Abstract
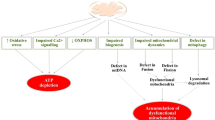
The mitochondrial theory of ageing proposes that accumulation of damage to mitochondrial function and DNA mutation lead to ageing of humans and animals. It has been suggested that mitochondria play dynamic roles in regulating synaptogenesis and morphological/functional responses of synaptic activity, and thus, deteriorating of mitochondrial function (e.g., deficits of the mitochondrial respiratory enzymes, reduced calcium influx, increased accumulation of mitochondrial DNA defects/apoptotic proteins and impairment of mitochondrial membrane potential) can lead to severe neuronal energy deficit, and in the long run, to modifications in neuronal synapses and neurodegeneration in the ageing brain. Hence, considering the mechanisms by which mitochondrial impairment can lead to neuronal death, the development of neuroprotective molecules that target various mitochondrial pathogenic processes can be effective in the treatment of ageing and age-related neurodegenerative diseases. This review addresses several aspects of the neuroprotective effects of propargylamine derivatives (e.g., the monoamine oxidase-B inhibitors, selegiline and rasagiline and the multifunctional drugs, ladostigil, M30 and VAR10303) in ageing with a special focus on mitochondrial molecular protective mechanisms.



Similar content being viewed by others
References
Akao Y, Maruyama W, Shimizu S, Yi H, Nakagawa Y, Shamoto-Nagai M, Youdim MB, Tsujimoto Y, Naoi M (2002a) Mitochondrial permeability transition mediates apoptosis induced by N-methyl(R)salsolinol, an endogenous neurotoxin, and is inhibited by Bcl- 2 and rasagiline, N-propargyl-1(R)-aminoindan. J Neurochem 82(4):913–923
Akao Y, Maruyama W, Yi H, Shamoto-Nagai M, Youdim MBH, Naoi M (2002b) An anti-Parkinson’s disease drug, N-propargyl-1(R)-aminoindan (rasagiline), enhances expression of anti-apoptotic Bcl-2 in human dopaminergic SH-SY5Y cells. Neurosci Lett 326(2):105–108
Anandatheerthavarada HK, Biswas G, Robin MA, Avadhani NG (2003) Mitochondrial targeting and a novel transmembrane arrest of Alzheimer’s amyloid precursor protein impairs mitochondrial function in neuronal cells. J Cell Biol 161(1):41–54
Avramovich-Tirosh Y, Amit T, Bar-Am O, Zheng H, Fridkin M, Youdim MB (2007) Therapeutic targets and potential of the novel brain- permeable multifunctional iron chelator-monoamine oxidase inhibitor drug, M-30, for the treatment of Alzheimer’s disease. J Neurochem 100(2):490–502
Badinter F, Weinreb O, Bar-Am O, Amit T, Youdim MB (2014) Neuroprotective effects of the major metabolite of the anti-Parkinsonian drug rasagiline, 1-(R)-aminoindan in animal model of aging. J Mol Neurosci 53(Suppl 1):S10
Bar-Am O, Amit M.B.H Y T (2004) Contrasting neuroprotective and neurotoxic actions of respective metabolites of anti-Parkinson drugs rasagiline and selegiline. Neurosci Lett 355(3):169–172
Bar-Am O, Weinreb O, Amit T, Youdim MB (2005) Regulation of Bcl-2 family proteins, neurotrophic factors, and APP processing in the neurorescue activity of propargylamine. FASEB J 19(13):1899–1901
Bar-Am O, Weinreb O, Amit T, Youdim MB (2009) The novel cholinesterase-monoamine oxidase inhibitor and antioxidant, ladostigil, confers neuroprotection in neuroblastoma cells and aged rats. J Mol Neurosci 37(2):135–145
Bar-Am O, Amit T, Kupershmidt L, Aluf Y, Mechlovich D, Kabha H, Danovitch L, Zurawski VR, Youdim MB, Weinreb O (2015) Neuroprotective and neurorestorative activities of a novel iron chelator-brain selective monoamine oxidase-A/monoamine oxidase-B inhibitor in animal models of Parkinson’s disease and aging. Neurobiol Aging 36(3):1529–1542
Barber AJ, Yu PH, Boulton AA (1993) Chronic effects of monoamine oxidase-B inhibitors on the behaviour of aged mice. Life Sci 53(9):739–747
Benedetti MS, Keane PE (1980) Differential changes in monoamine oxidase A and B activity in the aging rat brain. J Neurochem 35(5):1026–1032
Bernardi P, Petronilli V, Di Lisa F, Forte M (2001) A mitochondrial perspective on cell death. Trends Biochem Sci 26(2):112–117
Berr C, Balansard B, Arnaud J, Roussel AM, Alperovitch A (2000) Cognitive decline is associated with systemic oxidative stress: the EVA study. Etude du Vieillissement Arteriel. J Am Geriatr Soc 48(10):1285–1291
Bickford PC, Adams CE, Boyson SJ, Curella P, Gerhardt GA, Heron C, Ivy GO, Lin AM, Murphy MP, Poth K, Wallace DR, Young DA, Zahniser NR, Rose GM (1997) Long-term treatment of male F344 rats with deprenyl: assessment of effects on longevity, behavior, and brain function. Neurobiol Aging 18(3):309–318
Block ML (2008) NADPH oxidase as a therapeutic target in Alzheimer’s disease. BMC Neurosci 9(Suppl 2):S1–S8
Bondareff W (2013) Age-related changes in brain extracellular space affect processing of amyloid-beta peptides in Alzheimer’s disease. J Alzheimers Dis 35(1):1–6
Bongarzone ER, Pasquini JM, Soto EF (1995) Oxidative damage to proteins and lipids of CNS myelin produced by in vitro generated reactive oxygen species. J Neurosci Res 41(2):213–221
Boveris A, Navarro A (2008) Brain mitochondrial dysfunction in aging. IUBMB Life 60(5):308–314
Buccafusco JJ, Terry AV Jr, Goren T, Blaugrun E (2003) Potential cognitive actions of (n-propargly-(3r)-aminoindan-5-yl)-ethyl, methyl carbamate (tv3326), a novel neuroprotective agent, as assessed in old rhesus monkeys in their performance of versions of a delayed matching task. Neuroscience 119(3):669–678
Cameron HA, Kaliszewski CK, Greer CA (1991) Organization of mitochondria in olfactory bulb granule cell dendritic spines. Synapse 8(2):107–118
Carageorgiou H, Zarros A, Tsakiris S (2003) Selegiline long-term effects on brain acetylcholinesterase, (Na+, K+)-ATPase activities, antioxidant status and learning performance of aged rats. Pharmacol Res 48(3):245–251
Caspersen C, Wang N, Yao J, Sosunov A, Chen X, Lustbader JW, Xu HW, Stern D, McKhann G, Yan SD (2005) Mitochondrial Abeta: a potential focal point for neuronal metabolic dysfunction in Alzheimer’s disease. FASEB J 19(14):2040–2041
Chada SR, Hollenbeck PJ (2004) Nerve growth factor signaling regulates motility and docking of axonal mitochondria. Curr Biol 14(14):1272–1276
Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T, Korsmeyer SJ (2001) BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell 8(3):705–711
Cheng A, Hou Y, Mattson MP (2010) Mitochondria and neuroplasticity. ASN Neuro 2(5):e00045
Cohen G (2000) Oxidative stress, mitochondrial respiration, and Parkinson’s disease. Ann N Y Acad Sci 899:112–120
Cuello AC, Allard S, Ferretti MT (2012) Evidence for the accumulation of Abeta immunoreactive material in the human brain and in transgenic animal models. Life Sci 91(23–24):1141–1147
de Lima MN, Laranja DC, Caldana F, Bromberg E, Roesler R, Schroder N (2005) Reversal of age-related deficits in object recognition memory in rats with l-deprenyl. Exp Gerontol 40(6):506–511
Deng G, Su JH, Ivins KJ, Van Houten B, Cotman CW (1999) Bcl-2 facilitates recovery from DNA damage after oxidative stress. Exp Neurol 159(1):309–318
Devi L, Prabhu BM, Galati DF, Avadhani NG, Anandatheerthavarada HK (2006) Accumulation of amyloid precursor protein in the mitochondrial import channels of human Alzheimer’s disease brain is associated with mitochondrial dysfunction. J Neurosci 26(35):9057–9068
Droge W, Schipper HM (2007) Oxidative stress and aberrant signaling in aging and cognitive decline. Aging Cell 6(3):361–370
Finberg JP (2010) Pharmacology of rasagiline, a new MAO-B inhibitor drug for the treatment of Parkinson’s disease with neuroprotective potential. Rambam Maimonides Med J 1(1):e0003
Finberg JP (2014) Update on the pharmacology of selective inhibitors of MAO-A and MAO-B: focus on modulation of CNS monoamine neurotransmitter release. Pharmacol Ther 143:133–152
Finkel T (2011) Signal transduction by reactive oxygen species. J Cell Biol 194:7–15
Fowler CJ, Wiberg A, Oreland L, Marcusson J, Winblad B (1980) The effect of age on the activity and molecular properties of human brain monoamine oxidase. J Neural Transm 49(1–2):1–20
Frade JM, Barde YA (1998) Nerve growth factor: two receptors, multiple functions. BioEssays 20(2):137–145
Fridovich I (1975) Superoxide dismutases. Annu Rev Biochem 44:147–159
Friedman WJ, Greene LA (1999) Neurotrophin signaling via Trks and p75. Exp Cell Res 253(1):131–142
Gallagher IM, Clow A, Glover V (1998) Long-term administration of (–)-deprenyl increases mortality in male Wistar rats. J Neural Transm Suppl 52:315–320
Galluzzi L, Kroemer G (2007) Mitochondrial apoptosis without VDAC. Nat Cell Biol 9:487–489
Galva MD, Bondiolotti GP, Olasmaa M, Picotti GB (1995) Effect of aging on lazabemide binding, monoamine oxidase activity and monoamine metabolites in human frontal cortex. J Neural Transm Gen Sect 101(1–3):83–94
Gemma C, Mesches MH, Sepesi B, Choo K, Holmes DB, Bickford PC (2002) Diets enriched in foods with high antioxidant activity reverse age-induced decreases in cerebellar beta-adrenergic function and increases in proinflammatory cytokines. J Neurosci 22(14):6114–6120
Halbach OB (2010) Involvement of BDNF in age-dependent alterations in the hippocampus. Front Aging Neurosci 2
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 18(9):685–716
Halliwell B, Gutteridge JM (1986) Oxygen free radicals and iron in relation to biology and medicine: some problems and concepts. Arch Biochem Biophys 246(2):501–514
Hansson Petersen CA, Alikhani N, Behbahani H, Wiehager B, Pavlov PF, Alafuzoff I, Leinonen V, Ito A, Winblad B, Glaser E, Ankarcrona M (2008) The amyloid beta-peptide is imported into mitochondria via the TOM import machinery and localized to mitochondrial cristae. Proc Natl Acad Sci USA 105(35):13145–13150
Harman D (1972) Free radical theory of aging: dietary implications. Am J Clin Nutr 25(8):839–843
Harman D (1981) The aging process. Proc Natl Acad Sci USA 78(11):7124–7128
Harman D (1992) Free radical theory of aging. Mutat Res 275(3–6):257–266
Harper ME, Bevilacqua L, Hagopian K, Weindruch R, Ramsey JJ (2004) Ageing, oxidative stress, and mitochondrial uncoupling. Acta Physiol Scand 182(4):321–331
Irwin I, Finnegan KT, Delanney LE, Di Monte D, Langston JW (1992) The relationships between aging, monoamine oxidase, striatal dopamine and the effects of MPTP in C57BL/6 mice: a critical reassessment. Brain Res 572(1–2):224–231
Junn E, Mouradian MM (2001) Apoptotic signaling in dopamine-induced cell death: the role of oxidative stress, p38 mitogen-activated protein kinase, cytochrome c and caspases. J Neurochem 78(2):374–383
Kaur J, Singh S, Sharma D, Singh R (2003) Neurostimulatory and antioxidative effects of l-deprenyl in aged rat brain regions. Biogerontology 4(2):105–111
Kelso GF, Porteous CM, Hughes G, Ledgerwood EC, Gane AM, Smith RA, Murphy MP (2002) Prevention of mitochondrial oxidative damage using targeted antioxidants. Ann N Y Acad Sci 959:263–274
Kiray M, Bagriyanik HA, Pekcetin C, Ergur BU, Uysal N, Ozyurt D, Buldan Z (2006) Deprenyl and the relationship between its effects on spatial memory, oxidant stress and hippocampal neurons in aged male rats. Physiol Res 55(2):205–212
Kitani K, Minami C, Isobe K, Maehara K, Kanai S, Ivy GO, Carrillo MC (2002) Why (–)deprenyl prolongs survivals of experimental animals: increase of anti-oxidant enzymes in brain and other body tissues as well as mobilization of various humoral factors may lead to systemic anti-aging effects. Mech Ageing Dev 123(8):1087–1100
Kitani K, Kanai S, Miyasaka K, Carrillo MC, Ivy GO (2006) The necessity of having a proper dose of (–)deprenyl (D) to prolong the life spans of rats explains discrepancies among different studies in the past. Ann N Y Acad Sci 1067:375–382
Kornhuber J, Konradi C, Mack-Burkhardt F, Riederer P, Heinsen H, Beckmann H (1989) Ontogenesis of monoamine oxidase-A and -B in the human brain frontal cortex. Brain Res 499(1):81–86
Kumar H, Lim HW, More SV, Kim BW, Koppula S, Kim IS, Choi DK (2012) The role of free radicals in the aging brain and Parkinson’s disease: convergence and parallelism. Int J Mol Sci 13(8):10478–10504
Kupershmidt L, Amit T, Bar-Am O, Youdim MB, Weinreb O (2012a) The novel multi-target iron chelating-radical scavenging compound M30 possesses beneficial effects on major hallmarks of Alzheimer’s disease. Antioxid Redox Signal 17:860–877
Kupershmidt L, Amit T, Bar-Am O, Youdim MB, Weinreb O (2012b) Neuroprotection by the multitarget iron chelator M30 on age-related alterations in mice. Mech Ageing Dev 133(5):267–274
Linnane AW, Marzuki S, Ozawa T, Tanaka M (1989) Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet 1(8639):642–645
Lopez-Armada MJ, Riveiro-Naveira RR, Vaamonde-Garcia C, Valcarcel-Ares MN (2013) Mitochondrial dysfunction and the inflammatory response. Mitochondrion 13(2):106–118
Lu T, Pan Y, Kao SY, Li C, Kohane I, Chan J, Yankner BA (2004) Gene regulation and DNA damage in the ageing human brain. Nature 429(6994):883–891
Manczak M, Anekonda TS, Henson E, Park BS, Quinn J, Reddy PH (2006) Mitochondria are a direct site of A beta accumulation in Alzheimer’s disease neurons: implications for free radical generation and oxidative damage in disease progression. Hum Mol Genet 15(9):1437–1449
Marchi S, Giorgi C, Suski JM, Agnoletto C, Bononi A, Bonora M, De Marchi E, Missiroli S, Patergnani S, Poletti F et al (2012) Mitochondria-ros crosstalk in the control of cell death and aging. J Signal Transduct 2012:1–12
Markham A, Cameron I, Franklin P, Spedding M (2004) BDNF increases rat brain mitochondrial respiratory coupling at complex I, but not complex II. Eur J Neurosci 20(5):1189–1196
Markham A, Cameron I, Bains R, Franklin P, Kiss JP, Schwendimann L, Gressens P, Spedding M (2012) Brain-derived neurotrophic factor-mediated effects on mitochondrial respiratory coupling and neuroprotection share the same molecular signalling pathways. Eur J Neurosci 35(3):366–374
Martinez M, Hernandez AI, Martinez N, Ferrandiz ML (1996) Age-related increase in oxidized proteins in mouse synaptic mitochondria. Brain Res 731(1–2):246–248
Maruyama W, Akao Y, Youdim MB, Davis BA, Naoi M (2001) Transfection-enforced Bcl-2 overexpression and an anti-Parkinson drug, rasagiline, prevent nuclear accumulation of glyceraldehyde-3-phosphate dehydrogenase induced by an endogenous dopaminergic neurotoxin, N-methyl(R)salsolinol. J Neurochem 78(4):727–735
Maruyama W, Akao Y, Carrillo MC, Kitani K, Youdium MB, Naoi M (2002) Neuroprotection by propargylamines in Parkinson’s disease: suppression of apoptosis and induction of prosurvival genes. Neurotoxicol Teratol 24(5):675–682
Maruyama W, Weinstock M, Youdim MB, Nagai M, Naoi M (2003) Anti-apoptotic action of anti-Alzheimer drug, TV3326 [(N-propargyl)-(3R)-aminoindan-5-yl]-ethyl methyl carbamate, a novel cholinesterase- monoamine oxidase inhibitor. Neurosci Lett 341(3):233–236
Mechlovich D, Amit T, Mandel SA, Bar-Am O, Bloch K, Vardi P, Youdim MB (2010) The novel multifunctional, iron-chelating drugs M30 and HLA20 protect pancreatic beta-cell lines from oxidative stress damage. J Pharmacol Exp Ther 333(3):874–882
Naoi M, Maruyama W (2009) Functional mechanism of neuroprotection by inhibitors of type B monoamine oxidase in Parkinson’s disease. Expert Rev Neurother 9(8):1233–1250
Naoi M, Maruyama W, Yi H, Akao Y, Yamaoka Y, Shamoto-Nagai M (2007) Neuroprotection by propargylamines in Parkinson’s disease: intracellular mechanism underlying the anti-apoptotic function and search for clinical markers. J Neural Transm Suppl 72:121–131
Naoi M, Maruyama W, Inaba-Hasegawa K (2012) Type A and B monoamine oxidase in age-related neurodegenerative disorders: their distinct roles in neuronal death and survival. Curr Top Med Chem 12(20):2177–2188
Naoi M, Maruyama W, Yi H (2013) Rasagiline prevents apoptosis induced by PK11195, a ligand of the outer membrane translocator protein (18 kDa), in SH-SY5Y cells through suppression of cytochrome c release from mitochondria. J Neural Transm 120(11):1539–1551
Olanow CW (1990) Oxidation reactions in Parkinson’s disease. Neurology 40(10 Suppl 3):32–37 (discussion 37–39)
Panarsky R, Luques L, Weinstock M (2012) Anti-inflammatory effects of ladostigil and its metabolites in aged rat brain and in microglial cells. J Neuroimmune Pharmacol 7(2):488–498
Pollack M, Phaneuf S, Dirks A, Leeuwenburgh C (2002) The role of apoptosis in the normal aging brain, skeletal muscle, and heart. Ann N Y Acad Sci 959:93–107
Poon HF, Calabrese V, Calvani M, Butterfield DA (2006) Proteomics analyses of specific protein oxidation and protein expression in aged rat brain and its modulation by l-acetylcarnitine: insights into the mechanisms of action of this proposed therapeutic agent for CNS disorders associated with oxidative stress. Antioxid Redox Signal 8(3–4):381–394
Popov V, Medvedev NI, Davies HA, Stewart MG (2005) Mitochondria form a filamentous reticular network in hippocampal dendrites but are present as discrete bodies in axons: a three-dimensional ultrastructural study. J Comp Neurol 492(1):50–65
Reddy PH, Reddy TP (2011) Mitochondria as a therapeutic target for aging and neurodegenerative diseases. Curr Alzheimer Res 8(4):393–409
Reddy PH, Manczak M, Mao P, Calkins MJ, Reddy AP, Shirendeb U (2010) Amyloid-beta and mitochondria in aging and Alzheimer’s disease: implications for synaptic damage and cognitive decline. J Alzheimers Dis 20(Suppl 2):S499–S512
Reiter RJ (1995) Oxidative processes and antioxidative defense mechanisms in the aging brain. FASEB J 9(7):526–533
Rinaldi P, Polidori MC, Metastasio A, Mariani E, Mattioli P, Cherubini A, Catani M, Cecchetti R, Senin U, Mecocci P (2003) Plasma antioxidants are similarly depleted in mild cognitive impairment and in Alzheimer’s disease. Neurobiol Aging 24(7):915–919
Schulze ET, Geary EK, Susmaras TM, Paliga JT, Maki PM, Little DM (2011) Anatomical correlates of age-related working memory declines. J Aging Res 2011:606871
Segal RA, Greenberg ME (1996) Intracellular signaling pathways activated by neurotrophic factors. Annu Rev Neurosci 19:463–489
Sen T, Sen N, Tripathi G, Chatterjee U, Chakrabarti S (2006) Lipid peroxidation associated cardiolipin loss and membrane depolarization in rat brain mitochondria. Neurochem Int 49(1):20–27
Shoham S, Bejar C, Kovalev E, Schorer-Apelbaum D, Weinstock M (2007) Ladostigil prevents gliosis, oxidative-nitrative stress and memory deficits induced by intracerebroventricular injection of streptozotocin in rats. Neuropharmacology 52(3):836–843
Siegel GJ, Chauhan NB (2000) Neurotrophic factors in Alzheimer’s and Parkinson’s disease brain. Brain Res Brain Res Rev 33(2–3):199–227
Singh R, Mishra M, Singh S, Sharma D (2012) Effect of l-deprenyl treatment on electrical activity, Na+, K+ ATPase, and protein kinase C activities in hippocampal subfields (CA1 and CA3) of aged rat brain. Indian J Exp Biol 50(2):101–109
Smith RA, Murphy MP (2011) Mitochondria-targeted antioxidants as therapies. Discov Med 11(57):106–114
Smith RA, Porteous CM, Coulter CV, Murphy MP (1999) Selective targeting of an antioxidant to mitochondria. Eur J Biochem 263(3):709–716
Sofroniew MV, Howe CL, Mobley WC (2001) Nerve growth factor signaling, neuroprotection, and neural repair. Annu Rev Neurosci 24:1217–1281
Soong NW, Hinton DR, Cortopassi G, Arnheim N (1992) Mosaicism for a specific somatic mitochondrial DNA mutation in adult human brain. Nat Genet 2(4):318–323
Stauch KL, Purnell PR, Fox HS (2014) Aging synaptic mitochondria exhibit dynamic proteomic changes while maintaining bioenergetic function. Aging (Albany NY) 6(4):320–334
Strolin Benedetti M, Dostert P (1989) Monoamine oxidase, brain ageing and degenerative diseases. Biochem Pharmacol 38(4):555–561
Su B, Ji YS, Sun XL, Liu XH, Chen ZY (2014) Brain-derived neurotrophic factor (BDNF)-induced mitochondrial motility arrest and presynaptic docking contribute to BDNF-enhanced synaptic transmission. J Biol Chem 289(3):1213–1226
Subramanian MV, James TJ (2010) Age-related protective effect of deprenyl on changes in the levels of diagnostic marker enzymes and antioxidant defense enzymes activities in cerebellar tissue in Wistar rats. Cell Stress Chaperones 15(5):743–751
Swerdlow RH (2011) Brain aging, Alzheimer’s disease, and mitochondria. Biochim Biophys Acta 1812(12):1630–1639
Toman J, Fiskum G (2011) Influence of aging on membrane permeability transition in brain mitochondria. J Bioenerg Biomembr 43(1):3–10
Vander Heiden MG, Thompson CB (1999) Bcl-2 proteins: regulators of apoptosis or of mitochondrial homeostasis? Nat Cell Biol 1(8):E209–E216
Wang X, Su B, Siedlak SL, Moreira PI, Fujioka H, Wang Y, Casadesus G, Zhu X (2008) Amyloid-beta overproduction causes abnormal mitochondrial dynamics via differential modulation of mitochondrial fission/fusion proteins. Proc Natl Acad Sci USA 105(49):19318–19323
Weinreb O, Bar-Am O, Amit T, Chillag-Talmor O, Youdim MBH (2004) Neuroprotection via pro-survival protein kinase C isoforms associated with Bcl-2 family members. FASEB J 18(12):1471–1473
Weinreb O, Amit T, Bar-Am O, Youdim MB (2007a) Induction of neurotrophic factors GDNF and BDNF associated with the mechanism of neurorescue action of rasagiline and ladostigil: new insights and implications for therapy. Ann N Y Acad Sci 1122:155–168
Weinreb O, Drigues N, Sagi Y, Reznick AZ, Amit T, Youdim MB (2007b) The application of proteomics and genomics to the study of age-related neurodegeneration and neuroprotection. Antioxid Redox Signal 9(2):169–179
Weinreb O, Bar-Am O, Amit T, Drigues N, Sagi Y, Youdim MB (2008) The neuroprotective effect of ladostigil against hydrogen peroxide-mediated cytotoxicity. Chem Biol Interact 175(1–3):318–326
Weinreb O, Amit T, Bar-Am O, Youdim MB (2010) Rasagiline: a novel anti-Parkinsonian monoamine oxidase-B inhibitor with neuroprotective activity. Prog Neurobiol 92(3):330–344
Weinreb O, Amit T, Bar-Am O, Youdim MB (2012) Ladostigil: a novel multimodal neuroprotective drug with cholinesterase and brain-selective monoamine oxidase inhibitory activities for Alzheimer’s disease treatment. Curr Drug Targets 13(4):483–494
Weinstock M, Bejar C, Wang RH, Poltyrev T, Gross A, Finberg J, Youdim MBH (2000) TV3326, a novel neuroprotective drug with cholinesterase and monoamine oxidase inhibitory activities for the treatment of Alzheimer’s disease. J Neural Transm [Suppl] 60:S157–S170
Weinstock M, Luques L, Poltyrev T, Bejar C, Shoham S (2011) Ladostigil prevents age-related glial activation and spatial memory deficits in rats. Neurobiol Aging 32(6):1069–1078
Weinstock M, Bejar C, Schorer-Apelbaum D, Panarsky R, Luques L, Shoham S (2013) Dose-dependent effects of ladostigil on microglial activation and cognition in aged rats. J Neuroimmune Pharmacol 8(1):345–355
Xiao XQ, Lee NT, Carlier PR, Pang Y, Han YF (2000) Bis(7)-tacrine, a promising anti-Alzheimer’s agent, reduces hydrogen peroxide-induced injury in rat pheochromocytoma cells: comparison with tacrine. Neurosci Lett 290(3):197–200
Yankner BA, Lu T, Loerch P (2008) The aging brain. Annu Rev Pathol 3:41–66
Yogev-Falach M, Amit T, Bar-Am O, Sagi Y, Weinstock M, Youdim MBH (2002) The involvement of mitogen-activated protein (MAP) kinase in the regulation of amyloid precursor protein processing by novel cholinesterase inhibitors derived from rasagiline. FASEB J 16(12):1674–1676
Youdim MBH, Riederer PF (2004) A review of the mechanisms and role of monoamine oxidase inhibitors in Parkinson’s disease. Neurology 63(7 Suppl 1):S32–S35
Youdim MB, Bar Am O, Yogev-Falach M, Weinreb O, Maruyama W, Naoi M, Amit T (2005) Rasagiline: neurodegeneration, neuroprotection, and mitochondrial permeability transition. J Neurosci Res 79(1–2):172–179
Zecca L, Youdim MBH, Riederer P, Connor JR, Crichton RR (2004) Iron, brain ageing and neurodegenerative disorders. Nat Rev Neurosci 5(11):863–873
Zhang Y, Marcillat O, Giulivi C, Ernster L, Davies KJ (1990) The oxidative inactivation of mitochondrial electron transport chain components and ATPase. J Biol Chem 265(27):16330–16336
Zheng H, Weiner LM, Bar-Am O, Epsztejn S, Cabantchik ZI, Warshawsky A, Youdim MB, Fridkin M (2005) Design, synthesis, and evaluation of novel bifunctional iron-chelators as potential agents for neuroprotection in Alzheimer’s, Parkinson’s, and other neurodegenerative diseases. Bioorg Med Chem 13(3):773–783
Acknowledgments
We gratefully acknowledge the support of the Technion-Research and Development and Rappaport Family Research Institute, Technion-Israel Institute of Technology (Haifa, Israel).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bar-Am, O., Amit, T., Youdim, M.B. et al. Neuroprotective and neurorestorative potential of propargylamine derivatives in ageing: focus on mitochondrial targets. J Neural Transm 123, 125–135 (2016). https://doi.org/10.1007/s00702-015-1395-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-015-1395-3