/day, 6 mg/kg BW) led to more severe disease. Taken together, our data suggest that zinc aspartate can modulate activation, proliferation and cytokine production of effector T cells in vitro and in vivo and that activated autoreactive T cells may be potential therapeutic targets of tightly controlled zinc supplementation in autoimmune diseases like MS.






Similar content being viewed by others
Abbreviations
- BW:
-
Body weight
- CFA:
-
Complete Freund’s adjuvant
- CNS:
-
Central nervous system
- EAE:
-
Experimental autoimmune encephalomyelitis
- ELISA:
-
Enzyme-linked immunosorbent assay
- IL:
-
Interleukin
- i.p:
-
Intraperitoneal
- MLC:
-
Mixed lymphocyte cultures
- MS:
-
Multiple sclerosis
- NK:
-
Natural killer
- PBMC:
-
Peripheral blood mononuclear cells
- PLP:
-
Proteolipid protein
- PWM:
-
Pokeweed mitogen
- PTX:
-
Pertussis toxin
References
Bettelli E, Oukka M, Kuchroo VK (2007) T(H)-17 cells in the circle of immunity and autoimmunity. Nat Immunol 8(4):345–350. doi:10.1038/ni0407-345
Biton A, Ansorge S, Bank U, Täger M, Reinhold D, Brocke S (2011) Divergent actions by inhibitors of DP IV and APN family enzymes on CD4+ Teff cell motility and functions. Immunobiology 216(12):1295–1301
Bray TM, Bettger WJ (1990) The physiological role of zinc as an antioxidant. Free Radic Biol Med 8(3):281–291
Brocke S, Quigley L, McFarland HF, Steinman L (1996) Isolation and characterization of autoreactive T cells in experimental autoimmune encephalomyelitis of the mouse. Methods 9(3):458–462
Cakman I, Kirchner H, Rink L (1997) Zinc supplementation reconstitutes the production of interferon-alpha by leukocytes from elderly persons. J Interferon Cytokine Res 17(8):469–472
Campo CA, Wellinghausen N, Faber C, Fischer A, Rink L (2001) Zinc inhibits the mixed lymphocyte culture. Biol Trace Element Res 79(1):15–22. doi:10.1385/BTER:79:1:15
Chang KL, Hung TC, Hsieh BS, Chen YH, Chen TF, Cheng HL (2006) Zinc at pharmacologic concentrations affects cytokine expression and induces apoptosis of human peripheral blood mononuclear cells. Nutrition 22(5):465–474. doi:10.1016/j.nut.2005.11.009
Codarri L, Gyülvészi G, Tosevski V, Hesske L, Fontana A, Magnenat L, Suter T, Becher B (2011) RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat Immunol 12(6):560–567. doi:10.1038/ni.2027
Dyment DA, Ebers GC, Sadovnick AD (2004) Genetics of multiple sclerosis. Lancet Neurol 3(2):104–110. doi:10.1016/S1474-4422(03)00663-X
El-Behi M, Ciric B, Dai H, Yan Y, Cullimore M, Safavi F, Zhang GX, Dittel BN, Rostami A (2011) The encephalitogenicity of T(H)17 cells is dependent on IL-1- and IL-23-induced production of the cytokine GM-CSF. Nat Immunol 12(6):568–575. doi:10.1038/ni.2031
Faber C, Gabriel P, Ibs KH, Rink L (2004) Zinc in pharmacological doses suppresses allogeneic reaction without affecting the antigenic response. Bone Marrow Transplant 33(12):1241–1246. doi:10.1038/sj.bmt.1704509
Fleming KK, Bovaird JA, Mosier MC, Emerson MR, LeVine SM, Marquis JG (2005) Statistical analysis of data from studies on experimental autoimmune encephalomyelitis. J Neuroimmunol 170(1–2):71–84. doi:10.1016/j.jneuroim.2005.08.020
Fosmire GJ (1990) Zinc toxicity. Am J Clin Nutr 51(2):225–227
Fukada T, Yamasaki S, Nishida K, Murakami M, Hirano T (2011) Zinc homeostasis and signaling in health and diseases: zinc signaling. J Biol Inorg Chem 16(7):1123–1134. doi:10.1007/s00775-011-0797-4
Haak S, Croxford AL, Kreymborg K, Heppner FL, Pouly S, Becher B, Waisman A (2009) RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. J Clin Invest 119(1):61–69. doi:10.1172/JCI35997
Hemmer B, Nessler S, Zhou D, Kieseier B, Hartung HP (2006) Immunopathogenesis and immunotherapy of multiple sclerosis. Nat Clin Pract Neurol 2(4):201–211. doi:10.1038/ncpneuro0154
Hofstetter HH, Ibrahim SM, Koczan D, Kruse N, Weishaupt A, Toyka KV, Gold R (2005) Therapeutic efficacy of IL-17 neutralization in murine experimental autoimmune encephalomyelitis. Cell Immunol 237(2):123–130. doi:10.1016/j.cellimm.2005.11.002
Hofstetter HH, Toyka KV, Tary-Lehmann M, Lehmann PV (2007) Kinetics and organ distribution of IL-17-producing CD4 cells in proteolipid protein 139–151 peptide-induced experimental autoimmune encephalomyelitis of SJL mice. J Immunol 178(3):1372–1378
Ibs KH, Rink L (2003) Zinc-altered immune function. J Nutr 133(5 Suppl 1):146S–1452S
Kaltenberg J, Plum LM, Ober-Blöbaum JL, Hönscheid A, Rink L, Haase H (2010) Zinc signals promote IL-2-dependent proliferation of T cells. Eur J Immunol 40(5):1496–1503. doi:10.1002/eji.200939574
Keen CL, Gershwin ME (1990) Zinc deficiency and immune function. Annu Rev Nutr 10:415–431. doi:10.1146/annurev.nu.10.070190.002215
Kitabayashi C, Fukada T, Kanamoto M, Ohashi W, Hojyo S, Atsumi T, Ueda N, Azuma I, Hirota H, Murakami M, Hirano T (2010) Zinc suppresses Th17 development via inhibition of STAT3 activation. Int Immunol 22(5):375–386. doi:10.1093/intimm/dxq017
Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y (2006) IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 177(1):566–573
Leopold IH (1981) United States Patent 4,255,419
Noseworthy JH, Lucchinetti C, Rodriguez M, Weinshenker BG (2000) Multiple sclerosis. N Engl J Med 343(13):938–952
Penkowa M, Hidalgo J (2000) Metallothionein I + II expression and their role in experimental autoimmune encephalomyelitis. Glia 32(3):247–263. doi:10.1002/1098-1136(200012)32:3<247:AID-GLIA50>3.0.CO;2-T
Porter KG, McMaster D, Elmes ME, Love AH (1977) Anaemia and low serum-copper during zinc therapy. Lancet 2(8041):774. doi:10.1016/S0140-6736(77)90295-1
Prasad AS (1998) Zinc and immunity. Mol Cell Biochem 188(1–2):63–69. doi:10.1023/A:1006868305749
Prasad AS (2000) Effects of zinc deficiency on Th1 and Th2 cytokine shifts. J Infect Dis 182(Suppl 1):S62–S68. doi:10.1086/315916
Prasad AS, Brewer GJ, Schoomaker EB, Rabbani P (1978) Hypocupremia induced by zinc therapy in adults. JAMA 240:2166–2168
Prasad AS, Bao B, Beck FW, Kucuk O, Sarkar FH (2004) Antioxidant effect of zinc in humans. Free Radic Biol Med 37(8):1182–1190. doi:10.1016/j.freeradbiomed.2004.07.007
Prasad AS, Beck FW, Bao B, Fitzgerald JT, Snell DC, Steinberg JD, Cardozo LJ (2007) Zinc supplementation decreases incidence of infections in the elderly: effect of zinc on generation of cytokines and oxidative stress. Am J Clin Nutr 85:837–844
Reinhold D, Ansorge S, Grüngreiff K (1997) Zinc regulates DNA synthesis and IL-2, IL-6 and IL-10 production of PWM-stimulated PBMC and normalizes the periphere cytokine concentration in chronic liver disease. J Trace Elem Exp Med 10:19–27. doi:10.1002/(SICI)1520-670X(1997)10:1<19:AID-JTRA3>3.0.CO;2-#
Reinhold D, Ansorge S, Grüngreiff K (1999) Immunobiology of zinc and zinc therapy. Immunol Today 20(2):102–103. doi:10.1016/S0167-5699(98)01400-5
Reinhold D, Bank U, Entz D, Goihl A, Stoye D, Wrenger S, Brocke S, Thielitz A, Stefin S, Nordhoff K, Heimburg A, Täger M, Ansorge S (2011) PETIR-001, a dual inhibitor of dipeptidyl peptidase IV (DP IV) and aminopeptidase N (APN), ameliorates experimental autoimmune encephalomyelitis in SJL/J mice. Biol Chem 392(3):233–237. doi:10.1515/BC.2011.024
Rink L, Haase H (2007) Zinc homeostasis and immunity. Trends Immunol 28(1):1–4. doi:10.1016/j.it.2006.11.005
Röhn TA, Jennings GT, Hernandez M, Grest P, Beck M, Zou Y, Kopf M, Bachmann MF (2006) Vaccination against IL-17 suppresses autoimmune arthritis and encephalomyelitis. Eur J Immunol 36(11):2857–2867. doi:10.1002/eji.200636658
Schiffer RB, Herndon RM, Eskin T (1990) Effects of altered dietary trace metals upon experimental allergic encephalomyelitis. Neurotoxicology 11(3):443–450
Steinbrecher A, Reinhold D, Quigley L, Gado A, Tresser N, Izikson L, Born I, Faust J, Neubert K, Martin R, Ansorge S, Brocke S (2001) Targeting dipeptidyl peptidase IV (CD26) suppresses autoimmune encephalomyelitis and up-regulates TGF-beta 1 secretion in vivo. J Immunol 166(3):2041–2048
Steinman L (2007) A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat Med 13(2):139–145. doi:10.1038/nm1551
Uciechowski P, Kahmann L, Plümäkers B, Malavolta M, Mocchegiani E, Dedoussis G, Herbein G, Jajte J, Fulop T, Rink L (2008) TH1 and TH2 cell polarization increases with aging and is modulated by zinc supplementation. Exp Gerontol 43(5):493–498. doi:10.1016/j.exger.2007.11.006
Uyttenhove C, Van Snick J (2006) Development of an anti-IL-17A auto-vaccine that prevents experimental auto-immune encephalomyelitis. Eur J Immunol 36(11):2868–2874. doi:10.1002/eji.200636662
Varin A, Larbi A, Dedoussis GV, Kanoni S, Jajte J, Rink L, Monti D, Malavolta M, Marcellini F, Mocchegiani E, Herbein G, Fulop T Jr (2008) In vitro and in vivo effects of zinc on cytokine signalling in human T cells. Exp Gerontol 43(5):472–482. doi:10.1016/j.exger.2007.12.008
Weaver CT, Harrington LE, Mangan PR, Gavrieli M, Murphy KM (2006) Th17 an effector CD4 T cell lineage with regulatory T cell ties. Immunity 24(6):677–688. doi:10.1016/j.immuni.2006.06.002
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852. doi:10.1146/annurev.immunol.25.022106.141557
Wellinghausen N, Rink L (1998) The significance of zinc for leukocyte biology. J Leukoc Biol 64(5):571–577
Wellinghausen N, Kirchner H, Rink L (1997) The immunobiology of zinc. Immunol Today 18(11):519–521. doi:10.1016/S0167-5699(97)01146-8
Yamasaki S, Sakata-Sogawa K, Hasegawa A, Suzuki T, Kabu K, Sato E, Kurosaki T, Yamashita S, Tokunaga M, Nishida K, Hirano T (2007) Zinc is a novel intracellular second messenger. J Cell Biol 177(4):637–645. doi:10.1083/jcb.200702081
Yu M, Lee WW, Tomar D, Pryshchep S, Czesnikiewicz-Guzik M, Lamar DL, Li G, Singh K, Tian L, Weyand CM, Goronzy JJ (2011) Regulation of T cell receptor signaling by activation-induced zinc influx. J Exp Med 208(4):775–785. doi:10.1084/jem.20100031
Conflict of Interest Statement
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stoye, D., Schubert, C., Goihl, A. et al. Zinc aspartate suppresses T cell activation in vitro and relapsing experimental autoimmune encephalomyelitis in SJL/J mice. Biometals 25, 529–539 (2012). https://doi.org/10.1007/s10534-012-9532-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-012-9532-z