Abstract
Background
Worldwide, stomas represent a medical and social problem. Data from the literature on stoma management are extensive but not homogeneous. In Italy, no guidelines exist for this topic. Thus, clear and comprehensive clinical guidelines based on evidence-based data and best practice are need. In 2018, the Multidisciplinary Italian Study group for STOmas, called MISSTO, was founded. The aim was to elaborate guidelines for practice management of enteral and urinary stomas in adults.
Methods
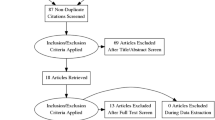
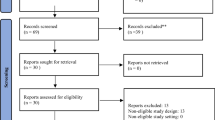
A systematic review of the literature was performed using PubMed, National Guideline Clearinghouse, and other databases. The research included guidelines, systematic reviews, meta-analyses, randomized clinical trials, cohort studies, and case reports. Five main topics were identified: “stoma preparation”, “stoma creation”, “stoma complications”, “stoma care”, and “stoma reversal”. The systematic review was performed for each topic, and studies were evaluated according to the GRADE system, AGREE II tool.
Results
Recommendations were elaborated in the form of statements with an established grade of recommendation for each statement. For low levels of scientific evidence statements, a consensus conference composed of expert members of the major Italian scientific societies in the field of stoma management and care was held. After discussing, correcting, validating, or eliminating the statements by the experts, the final version of the guidelines was elaborated and prepared for publication. This manuscript is focused on statements on the surgical management of enteral stomas.
Conclusions
These guidelines are the first Italian guidelines on multidisciplinary management of enteral stomas with the aim of assisting surgeons during stoma management and care.
Similar content being viewed by others
References
Goldberg M, Aukett LK, Carmel J et al (2010) Management of the patient with a fecal ostomy. J Wound Ostomy Cont Nurs 37:596–598. https://doi.org/10.1097/WON.0b013e3181f97e37
Italian Republic Senate (2015) Draft Law n. 2101. http://www.senato.it/leg/17/BGT/Schede/Ddliter/46103.htm#
De Salvo G (2005) Quanti siamo, sei anni dopo. Ritrovarci 2:15–16
Robertson I, Leung E, Hughes D et al (2005) Prospective analysis of stoma-related complications. Color Dis 7:279–285. https://doi.org/10.1111/j.1463-1318.2005.00785.x
Sheetz KH, Waits SA, Krell RW et al (2014) Complication rates of ostomy surgery are high and vary significantly between hospitals. Dis Colon Rectum 57:632–637. https://doi.org/10.1097/DCR.0000000000000038
Rojanasarot S (2017) The impact of early involvement in a postdischarge support program for ostomy surgery patients on preventable healthcare utilization. J Wound Ostomy Cont Nurs. https://doi.org/10.1097/WON.0000000000000395
Ferrara F, Rizzo G, Bondurri A et al (2019) Italian guidelines for the management of enteral and urinary stomas. Dis Colon Rectum 62:3–4. https://doi.org/10.1097/DCR.0000000000001235
Brouwers MC, Kho ME, Browman GP et al (2010) AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ 182:839–842. https://doi.org/10.1503/cmaj.090449
Guyatt G, Gutterman D, Baumann MH et al (2006) Grading strength of recommendations and quality of evidence in clinical guidelines. Chest 129:174–181. https://doi.org/10.1378/chest.129.1.174
Forsmo HM, Pfeffer F, Rasdal A et al (2016) Pre- and postoperative stoma education and guidance within an enhanced recovery after surgery (ERAS) programme reduces length of hospital stay in colorectal surgery. Int J Surg 36:121–126. https://doi.org/10.1016/j.ijsu.2016.10.031
Hendren S, Hammond K, Glasgow SC et al (2015) Clinical practice guidelines for ostomy surgery. Dis Colon Rectum 58:375–387. https://doi.org/10.1097/DCR.0000000000000347
Scottish Intercollegiate Guidelines Network (SIGN). (2011) Diagnosis and management of colorectal cancer
Ronco M, Iona L, Fabbro C et al (2012) Patient education outcomes in surgery: a systematic review from 2004 to 2010. Int J Evid Based Healthc 10:309–323. https://doi.org/10.1111/j.1744-1609.2012.00286.x
Lo S-F, Wang Y-T, Wu L-Y et al (2009) A cost-effectiveness analysis of a multimedia learning education program for stoma patients. J Clin Nurs 19:1844–1854. https://doi.org/10.1111/j.1365-2702.2009.02931.x
Chaudhri S, Brown L, Hassan I, Horgan AF (2005) Preoperative intensive, community-based vs. traditional stoma education: a randomized controlled trial. Dis Colon Rectum 48:504–509. https://doi.org/10.1007/s10350-004-0897-0
Zhang J, Wong FKY, You L et al (2013) Effects of enterostomal nurse telephone follow-up on postoperative adjustment of discharged colostomy patients. Cancer Nurs 36:419–428. https://doi.org/10.1097/NCC.0b013e31826fc8eb
Haugen V, Bliss DZ, Savik K (2006) Perioperative factors that affect long-term adjustment to an incontinent ostomy. J Wound Ostomy Cont Nurs 33:525–535
Millan M, Tegido M, Biondo S, García-Granero E (2009) Preoperative stoma siting and education by stomatherapists in colorectal cancer patients: a descriptive study of 12 colorectal surgery units in spain. Color Dis. https://doi.org/10.1111/j.1463-1318.2009.01942.x
Altuntas YE, Kement M, Gezen C et al (2012) The role of group education on quality of life in patients with a stoma. Eur J Cancer Care (Engl) 21:776–781. https://doi.org/10.1111/j.1365-2354.2012.01360.x
Faury S, Koleck M, Foucaud J et al (2017) Patient education interventions for colorectal cancer patients with stoma: a systematic review. Patient Educ Couns 100:1807–1819. https://doi.org/10.1016/j.pec.2017.05.034
Bass EM, Del Pino A, Tan A et al (1997) Does preoperative stoma marking and education by the enterostomal therapist affect outcome? Dis Colon Rectum 40:440–442
Mahjoubi B, Kiani Goodarzi K, Mohammad-Sadeghi H (2010) Quality of life in stoma patients: appropriate and inappropriate stoma sites. World J Surg 34:147–152. https://doi.org/10.1007/s00268-009-0275-0
Nastro P, Knowles CH, McGrath A et al (2010) Complications of intestinal stomas. Br J Surg 97:1885–1889. https://doi.org/10.1002/bjs.7259
Person B, Ifargan R, Lachter J et al (2012) The impact of preoperative stoma site marking on the incidence of complications, quality of life, and patient’s independence. Dis Colon Rectum 55:783–787. https://doi.org/10.1097/DCR.0b013e31825763f0
Baykara ZG, Demir SG, Karadag A et al (2014) A multicenter, retrospective study to evaluate the effect of preoperative stoma site marking on stomal and peristomal complications. Ostomy Wound Manage 60:16–26
Jayarajah U, Samarasekara AMP, Samarasekera DN (2016) A study of long-term complications associated with enteral ostomy and their contributory factors. BMC Res Notes 9:500. https://doi.org/10.1186/s13104-016-2304-z
McKenna LS, Taggart E, Stoelting J et al (2016) The impact of preoperative stoma marking on health-related quality of life. J Wound Ostomy Cont Nurs 43:57–61. https://doi.org/10.1097/WON.0000000000000180
Carlsson E, Fingren J, Hallén A-M et al (2016) The prevalence of ostomy-related complications 1 year after ostomy surgery: a prospective, descriptive, clinical study. Ostomy Wound Manage 62:34–48
Colwell JC, Gray M (2007) Does preoperative teaching and stoma site marking affect surgical outcomes in patients undergoing ostomy surgery? J Wound Ostomy Cont Nurs 34:492–496. https://doi.org/10.1097/01.WON.0000290726.08323.a6
Arumugam PJ, Bevan L, Macdonald L et al (2003) A prospective audit of stomas–analysis of risk factors and complications and their management. Colorectal Dis 5:49–52
Pittman J, Rawl SM, Schmidt CM et al (2008) Demographic and clinical factors related to ostomy complications and quality of life in veterans with an ostomy. J Wound Ostomy Cont Nurs 35:493–503. https://doi.org/10.1097/01.WON.0000335961.68113.cb
Koc U, Karaman K, Gomceli I et al (2017) A retrospective analysis of factors affecting early stoma complications. Ostomy Wound Manage 63:28–32
Arolfo S, Borgiotto C, Bosio G, Mistrangelo M, Allaix ME, Morino M (2018) Preoperative stoma site marking: a simple practice to reduce stoma-related complications. Tech Coloproctol 22:683–687. https://doi.org/10.1007/s10151-018-1857-3
Salvadalena G, Hendren S, McKenna L et al (2015) WOCN society and aua position statement on preoperative stoma site marking for patients undergoing urostomy surgery. J Wound Ostomy Cont Nurs 42:249–252. https://doi.org/10.1097/WON.0000000000000118
Roveron G, De Toma G, Barbierato M (2016) Italian society of surgery and association of stoma care nurses joint position statement on preoperative stoma siting. J Wound Ostomy Cont Nurs 43:165–169. https://doi.org/10.1097/WON.0000000000000204
Ludwig KA, Milsom JW, Garcia-Ruiz A, Fazio VW (1996) Laparoscopic techniques for fecal diversion. Dis Colon Rectum 39:285–288
Oliveira L, Reissman P, Nogueras J, Wexner SD (1997) Laparoscopic creation of stomas. Surg Endosc 11:19–23
Hollyoak MA, Lumley J, Stitz RW (1998) Laparoscopic stoma formation for faecal diversion. Br J Surg 85:226–228. https://doi.org/10.1046/j.1365-2168.1998.00516.x
Schwandner O, Schiedeck TH, Bruch HP (1998) Stoma creation for fecal diversion: is the laparoscopic technique appropriate? Int J Colorectal Dis 13:251–255
Young CJ, Eyers AA, Solomon MJ (1998) Defunctioning of the anorectum: historical controlled study of laparoscopic vs. open procedures. Dis Colon Rectum 41:190–194
Liu J, Bruch HP, Farke S et al (2005) Stoma formation for fecal diversion: a plea for the laparoscopic approach. Tech Coloproctol 9:9–14. https://doi.org/10.1007/s10151-005-0185-6
Scheidbach H, Ptok H, Schubert D et al (2009) Palliative stoma creation: comparison of laparoscopic vs conventional procedures. Langenbeck’s Arch Surg 394:371–374. https://doi.org/10.1007/s00423-007-0220-3
Gorgun E, Gezen FC, Aytac E et al (2015) Laparoscopic versus open fecal diversion: does laparoscopy offer better outcomes in short term? Tech Coloproctol 19:293–300. https://doi.org/10.1007/s10151-015-1295-4
Ivatury SJ, Bostock Rosenzweig IC, Holubar SD (2016) Short-term outcomes after open and laparoscopic colostomy creation. Dis Colon Rectum 59:543–550. https://doi.org/10.1097/DCR.0000000000000581
Ng KH, Ng DCK, Cheung HYS et al (2008) Obstructive complications of laparoscopically created defunctioning ileostomy. Dis Colon Rectum 51:1664–1668. https://doi.org/10.1007/s10350-008-9351-z
Hiranyakas A, Rather A, da Silva G et al (2013) Loop ileostomy closure after laparoscopic versus open surgery: is there a difference? Surg Endosc 27:90–94. https://doi.org/10.1007/s00464-012-2422-1
Williams NS, Nasmyth DG, Jones D, Smith AH (1986) De-functioning stomas: a prospective controlled trial comparing loop ileostomy with loop transverse colostomy. Br J Surg 73:566–570
Khoury GA, Lewis MC, Meleagros L, Lewis AA (1987) Colostomy or ileostomy after colorectal anastomosis?: a randomised trial. Ann R Coll Surg Engl 69:5–7
Gooszen AW, Geelkerken RH, Hermans J et al (1998) Temporary decompression after colorectal surgery: randomized comparison of loop ileostomy and loop colostomy. Br J Surg 85:76–79. https://doi.org/10.1046/j.1365-2168.1998.00526.x
Edwards DP, Leppington-Clarke A, Sexton R et al (2001) Stoma-related complications are more frequent after transverse colostomy than loop ileostomy: a prospective randomized clinical trial. Br J Surg 88:360–363. https://doi.org/10.1046/j.1365-2168.2001.01727.x
Law WL, Chu KW, Choi HK (2002) Randomized clinical trial comparing loop ileostomy and loop transverse colostomy for faecal diversion following total mesorectal excision. Br J Surg 89:704–708. https://doi.org/10.1046/j.1365-2168.2002.02082.x
Lertsithichai P, Rattanapichart P (2004) Temporary ileostomy versus temporary colostomy: a meta-analysis of complications. Asian J Surg 27:202–210. https://doi.org/10.1016/S1015-9584(09)60033-6
Güenaga KF, Lustosa SAS, Saad SS et al (2007) Ileostomy or colostomy for temporary decompression of colorectal anastomosis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd004647.pub2
Rondelli F, Reboldi P, Rulli A et al (2009) Loop ileostomy versus loop colostomy for fecal diversion after colorectal or coloanal anastomosis: a meta-analysis. Int J Colorectal Dis 24:479–488. https://doi.org/10.1007/s00384-009-0662-x
Chen J, Wang DR, Zhang JR et al (2013) Meta-analysis of temporary ileostomy versus colostomy for colorectal anastomoses. Acta Chir Belg 113:330–339
Geng HZ, Nasier D, Liu B et al (2015) Meta-analysis of elective surgical complications related to defunctioning loop ileostomy compared with loop colostomy after low anterior resection for rectal carcinoma. Ann R Coll Surg Engl 97:494–501. https://doi.org/10.1308/003588415X14181254789240
Li W, Stocchi L, Cherla D et al (2017) Factors associated with hospital readmission following diverting ileostomy creation. Tech Coloproctol 21:641–648. https://doi.org/10.1007/s10151-017-1667-z
Shaffer VO, Owi T, Kumarusamy MA et al (2017) Decreasing hospital readmission in ileostomy patients: results of novel pilot program. J Am Coll Surg 224:425–430. https://doi.org/10.1016/j.jamcollsurg.2016.12.030
Iqbal A, Raza A, Huang E et al (2017) Cost effectiveness of a novel attempt to reduce readmission after ileostomy creation. JSLS J Soc Laparoendosc Surg 21(e2016):00082. https://doi.org/10.4293/JSLS.2016.00082
Fish DR, Mancuso CA, Garcia-Aguilar JE et al (2017) Readmission after ileostomy creation. Ann Surg 265:379–387. https://doi.org/10.1097/SLA.0000000000001683
Paquette IM, Solan P, Rafferty JF et al (2013) Readmission for dehydration or renal failure after ileostomy creation. Dis Colon Rectum 56:974–979. https://doi.org/10.1097/DCR.0b013e31828d02ba
Messaris E, Sehgal R, Deiling S et al (2012) Dehydration is the most common indication for readmission after diverting ileostomy creation. Dis Colon Rectum 55:175–180. https://doi.org/10.1097/DCR.0b013e31823d0ec5
Baker ML, Williams RN, Nightingale JMD (2011) Causes and management of a high-output stoma. Color Dis 13:191–197. https://doi.org/10.1111/j.1463-1318.2009.02107.x
Gooszen AW, Geelkerken RH, Hermans J et al (2000) Quality of life with a temporary stoma: ileostomy vs. colostomy. Dis Colon Rectum 43:650–655
Silva MA, Ratnayake G, Deen KI (2003) Quality of life of stoma patients: temporary ileostomy versus colostomy. World J Surg 27:421–424. https://doi.org/10.1007/s00268-002-6699-4
Bruns BR, DuBose J, Pasley J et al (2015) Loop versus end colostomy reversal: has anything changed? Eur J Trauma Emerg Surg 41:539–543. https://doi.org/10.1007/s00068-014-0444-1
Mak GZ, Harberg FJ, Hiatt P et al (2000) T-tube ileostomy for meconium ileus: four decades of experience. J Pediatr Surg 35:349–352
Lizarralde E (1981) Typhoid perforation of the ileum in children. J Pediatr Surg 16:1012–1016
Hojo K (1985) Total colectomy, rectal mucosectomy and ileoanal anastomosis for familial polyposis coli–use of tube ileostomy. Jpn J Clin Oncol 15:661–669
Zong Z, Zhou T, Jiang Z et al (2016) Temporary tube stoma versus conventional loop stoma for the protection of a low anastomosis in colorectal surgery: a systematic review and meta-analysis. Am Surg 82:251–258
Senapati A, Phillips RK (1991) The trephine colostomy: a permanent left iliac fossa end colostomy without recourse to laparotomy. Ann R Coll Surg Engl 73:305–306
Parithivel VS, Schein M, Gerst PH (2003) Colonoscopy-assisted “trephine” sigmoid colostomy. Dig Surg 20:103–106. https://doi.org/10.1159/000069380
Brand M, Dujovny N (2008) Preoperative considerations and creation of normal ostomies. Clin Colon Rectal Surg 21:005–016. https://doi.org/10.1055/s-2008-1055316
Speirs M, Leung E, Hughes D et al (2006) Ileostomy rod–Is it bridge too far? Color Dis 8:484–487. https://doi.org/10.1111/j.1463-1318.2005.00923.x
Zindel J, Gygax C, Studer P et al (2017) A sustaining rod increases necrosis of loop ileostomies: a randomized controlled trial. Int J Colorectal Dis 32:875–881. https://doi.org/10.1007/s00384-017-2813-9
Uchino M, Ikeuchi H, Bando T et al (2017) Is an ostomy rod useful for bridging the retraction during the creation of a loop ileostomy? a randomized control trial. World J Surg 41:2128–2135. https://doi.org/10.1007/s00268-017-3978-7
Franklyn J, Varghese G, Mittal R et al (2017) A prospective randomized controlled trial comparing early postoperative complications in patients undergoing loop colostomy with and without a stoma rod. Color Dis 19:675–680. https://doi.org/10.1111/codi.13600
Whiteley I, Russell M, Nassar N, Gladman MA (2016) Outcomes of support rod usage in loop stoma formation. Int J Colorectal Dis 31:1189–1195. https://doi.org/10.1007/s00384-016-2569-7
Dziki Ł, Mik M, Trzciński R et al (2015) Evaluation of the early results of a loop stoma with a plastic rod in comparison to a loop stoma made with a skin bridge. Pol Prz Chir Polish J Surg 87:31–34. https://doi.org/10.1515/pjs-2015-0014
Harish K (2008) The loop stoma bridge-A new technique. J Gastrointest Surg 12:958–961. https://doi.org/10.1007/s11605-007-0413-7
Lafreniere R, Ketcham AS (1985) The Penrose drain: a safe, atraumatic colostomy bridge. Am J Surg 149:288–291
Scarpa M, Sadocchi L, Ruffolo C et al (2007) Rod in loop ileostomy: just an insignificant detail for ileostomy-related complications? Langenbeck’s Arch Surg 392:149–154. https://doi.org/10.1007/s00423-006-0105-x
Parmar KL, Zammit M, Smith A et al (2011) A prospective audit of early stoma complications in colorectal cancer treatment throughout the Greater Manchester and Cheshire colorectal cancer network. Color Dis 13:935–938. https://doi.org/10.1111/j.1463-1318.2010.02325.x
Cottam J, Richards K, Hasted A, Blackman A (2007) Results of a nationwide prospective audit of stoma complications within 3 weeks of surgery. Color Dis 9:834–838. https://doi.org/10.1111/j.1463-1318.2007.01213.x
Lin AY (2013) End ileostomy and loop ileostomy. In: Fleshman J (ed) Atlas of surgical techniques for colon, rectum and anus. Saunders, Philadelphia, pp 354–362
Lin AY, Birnbaum EH (2013) Colostomy: end and divided loop. In: Fleshman J (ed) Atlas of surgical techniques for colon, rectum and anus. Saunders, Philadelphia, pp 363–370
Gore DC (2010) Brooke ileostomy. In: Townsend C, Evers B (eds) Atlas of general surgical techniques. Saunders, Philadelphia, pp 627–634
Gore DC (2010) Loop colostomy. In: Townsend C, Evers B (eds) Atlas of general surgical techniques. Saunders, Philadelphia, pp 635–640
Gore DC (2010) Diverting end colostomy with mucous fistula or hartmann’s pouch. In: Townsend C, Evers B (eds) Atlas of general surgical techniques. Saunders, Philadelphia, pp 617–626
Couëtte C, Dumont F, Thibaudeau E (2018) Techniques des colostomies et traitement de leurs complications. EMC Techniques chirurgicales. Elsevier Masson, Paris, pp 1–25
Godiris-Petit G, Leyre P, Trésallet C, Ménégaux F (2010) Entérostomies chirurgicales. EMC Techniques chirurgicales. Elsevier Masson, Paris, pp 1–13
Hardt J, Seyfried S, Weiß C et al (2016) A pilot single-centre randomized trial assessing the safety and efficacy of lateral pararectus abdominis compared with transrectus abdominis muscle stoma placement in patients with temporary loop ileostomies: the PATRASTOM trial. Color Dis 18:O81–O90. https://doi.org/10.1111/codi.13251
Antoniou SA, Agresta F, Garcia Alamino JM et al (2018) European hernia society guidelines on prevention and treatment of parastomal hernias. Hernia 22:183–198. https://doi.org/10.1007/s10029-017-1697-5
Li W, Benlice C, Stocchi L et al (2017) Does stoma site specimen extraction increase postoperative ileostomy complication rates? Surg Endosc 31:3552–3558. https://doi.org/10.1007/s00464-016-5384-x
Hardt J, Meerpohl JJ, Metzendorf MI et al (2013) Lateral pararectal versus transrectal stoma placement for prevention of parastomal herniation. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD009487.pub2
Chapman SJ, Wood B, Drake TM et al (2017) Systematic review and meta-analysis of prophylactic mesh during primary stoma formation to prevent parastomal hernia. Dis Colon Rectum 60:107–115. https://doi.org/10.1097/DCR.0000000000000670
Cornille JB, Pathak S, Daniels IR, Smart NJ (2017) Prophylactic mesh use during primary stoma formation to prevent parastomal hernia. Ann R Coll Surg Engl 99:2–11. https://doi.org/10.1308/rcsann.2016.0186
Cross AJ, Buchwald PL, Frizelle FA, Eglinton TW (2017) Meta-analysis of prophylactic mesh to prevent parastomal hernia. Br J Surg 104:179–186. https://doi.org/10.1002/bjs.10402
Hauters P, Cardin JL, Lepere M et al (2016) Long-term assessment of parastomal hernia prevention by intra-peritoneal mesh reinforcement according to the modified Sugarbaker technique. Surg Endosc 30:5372–5379. https://doi.org/10.1007/s00464-016-4891-0
López-Cano M, Brandsma HT, Bury K et al (2017) Prophylactic mesh to prevent parastomal hernia after end colostomy: a meta-analysis and trial sequential analysis. Hernia 21:177–189. https://doi.org/10.1007/s10029-016-1563-x
Patel SV, Zhang L, Chadi SA, Wexner SD (2017) Prophylactic mesh to prevent parastomal hernia: a meta-analysis of randomized controlled studies. Tech Coloproctol 21:5–13. https://doi.org/10.1007/s10151-016-1559-7
Pianka F, Probst P, Keller AV et al (2017) Prophylactic mesh placement for the PREvention of paraSTOmal hernias: the PRESTO systematic review and meta-analysis. PLoS One 12:1–15. https://doi.org/10.1371/journal.pone.0171548
Wang S, Wang W, Zhu B et al (2016) Efficacy of prophylactic mesh in end-colostomy construction: a systematic review and meta-analysis of randomized controlled trials. World J Surg 40:2528–2536. https://doi.org/10.1007/s00268-016-3576-0
Zhu J, Pu Y, Yang X et al (2016) Prophylactic mesh application during colostomy to prevent parastomal hernia: a meta-analysis. Gastroenterol Res Pract 2016:1–10. https://doi.org/10.1155/2016/1694265
Odensten C, Strigård K, Rutegård J et al (2019) Use of prophylactic mesh when creating a colostomy does not prevent parastomal hernia: a randomized controlled trial-stomamesh. Ann Surg 269:427–431. https://doi.org/10.1097/SLA.0000000000002542
Findlay JM, Wood CPJ, Cunningham C (2018) Prophylactic mesh reinforcement of stomas: a cost-effectiveness meta-analysis of randomised controlled trials. Tech Coloproctol 22:265–270. https://doi.org/10.1007/s10151-018-1774-5
Berger D (2008) Prevention of parastomal hernias by prophylactic use of a specially designed intraperitoneal onlay mesh (Dynamesh IPST). Hernia 12:243–246. https://doi.org/10.1007/s10029-007-0318-0
Köhler G, Hofmann A, Lechner M et al (2016) Prevention of parastomal hernias with 3D funnel meshes in intraperitoneal onlay position by placement during initial stoma formation. Hernia 20:151–159. https://doi.org/10.1007/s10029-015-1380-7
Conde-Muíño R, Díez J-L, Martínez A et al (2017) Preventing parastomal hernias with systematic intraperitoneal specifically designed mesh. BMC Surg 17:41. https://doi.org/10.1186/s12893-017-0237-7
Ng ZQ, Tan P, Theophilus M (2017) Stapled mesh stoma reinforcement technique (smart) in the prevention of parastomal hernia: a single-centre experience. Hernia 21:469–475. https://doi.org/10.1007/s10029-016-1548-9
Canda AE, Terzi C, Agalar C et al (2018) Preventing parastomal hernia with modified stapled mesh stoma reinforcement technique (SMART) in patients who underwent surgery for rectal cancer: a case–control study. Hernia 22:379–384. https://doi.org/10.1007/s10029-017-1723-7
Guarnero V, Hoffmann H, Hetzer F et al (2016) A new stomaplasty ring (Koring™) to prevent parastomal hernia: an observational multicenter Swiss study. Tech Coloproctol 20:293–297. https://doi.org/10.1007/s10151-016-1452-4
Lykke A, Andersen JFB, Jorgensen LN, Mynster T (2017) Prevention of parastomal hernia in the emergency setting. Langenbeck’s Arch Surg. https://doi.org/10.1007/s00423-017-1596-3
Shabbir J, Chaudhary BN, Dawson R (2012) A systematic review on the use of prophylactic mesh during primary stoma formation to prevent parastomal hernia formation. Color Dis 14:931–936. https://doi.org/10.1111/j.1463-1318.2011.02835.x
Hammond TM, Huang A, Prosser K et al (2008) Parastomal hernia prevention using a novel collagen implant: a randomised controlled phase 1 study. Hernia 12:475–481. https://doi.org/10.1007/s10029-008-0383-z
Jänes A, Cengiz Y, Israelsson LA (2010) Experiences with a prophylactic mesh in 93 consecutive ostomies. World J Surg 34:1637–1640. https://doi.org/10.1007/s00268-010-0492-6
Serra-Aracil X, Bombardo-Junca J, Moreno-Matias J et al (2009) Randomized, controlled, prospective trial of the use of a mesh to prevent parastomal hernia. Ann Surg 249:583–587. https://doi.org/10.1097/SLA.0b013e31819ec809
Fortelny RH, Hofmann A, May C et al (2015) Prevention of a parastomal hernia by biological mesh reinforcement. Front Surg 2:53. https://doi.org/10.3389/fsurg.2015.00053
Köckerling F, Alam NN, Antoniou SA et al (2018) What is the evidence for the use of biologic or biosynthetic meshes in abdominal wall reconstruction? Hernia 22:249–269. https://doi.org/10.1007/s10029-018-1735-y
Kroese LF, De Smet GHJ, Jeekel J et al (2016) Systematic review and meta-analysis of extraperitoneal versus transperitoneal colostomy for preventing parastomal hernia. Dis Colon Rectum 59:688–695. https://doi.org/10.1097/DCR.0000000000000605
Lian L, Wu XR, He XS et al (2012) Extraperitoneal vs. intraperitoneal route for permanent colostomy: a meta-analysis of 1,071 patients. Int J Colorectal Dis 27:59–64. https://doi.org/10.1007/s00384-011-1293-6
Mari FS, Di Cesare T, Novi L et al (2015) Does ghost ileostomy have a role in the laparoscopic rectal surgery era? A randomized controlled trial. Surg Endosc 29:2590–2597. https://doi.org/10.1007/s00464-014-3974-z
Miccini M, Bonapasta SA, Gregori M et al (2010) Ghost ileostomy: real and potential advantages. Am J Surg 200:e55–e57. https://doi.org/10.1016/j.amjsurg.2009.12.017
Gullà N, Trastulli S, Boselli C et al (2011) Ghost ileostomy after anterior resection for rectal cancer: a preliminary experience. Langenbeck’s Arch Surg 396:997–1007. https://doi.org/10.1007/s00423-011-0793-8
Cerroni M, Cirocchi R, Morelli U et al (2011) Ghost Ileostomy with or without abdominal parietal split. World J Surg Oncol 9:92. https://doi.org/10.1186/1477-7819-9-92
Mori L, Vita M, Razzetta F et al (2013) Ghost ileostomy in anterior resection for rectal carcinoma: is it worthwhile? Dis Colon Rectum 56:29–34. https://doi.org/10.1097/DCR.0b013e3182716ca1
Lago V, Domingo S, Matute L et al (2017) Ghost ileostomy in advanced ovarian cancer. Gynecol Oncol 147:488. https://doi.org/10.1016/j.ygyno.2017.08.017
Donahue TF, Bochner BH, Sfakianos JP et al (2014) Risk factors for the development of parastomal hernia after radical cystectomy. J Urol 191:1708–1713. https://doi.org/10.1016/j.juro.2013.12.041
Liu NW, Hackney JT, Gellhaus PT et al (2014) Incidence and risk factors of parastomal hernia in patients undergoing radical cystectomy and ileal conduit diversion. J Urol 191:1313–1318. https://doi.org/10.1016/j.juro.2013.11.104
Narang SK, Alam NN, Campain NJ et al (2017) Parastomal hernia following cystectomy and ileal conduit urinary diversion: a systematic review. Hernia 21:163–175. https://doi.org/10.1007/s10029-016-1561-z
Funahashi K, Suzuki T, Nagashima Y et al (2014) Risk factors for parastomal hernia in Japanese patients with permanent colostomy. Surg Today 44:1465–1469. https://doi.org/10.1007/s00595-013-0721-3
De Raet J, Delvaux G, Haentjens P, Van Nieuwenhove Y (2008) Waist circumference is an independent risk factor for the development of parastomal hernia after permanent colostomy. Dis Colon Rectum 51:1806–1809. https://doi.org/10.1007/s10350-008-9366-5
Harilingam M, Sebastian J, Twum-Barima C et al (2017) Patient-related factors influence the risk of developing intestinal stoma complications in early post-operative period. ANZ J Surg 87:E116–E120. https://doi.org/10.1111/ans.13397
Nybæk H, Knudsen DB, Laursen TN et al (2009) Skin problems in ostomy patients: a case-control study of risk factors. Acta Derm Venereol 89:64–67. https://doi.org/10.2340/00015555-0536
Sung YH, Kwon I, Jo S, Park S (2010) Factors affecting ostomy-related complications in Korea. J Wound Ostomy Cont Nurs 37:166–172. https://doi.org/10.1097/WON.0b013e3181cf7b76
Caricato M, Ausania F, Ripetti V et al (2007) Retrospective analysis of long-term defunctioning stoma complications after colorectal surgery. Color Dis 9:559–561. https://doi.org/10.1111/j.1463-1318.2006.01187.x
Persson E, Berndtsson I, Carlsson E et al (2010) Stoma-related complications and stoma size - a 2-year follow up. Color Dis 12:971–976. https://doi.org/10.1111/j.1463-1318.2009.01941.x
Wu XR, Mukewar S, Kiran RP et al (2013) Risk factors for peristomal pyoderma gangrenosum complicating inflammatory bowel disease. J Crohn’s Colitis 7:e171–e177. https://doi.org/10.1016/j.crohns.2012.08.001
Alves A, Panis Y, Lelong B et al (2008) Randomized clinical trial of early versus delayed temporary stoma closure after proctectomy. Br J Surg 95:693–698. https://doi.org/10.1002/bjs.6212
Park J, Danielsen AK, Angenete E et al (2018) Quality of life in a randomized trial of early closure of temporary ileostomy after rectal resection for cancer (EASY trial). Br J Surg 105:244–251. https://doi.org/10.1002/bjs.10680
Lasithiotakis K, Aghahoseini A, Alexander D (2016) Is early reversal of defunctioning ileostomy a shorter, easier and less expensive operation? World J Surg 40:1737–1740. https://doi.org/10.1007/s00268-016-3448-7
Nelson T, Pranavi A, Sureshkumar S et al (2018) Early versus conventional stoma closure following bowel surgery: a randomized controlled trial. Saudi J Gastroenterol 24:52. https://doi.org/10.4103/sjg.SJG_445_17
Farag S, Rehman S, Sains P et al (2017) Early vs delayed closure of loop defunctioning ileostomy in patients undergoing distal colorectal resections: an integrated systematic review and meta-analysis of published randomized controlled trials. Color Dis 19:1050–1057. https://doi.org/10.1111/codi.13922
Menahem B, Lubrano J, Vallois A, Alves A (2018) Early closure of defunctioning loop ileostomy: is it beneficial for the patient? a meta-analysis. World J Surg 42:3171–3178. https://doi.org/10.1007/s00268-018-4603-0
Di Carlo I, Toro A, Pannofino O et al (2010) Laparoscopic versus open restoration of intestinal continuity after Hartmann procedure. Hepatogastroenterology 57:232–235
Keck JO, Collopy BT, Ryan PJ et al (1994) Reversal of hartmann’s procedure: effect of timing and technique on ease and safety. Dis Colon Rectum 37:243–248
Slawik S, Dixon AR (2008) Laparoscopic reversal of Hartmann’s rectosigmoidectomy. Colorectal Dis 10:81–83. https://doi.org/10.1111/j.1463-1318.2007.01243.x
Fleming FJ, Gillen P (2009) Reversal of Hartmann’s procedure following acute diverticulitis: is timing everything? Int J Colorectal Dis 24:1219–1225. https://doi.org/10.1007/s00384-009-0747-6
Löffler T, Rossion I, Bruckner T et al (2012) HAnd suture versus stapling for closure of loop ileostomy (HASTA trial). Ann Surg 256:828–836. https://doi.org/10.1097/SLA.0b013e318272df97
Shelygin YA, Chernyshov SV, Rybakov EG (2010) Stapled ileostomy closure results in reduction of postoperative morbidity. Tech Coloproctol 14:19–23. https://doi.org/10.1007/s10151-009-0550-y
Hull TL, Kobe I, Fazio VW (1996) Comparison of handsewn with stapled loop ileostomy closures. Dis Colon Rectum 39:1086–1089
Hasegawa H, Radley S, Morton DG, Keighley MR (2000) Stapled versus sutured closure of loop ileostomy: a randomized controlled trial. Ann Surg 231:202–204
Madani R, Day N, Kumar L et al (2018) Hand-Sewn versus stapled closure of loop ileostomy: a meta-analysis. Dig Surg. https://doi.org/10.1159/000487310
Löffler T, Rossion I, Gooßen K et al (2015) Hand suture versus stapler for closure of loop ileostomy—a systematic review and meta-analysis of randomized controlled trials. Langenbeck’s Arch Surg 400:193–205. https://doi.org/10.1007/s00423-014-1265-8
Sajid MS, Craciunas L, Baig MK, Sains P (2013) Systematic review and meta-analysis of published, randomized, controlled trials comparing suture anastomosis to stapled anastomosis for ileostomy closure. Tech Coloproctol 17:631–639. https://doi.org/10.1007/s10151-013-1027-6
Markides GA, Wijetunga IU, Brown SR, Anwar S (2015) Meta-analysis of handsewn versus stapled reversal of loop ileostomy. ANZ J Surg 85:217–224. https://doi.org/10.1111/ans.12684
Celentano V, Giglio MC, Bucci L (2015) Laparoscopic versus open Hartmann’s reversal: a systematic review and meta-analysis. Int J Colorectal Dis 30:1603–1615. https://doi.org/10.1007/s00384-015-2325-4
Siddiqui MRS, Sajid MS, Baig MK (2010) Open vs laparoscopic approach for reversal of Hartmann’s procedure: a systematic review. Colorectal Dis 12:733–741. https://doi.org/10.1111/j.1463-1318.2009.01892.x
Sherman KL, Wexner SD (2017) Considerations in stoma reversal. Clin Colon Rectal Surg 30:172–177. https://doi.org/10.1055/s-0037-1598157
Banerjee A (1997) Pursestring skin closure after stoma reversal. Dis Colon Rectum 40:993–994
Rondelli F, Franco L, Balzarotti Canger RC et al (2018) Purse-string closure versus conventional primary closure of wound following stoma reversal: meta-analysis of randomized controlled trials. Int J Surg 52:208–213. https://doi.org/10.1016/j.ijsu.2018.02.027
Sajid MS, Bhatti MI, Miles WFA (2015) Systematic review and meta-analysis of published randomized controlled trials comparing purse-string vs conventional linear closure of the wound following ileostomy (stoma) closure. Gastroenterol Rep 3:156–161. https://doi.org/10.1093/gastro/gou038
Hsieh M-C, Kuo L-T, Chi C-C et al (2015) Pursestring closure versus conventional primary closure following stoma reversal to reduce surgical site infection rate. Dis Colon Rectum 58:808–815. https://doi.org/10.1097/DCR.0000000000000401
McCartan DP, Burke JP, Walsh SR, Coffey JC (2013) Purse-string approximation is superior to primary skin closure following stoma reversal: a systematic review and meta-analysis. Tech Coloproctol 17:345–351. https://doi.org/10.1007/s10151-012-0970-y
Li LT, Hicks SC, Davila JA et al (2014) Circular closure is associated with the lowest rate of surgical site infection following stoma reversal: a systematic review and multiple treatment meta-analysis. Colorectal Dis 16:406–416. https://doi.org/10.1111/codi.12556
Li LT, Brahmbhatt R, Hicks SC et al (2014) Prevalence of surgical site infection at the stoma site following four skin closure techniques: a retrospective cohort study. Dig Surg 31:73–78. https://doi.org/10.1159/000354426
Camacho-Mauries D, Rodriguez-Díaz JL, Salgado-Nesme N et al (2013) Randomized clinical trial of intestinal ostomy takedown comparing pursestring wound closure vs conventional closure to eliminate the risk of wound infection. Dis Colon Rectum 56:205–211. https://doi.org/10.1097/DCR.0b013e31827888f6
Alvandipour M, Gharedaghi B, Khodabakhsh H, Karami MY (2016) Purse-string versus linear conventional skin wound closure of an ileostomy: a randomized clinical trial. Ann Coloproctol 32:144. https://doi.org/10.3393/ac.2016.32.4.144
O’Leary DP, Carter M, Wijewardene D et al (2017) The effect of purse-string approximation versus linear approximation of ileostomy reversal wounds on morbidity rates and patient satisfaction: the “STOMA” trial. Tech Coloproctol 21:863–868. https://doi.org/10.1007/s10151-017-1713-x
Lee JT, Marquez TT, Clerc D et al (2014) Pursestring closure of the stoma site leads to fewer wound infections. Dis Colon Rectum 57:1282–1289. https://doi.org/10.1097/DCR.0000000000000209
Juratli MA, Nour-Eldin N-EA, Ackermann H et al (2018) Purse-string closure technique reduces the incidence of incisional hernias following the reversal of temporary ileostomy. Int J Colorectal Dis 33:973–977. https://doi.org/10.1007/s00384-018-2986-x
Livingston DH, Miller FB, Richardson JD (1989) Are the risks after colostomy closure exaggerated? Am J Surg 158:17–20
Riesener KP, Lehnen W, Höfer M et al (1997) Morbidity of ileostomy and colostomy closure: impact of surgical technique and perioperative treatment. World J Surg 21:103–108
Demetriades D, Pezikis A, Melissas J et al (1988) Factors influencing the morbidity of colostomy closure. Am J Surg 155:594–596
Mirbagheri N, Dark J, Skinner S (2013) Factors predicting stomal wound closure infection rates. Tech Coloproctol 17:215–220. https://doi.org/10.1007/s10151-012-0908-4
Haase O, Raue W, Böhm B et al (2005) Subcutaneous gentamycin implant to reduce wound infections after loop-ileostomy closure: a randomized, double-blind, placebo-controlled trial. Dis Colon Rectum 48:2025–2031. https://doi.org/10.1007/s10350-005-0164-z
Acknowledgements
Special thanks to AIOSS for the original contribution and support to the project, with great ability to aggregate different healthcare professionals and scientific societies.
Contributing Members of the MISSTO project: Maria Dolores D’Elia, Patrizio Capelli, Roberto Dino Villani, Adolfo Renzi, Salvatore Siracusano, Maria Russo, Concetta Balzotti, Stefano Mancini, Roberto Aloesio, Loriano Bagnoli, Antonio Ferrazzano, Antonio D’Elia and Maria De Pasquale, on behalf of: AIOSS (Italian Association of Stoma Care Operators), SIC (Italian Society of Surgery), ACOI (Italian Association of Hospital Surgeons), SICCR (Italian Society of ColoRectal Surgery), SICO (Italian Society of Surgical Oncology), SIUCP (Italian Unitary Society of ColoProctology), SIU (Italian Society of Urology), AIURO (Italian Association of Urologic Nurses), AMICI (Association of Inflammatory Bowel Diseases), FAIS (Federative Association of Incontinent and Stoma Patients), and AISTOM (Italian Association of Stoma Patients).
Funding
Travels and/or accommodations to meetings for the study group were supported by AIOSS (Associazione Italiana Operatori Sanitari Stomaterapia); travels to the first meeting for urologists were supported by SIU (Società Italiana di Urologia). These supports did not influence the content of the guidelines.
Author information
Authors and Affiliations
Consortia
Contributions
All authors contributed to the study conception, design, material preparation, data collection, and analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000.
Informed consent
No need of informed consent since no human subject was included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Multidisciplinary Italian Study group for STOmas (MISSTO) contributors are listed in the Acknowledgement section.
About this article
Cite this article
Ferrara, F., Parini, D., Bondurri, A. et al. Italian guidelines for the surgical management of enteral stomas in adults. Tech Coloproctol 23, 1037–1056 (2019). https://doi.org/10.1007/s10151-019-02099-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-019-02099-3