Abstract
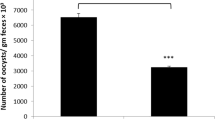
Coccidiosis in poultry is caused by protozoan parasites of the genus Eimeria, which is responsible for worldwide economic losses. The methanolic extract of Azadirachta indica (neem) leaves was used in vivo for its pharmacological, antioxidant, and anticoccidial properties. Four groups of mice were investigated. The first group was inoculated only with sterile saline and served as the control group. The second group was treated by oral gavage with neem extract (500 mg/kg) daily for 4 days. The third and fourth groups were infected with 103 sporulated oocysts of Eimeria papillata. The fourth group was also treated once daily with neem extract for 4 days. Paraffin sections from the jejunum as well as jejunal homogenate were prepared for the histopathological and biochemical investigations, respectively. The data showed that mice infected with E. papillata revealed an output of 6.5 × 105 ± 29,753 oocysts per gram feces on day 4 postinoculation. This output is significantly decreased to 2.7 × 105 ± 37,341 oocysts in neem-treated mice. Infection with E. papillata induced marked histopathological alterations in the jejunum in the form of inflammation, vacuolation of the epithelium, and destruction of some villi. Also, the neem extract greatly diminished body weight loss of infected mice. Moreover, the number of goblet cells stained with Alcian blue within the infected villi was significantly lowered (P ≤ 0.05). In addition, E. papillata enhanced lipid peroxidation and nitric oxide production in both serum and jejunum with concomitant reduction in glutathione. Neem induced marked improvements in all of the studied parameters as well as the histopathological features of the jejunum. Our study revealed that neem as a natural product has protective effects against E. papillata-induced coccidiosis.



Similar content being viewed by others
References
Abdel-Ghaffar F, Semmler M, Al-Rasheid KA, Strassen B, Fischer K, Aksu G, Klimpel S, Mehlhorn H (2010) The effects of different plant extracts on intestinal cestodes and on trematodes. Parasitol Res 108:979–984
Allen A, Hutton DA, Leonard AJ, Pearson JP, Sellers LA (1986) The role of mucus in the protection of the gastroduodenal mucosa. Scand J Gastroenterol Supp 125:71–78
Al-Quraishy S, Delic D, Sies H, Wunderlich F, Abdel-Baki AA, Dkhil MA (2011) Differential miRNA expression in the mouse jejunum during garlic treatment of Eimeria papillata infections. Parasitol Res 109:387–394
Anyaehie UB (2009) Medicinal properties of fractionated acetone/water neem (Azadirachta indica) leaf extract from Nigeria: a review. Nig J Physiol Sci 24:157–169
Balasenthil S, Arivazhagan S, Ramachandran CR, Ramachandran V, Nagini S (1999) Chemopreventive potential of neem (Azadirachta indica) on 7,12-dimethylbenz[a]anthracene (DMBA) induced hamster buccal pouch carcinogenesis. J Ethnopharmacol 67:189–195
Berkels R, Purol-Schnabel S, Roesen R (2004) Measurement of nitric oxide by reconversion of nitrate/nitrite to NO. Methods Mol Biol 279:1–8
Bhanwra S, Singh J, Khosla P (2000) Effect of Azadirachta indica (neem) leaf aqueous extract on paracetamol-induced liver damage in rats. Ind J Physiol Pharmacol 44:64–68
Cam Y, Atasever A, Eraslan G, Kibar M, Atalay O, Beyaz L, Inci A, Liman BC (2008) Eimeria stiedae: experimental infection in rabbits and the effect of treatment with toltrazuril and ivermectin. Exp Parasitol 119:164–172
Cheng H (1974) Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. II. Mucous cells. Am J Anat 141:481–501
Deplancke B, Gaskins HR (2001) Microbial modulation of innate defense: goblet cells and the intestinal mucus layer. Am J Clin Nutr 73:1131S–1141S
Dkhil MA, Abdel-Baki AS, Wunderlich F, Sies H, Al-Quraishy S (2011) Anticoccidial and antiinflammatory activity of garlic in murine Eimeria papillata infections. Vet Parasitol 175:66–72
Dommels YE, Butts CA, Zhu S, Davy M, Martell S, Hedderley D, Barnett MP, McNabb WC, Roy NC (2007) Characterization of intestinal inflammation and identification of related gene expression changes in mdr1a(−/−) mice. Genes Nutr 2:209–223
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Ezz-Din D, Gabry MS, Farrag AH, Abdel Moneim AE (2011) Physiological and histological impact of Azadirachta indica (neem) leaves extract in a rat model of cisplatin-induced hepato and nephrotoxicity. J Med Plants Res 5:5499–5506
Fukui K, Takatsu H, Koike T, Urano S (2011) Hydrogen peroxide induces neurite degeneration: prevention by tocotrienols. Free Rad Res 45:681–691
Georgieva N, Koinarski V, Gadjeva V (2006) Antioxidant status during the course of Eimeria tenella infection in broiler chickens. Vet J 172:488–492
Kaur G, Sarwar Alam M, Athar M (2004) Nimbidin suppresses functions of macrophages and neutrophils: relevance to its antiinflammatory mechanisms. Phytother Res 18:419–424
Kayano ACA, Lopes SCP, Bueno FG, Cabral EC, Souza-Neiras WC, Yamauchi LM, Foglio MA, Eberlin MN, Mello JCP, Costa FTM (2011) In vitro and in vivo assessment of the antimalarial activity of Caesalpinia pluviosa. Malaria J 10:112–122
Khalafalla RE, Daugschies A, Dyachenko V (2011) Cross-reactivity of anti-Eimeria tenella antibody fragments on merozoites and sporozoites of different chicken Eimeria species. Parasitol Res 108:745–749
MacDonald TT, Monteleone G (2005) Immunity, inflammation, and allergy in the gut. Science 307:1920–1925
Manikandan P, Letchoumy PV, Gopalakrishnan M, Nagini S (2008) Evaluation of Azadirachta indica leaf fractions for in vitro antioxidant potential and in vivo modulation of biomarkers of chemoprevention in the hamster buccal pouch carcinogenesis model. Food Chem Toxicol 46:2332–2343
Mehlhon H, Ortiman FG, Ha-berkorn A (1984) Effects of sym. Trizanones on developmental stages of Eimeria tenella, E. maxima and E. acervulina: a light and electron microscopy study. Parasitol Res 70:173–182
Mehlhorn H (ed) (2008) Encyclopedic reference of parasitology, vol 1, 3rd edn. Berlin, Springer
Metwaly MS, Dkhil MA, Al-Quraishy S (2012) The potential role of Phoenix dactylifera on Eimeria papillata-induced infection in mice. Parasitol Res 111:681–687
Morgan ED (2009) Azadirachtin, a scientific gold mine. Bioorg Med Chem 17:4096–4105
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analyt Biochem 95:351–358
Özdemir ÖM, Ergin H, Yenisey C, Türk NS, Şimşek NG (2010) Protective effects of clarithromycin in rats with hypoxia/reoxygenation-induced intestinal injury. J Pediatric Surg 45:2169–2174
Peer PA, Trivedi PC, Nigade PB, Ghaisas MM, Deshpande AD (2008) Cardioprotective effect of Azadirachta indica A. Juss. on isoprenaline induced myocardial infarction in rats. Int J Cardiol 126:123–126
Sato Y, Itagaki S, Oikawa S, Ogura J, Kobayashi M, Hirano T, Sugawara M, Iseki K (2011) Protective effect of soy isoflavone genistein on ischemia-reperfusion in the rat small intestine. Biol Pharmaceut Bullet 34:1448–1454
Schito ML, Barta JR, Chobotar B (1996) Comparison of four murine Eimeria species in immunocompetent and immunodeficient mice. J Parasitol 82:255–262
Schmahl G, Al-Rasheid KA, Abdel-Ghaffar F, Klimpel S, Mehlhorn H (2010) The efficacy of neem seed extracts (Tre-san, MiteStop) on a broad spectrum of pests and parasites. Parasitol Res 107:261–269
Toulah FH, Ismeel HA, Khan S (2010) Effect of treatment with neem (Azadirachta indica) compared with Baycox drug on the caecum of chicken experimentally infected with Eimeria tenella. J Egypt Soc Parasitol 40:93–106
Tsakiris S, Schulpis KH, Marinou M, Behrakis P (2004) Protective effect of L-cysteine and glutathione on the modulated suckling rat brain Na+, K+, -ATPase and Mg2+ -ATPase activities induced by the in vitro galactosaemia. Pharmacol Res 495:475–479
Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39:44–84
Yanpallewar SU, Sen S, Tapas S, Kumar M, Raju SS, Acharya SB (2003) Effect of Azadirachta indica on paracetamol-induced hepatic damage in albino rats. Phytomedicine 10:391–396
Yunus M, Horii Y, Makimura S, Smith AL (2005) Murine goblet cell hypoplasia during Eimeria pragensis infection is ameliorated by clindamycin treatment. J Vet Med Sci 67:311–315
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding the work through the research group project no. RGP-VPP-198.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

Figure S1
Unsporulated (a) and sporulated (b) oocyst of E. papillata. OM outer membrane, IM inner membrane, SPW sporoblast wall, SP sporoblast, Sc sporocyst, Scw sporocyst wall, RB residual body. (JPEG 33 kb)

Figure S2
Sections of mouse jejunum infected with E. papillata on day 4 p.i. a Noninfected jejunum with normal architecture. b Noninfected treated mouse jejunum with normal structure. c Infected jejunum with some pathological changes and developmental stages appearing in the inner epithelium. d Infected treated mouse with less parasites. Sections are stained with hematoxylin and eosin. Bar = 25 μm (JPEG 595 kb)

Figure S3
Changes in goblet cell numbers in mouse jejunum infected with E. papillata on day 4 p.i. a Noninfected jejunum, b noninfected treated mouse jejunum, c infected jejunum with decreased number of goblet cells, d infected treated mouse. Sections are stained with Alcian blue. Bar = 25 μm (JPEG 472 kb)
Rights and permissions
About this article
Cite this article
Dkhil, M.A., Al-Quraishy, S., Abdel Moneim, A.E. et al. Protective effect of Azadirachta indica extract against Eimeria papillata-induced coccidiosis. Parasitol Res 112, 101–106 (2013). https://doi.org/10.1007/s00436-012-3109-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-3109-1