Summary
Background
Cytomegalovirus (CMV) is endemic worldwide, with marked differences in the seroprevalence rates between countries. The aim of this study was to analyze the seroprevalence of CMV infections in Croatia.
Methods
During a 3-year period (2013–2015) 2438 consecutive serum samples collected from Croatian residents were tested for the presence of CMV IgM and IgG antibodies using enzyme-linked immunoassay. The IgM/IgG positive samples were further tested for IgG avidity.
Results
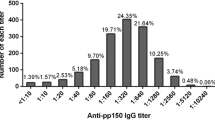
The overall seroprevalence rates for CMV IgG and IgM antibodies were 74.4 % and 4.3 %, respectively. The IgG seroprevalence showed significant differences between population groups: children/adolescents 54.6 %, general adult population 77.2 %, hemodialysis patients 91.4 % (p < 0.001). Seropositivity of CMV was strongly age-dependent with prevalences ranging from 53.0 % in children less than 10 years old to 93.8 % in persons above 60 years (p < 0.001). There was no difference in the prevalence rate between women with normal pregnancy and women with poor obstetric history. Gender and place of residence were not associated with CMV seropositivity. Using IgG avidity, current/recent primary CMV infection was confirmed by a low/borderline avidity index (AI) in 46.7 % participants, while in 53.3 % a high AI indicated CMV reactivation or reinfection. Primary infections were detected mainly in children and adolescents (83.2 % and 70.5 %, respectively), while reactivation/reinfection was common in persons older than 40 (77.0–100 %). Reactivation/reinfection was most commonly detected in hemodialysis patients (92.3 %). Logistic regression showed that older age and being on hemodialysis were significant predictors of CMV seropositivity.
Conclusion
Infections with CMV are widespread in the Croatian population. Older age and being on hemodialysis appear to be the main risk factors for CMV infection.



Similar content being viewed by others
References
Adjei AA, Armah HB, Gbagbo F, Boamah I, Adu-Gyamfi C, Asare I. Seroprevalence of HHV-8, CMV, and EBV among the general population in Ghana, West Africa. BMC Infect Dis. 2008;8:111.
Cannon MJ, Schmid DS, Hyde TB. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev Med Virol. 2010;20:202–13.
Hodinka RL. Human cytomegalovirus. In: Versalovic J, Carroll KC, Funke G, Jorgensen JH, Landry ML, Warnick DW, editors. Manual of clinical microbiology, 10th edn. Washington, DC: ASM Press; 2011. pp. 1558–74.
Lancini D, Faddy HM, Flower R, Hogan C. Cytomegalovirus disease in immunocompetent adults. Med J Aust. 2014;201:578–80.
Karakozis S, Gongora E, Caceres M, Brun E, Cook JW. Life-threatening cytomegalovirus colitis in the immunocompetent patient: report of a case and review of the literature. Dis Colon Rectum. 2001;44:1716–20.
Rafailidis PI, Mourtzoukou EG, Varbobitis IC, Falagas ME. Severe cytomegalovirus infection in apparently immunocompetent patients: a systematic review. Virol J. 2008;5:47.
Orasch C, Conen A. Severe primary cytomegalovirus infection in the immunocompetent adult patient: a case series. Scand J Infect Dis. 2012;44:987–91.
Cavdar C, Celtik A, Saglam F, et al. Cytomegalovirus disease in renal transplant recipients: a single-center experience. Ren Fail. 2008;30:503–6.
Beader N, Kalenić S, Labar B. Diagnostic approach and therapy for cytomegalovirus (CMV) infection following allogeneic stem cell transplantation. Lijec Vjesn. 2011;133:389–96.
Varga M, Görög D, Kári D, et al. Cytomegalovirus seroprevalence among solid organ donors in Hungary: correlations with age, gender, and blood group. Transplant Proc. 2011;43:1233–5.
Wang C, Zhang X, Bialek S, Cannon MJ. Attribution of congenital cytomegalovirus infection to primary versus non-primary maternal infection. Clin Infect Dis. 2011;52:e11–e13.
Aarnisalo J, Ilonen J, Vainionpää R, Volanen I, Kaitosaari T, Simell O. Development of antibodies against cytomegalovirus, varicella-zoster virus and herpes simplex virus in Finland during the first eight years of life: a prospective study. Scand J Infect Dis. 2003;35:750–3.
Ataman S, Colak D, Günseren F, et al. Investigation of cytomegalovirus seroepidemiology in Antalya with a population-based cross-sectional study and review of related data in Turkey. Mikrobiyol Bul. 2007;41:545–55.
Lopo S, Vinagre E, Palminha P, Paixao MT, Nogueira P, Freitas MG. Seroprevalence to cytomegalovirus in the Portuguese population, 2002–2003. Euro Surveill. 2011;16(25):pii: 19896.
Zebrun AB, Kuliasheva LB, Ermolenko KD, Zakrevskaia AV. Spread of herpesvirus infections in children and adults in St. Petersburg according to seroepidemiologic study data. Zh Mikrobiol Epidemiol Immunobiol. 2013;6:30–6.
Dolgikh TI, Dalmatov VV, Zaparii NS, Kadtsyna TV. Cytomegalovirus infection in Omsk region. Zh Mikrobiol Epidemiol Immunobiol. 2008;3:85–7.
Lübeck PR, Doerr HW, Rabenau HF. Epidemiology of human cytomegalovirus (HCMV) in an urban region of Germany: what has changed? Med Microbiol Immunol. 2010;199:53–60.
Lepage N, Leroyer A, Cherot-Kornobis N, Lartigau I, Miczek S, Sobaszek A. Cytomegalovirus seroprevalence in exposed and unexposed populations of hospital employees. Eur J Clin Microbiol Infect Dis. 2011;30:65–70.
Korndewal MJ, Mollema L, Tcherniaeva I, et al. Cytomegalovirus infection in the Netherlands: seroprevalence, risk factors, and implications. J Clin Virol. 2015;63:53–8.
Odland JØ, Sergejeva IV, Ivaneev MD, Jensen IP, Stray-Pedersen B. Seropositivity of cytomegalovirus, parvovirus and rubella in pregnant women and recurrent aborters in Leningrad County, Russia. Acta Obstet Gynecol Scand. 2001;80:1025–9.
Leuridan E, Ieven M, Hens N, Damme P van. High susceptibility to cytomegalovirus infection of pregnant women in Flanders, Belgium. Facts Views Vis Obgyn. 2012;4:76–81.
N’Diaye DS, Yazdanpanah Y, Krivine A, et al. Predictive factors of cytomegalovirus seropositivity among pregnant women in Paris, France. PLOS ONE. 2014;9(2):e89857.
Odland ML, Strand KM, Nordbø SA, Forsmo S, Austgulen R, Iversen AC. Changing patterns of cytomegalovirus seroprevalence among pregnant women in Norway between 1995 and 2009 examined in the Norwegian Mother and Child Cohort Study and two cohorts from Sor-Trondelag County: a cross-sectional study. BMJ Open. 2013;3(9):e003066.
Aynioglu A, Aynioglu O, Altunok ES. Seroprevalence of toxoplasma gondii, rubella and cytomegalovirus among pregnant females in north-western Turkey. Acta Clin Belg. 2015; doi:10.1179/2295333715Y.0000000021.
Trkulic M, Jovanovic D, Ostojic G, Kovacevic Z, Taseski J. Cytomegalovirus infection in patients with kidney diseases. Vojnosanit Pregl. 2000;57:63–7.
Ocak S, Duran N, Eskiocak AF. Seroprevalence of cytomegalovirus antibodies in hemodialysis patients. Turk J Med Sci. 2006;36:155–8.
Betjes MG, Litjens NH, Zietse R. Seropositivity for cytomegalovirus in patients with end-stage renal disease is strongly associated with atherosclerotic disease. Nephrol Dial Transplant. 2007;22:3298–303.
Vilibic-Cavlek T, Ljubin-Sternak S, Ban M, Kolaric B, Sviben M, Mlinaric-Galinovic G. Seroprevalence of TORCH infections in women of childbearing age in Croatia. J Matern Fet Neonatal Med. 2011;24:280–3.
Vilibic-Cavlek T, Kolaric B, Ljubin-Sternak S, Kos M, Kaic B, Mlinaric-Galinovic G. Prevalence and dynamics of cytomegalovirus infection among patients undergoing chronic hemodialysis. Indian J Nephrol. 2015;25:95–8.
Ory Manchón F de, Sanz Moreno JC, Castañeda López R, Ramírez Fernández R, León Rega P, Pachón del Amo I. Cytomegalovirus seroepidemiology in the community of Madrid. Rev Esp Salud Publica. 2001;75:55–62.
Francisse S, Revelard P, de Maertelaer V, Strebelle E, Englert Y, Liesnard C. Human cytomegalovirus seroprevalence and risk of seroconversion in a fertility clinic population. Obstet Gynecol. 2009;114:285–91.
Enders G, Daiminger A, Lindemann L, et al. Cytomegalovirus (CMV) seroprevalence in pregnant women, bone marrow donors and adolescents in Germany, 1996–2010. Med Microbiol Immunol. 2012;201:303–9.
Rijckevorsel GG van, Bovée LP, Damen M, Sonder GJ, Schim van der Loeff MF, Hoek A van den. Increased seroprevalence of IgG-class antibodies against cytomegalovirus, parvovirus B19, and varicella-zoster virus in women working in child day care. BMC Public Health. 2012;12:475.
Sibrowski W, Kühnl P, Kalmar G, Albert S, Böhm BO, Doerr HW. Cytomegalovirus diagnosis in blood donors and risk patients. Beitr Infusionsther. 1990;26:37–9.
Pembrey L, Raynor P, Griffiths P, Chaytor S, Wright J, Hall AJ. Seroprevalence of cytomegalovirus, Epstein Barr virus and varicella zoster virus among pregnant women in Bradford: a cohort study. PLOS ONE. 2013;8(11):e81881.
Alanen A, Kahala K, Vahlberg T, Koskela P, Vainionpää R. Seroprevalence, incidence of prenatal infections and reliability of maternal history of varicella zoster virus,cytomegalovirus, herpes simplex virus and parvovirus B19 infection in South-Western Finland. BJOG. 2005;112:50–6.
Wujcicka W, Gaj Z, Wilczyński J, Sobala W, Spiewak E, Nowakowska D. Impact of socioeconomic risk factors on the seroprevalence of cytomegalovirus infections in a cohort of pregnant Polish women between 2010 and 2011. Eur J Clin Microbiol Infect Dis. 2014;33:1951–8.
Uyar Y, Balci A, Akcali A, Cabar C. Prevalence of rubella and cytomegalovirus antibodies among pregnant women in Northern Turkey. New Microbiol. 2008;31:451–5.
Barlinn R, Vainio K, Samdal HH, Nordbø SA, Nøkleby H, Dudman SG. Susceptibility to cytomegalovirus, parvovirus B19 and age-dependent differences in levels of rubella antibodies among pregnant women. J Med Virol. 2014;86:820–6.
Knowles SJ, Grundy K, Cahill I, Cafferkey MT, Geary M. Low cytomegalovirus sero-prevalence in Irish pregnant women. Ir Med J. 2005;98:210–2.
Acknowledgements
This research was supported by the Ministry of Science, Education, and Sports of the Republic of Croatia, Grant No 005-0053443-3447 (to GMG). The authors thank Ljiljana Milasincic, Snjezana Artl, Vesna Cupic, Dubravka Landripet and Ana Pesut for technical assistance. Izabela Vrtar was project associate.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T. Vilibic-Cavlek, B. Kolaric, N. Beader, I. Vrtar, I. Tabain, and G. Mlinaric-Galinovic declare that they have no competing interests.
Ethical standards
All studies on humans described in this manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current revised form). Informed consent was obtained from all participants included in the study. In the case of underage patients consent was obtained from a parent of legal guardian.
Rights and permissions
About this article
Cite this article
Vilibic-Cavlek, T., Kolaric, B., Beader, N. et al. Seroepidemiology of cytomegalovirus infections in Croatia. Wien Klin Wochenschr 129, 129–135 (2017). https://doi.org/10.1007/s00508-016-1069-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-016-1069-7