Abstract
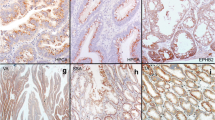
Serrated colorectal adenocarcinoma (SAC) is a morphologically distinct subtype of colorectal cancer (CRC), in which increased HIF-1α mRNA expression and HIF-1α protein stabilization are typical features. Here we aimed to further elucidate HIF-1α protein expression in serrated and non-serrated colorectal carcinomas (CRCs) and their precursor lesions and its association with vascular endothelial growth factor (VEGF) and microvascular density (MVD). HIF-1α and VEGF expressions were determined immunohistochemically in 134 serrated polyps (SPs), 104 non-serrated adenomas (NSAs), 81 SACs, and 74 matched conventional adenocarcinomas (CCs) and were correlated with morphology, clinicopathological features, and MVD. In premalignant lesions, both HIF-1α and VEGF were expressed in the vast majority of SPs and NSAs. In CRCs, HIF-1α protein was also present in 77.8 % of SACs, while only 20.3 % of CCs were HIF-1α proficient. MVD was significantly higher in SACs, but the serrated morphology was the only significant predictor of MVD in CRC in multivariate analyses. HIF-1α protein is often stabilized in well-vascularized SACs, suggesting hypoxia-independent stabilization of HIF-1α. Moreover, HIF-1α stabilization did not associate with oncogenic activation of BRAF or KRAS or Von Hippel-Lindau (VHL) mutation. Prevalent HIF-1α expression in SAC and its precursors support the importance of HIF-1α-mediated pathways for the serrated route of colorectal carcinogenesis.

Similar content being viewed by others
Abbreviations
- CC:
-
Conventional carcinoma
- CRC:
-
Colorectal cancer
- HIF:
-
Hypoxia-inducible transcription factor
- HP:
-
Hyperplastic polyp
- MVD:
-
Microvessel density
- NSA:
-
Non-serrated adenoma
- SP:
-
Serrated polyp
- SAC:
-
Serrated adenocarcinoma
- TSA:
-
Traditional serrated adenoma
References
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics. CA Cancer J Clin 64:9–29. doi:10.3322/caac.21208
Jass JR (2007) Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 50:113–130. doi:10.1111/j.1365-2559.2006.02549.x
Tuppurainen K, Makinen JM, Junttila O, et al. (2005) Morphology and microsatellite instability in sporadic serrated and non-serrated colorectal cancer. J Pathol 207:285–294. doi:10.1002/path.1850
Laiho P, Kokko A, Vanharanta S, et al. (2007) Serrated carcinomas form a subclass of colorectal cancer with distinct molecular basis. Oncogene 26:312–320. doi:10.1038/sj.onc.1209778
Mäkinen MJ (2007) Colorectal serrated adenocarcinoma. Histopathology 50:131–150. doi:10.1111/j.1365-2559.2006.02548.x
O’Brien MJ, Yang S, Mack C, et al. (2006) Comparison of microsatellite instability, CpG island methylation phenotype, BRAF and KRAS status in serrated polyps and traditional adenomas indicates separate pathways to distinct colorectal carcinoma end points. Am J Surg Pathol 30:1491–1501. doi:10.1097/01.pas.0000213313.36306.85
Stefanius K, Ylitalo L, Tuomisto A, et al. (2011) Frequent mutations of KRAS in addition to BRAF in colorectal serrated adenocarcinoma. Histopathology 58:679–692. doi:10.1111/j.1365-2559.2011.03821.x
García-Solano J, Conesa-Zamora P, Carbonell P, et al. (2012) Colorectal serrated adenocarcinoma shows a different profile of oncogene mutations, MSI status and DNA repair protein expression compared to conventional and sporadic MSI-H carcinomas. Int J Cancer 131:1790–1799. doi:10.1002/ijc.27454
García-Solano J, Conesa-Zamora P, Trujillo-Santos J, et al. (2011) Tumour budding and other prognostic pathological features at invasive margins in serrated colorectal adenocarcinoma: a comparative study with conventional carcinoma. Histopathology 59:1046–1056. doi:10.1111/j.1365-2559.2011.04043.x
García-Solano J, Pérez-Guillermo M, Conesa-Zamora P, et al. (2010) Clinicopathologic study of 85 colorectal serrated adenocarcinomas: further insights into the full recognition of a new subset of colorectal carcinoma. Hum Pathol 41:1359–1368. doi:10.1016/j.humpath.2010.04.002
Zhong H, De Marzo AM, Laughner E, et al. (1999) Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 59:5830–5835
Semenza GL (2010) Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 29:625–634. doi:10.1038/onc.2009.441
Kaelin WG, Ratcliffe PJ (2008) Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell 30:393–402. doi:10.1016/j.molcel.2008.04.009
Schödel J, Oikonomopoulos S, Ragoussis J, et al. (2011) High-resolution genome-wide mapping of HIF-binding sites by ChIP-seq. Blood 117:e207–e217. doi:10.1182/blood-2010-10-314427
Kelly BD, Hackett SF, Hirota K, et al. (2003) Cell type-specific regulation of angiogenic growth factor gene expression and induction of angiogenesis in nonischemic tissue by a constitutively active form of hypoxia-inducible factor 1. Circ Res 93:1074–1081. doi:10.1161/01.RES.0000102937.50486.1B
Kikuchi H, Pino MS, Zeng M, et al. (2009) Oncogenic KRAS and BRAF differentially regulate hypoxia-inducible factor-1alpha and -2alpha in colon cancer. Cancer Res 69:8499–8506. doi:10.1158/0008-5472.CAN-09-2213
Zhong H, Chiles K, Feldser D, et al. (2000) Modulation of hypoxia-inducible factor 1alpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: implications for tumor angiogenesis and therapeutics. Cancer Res 60:1541–1545
Krieg M, Haas R, Brauch H, et al. (2000) Up-regulation of hypoxia-inducible factors HIF-1alpha and HIF-2alpha under normoxic conditions in renal carcinoma cells by von Hippel-Lindau tumor suppressor gene loss of function. Oncogene 19:5435–5443. doi:10.1038/sj.onc.1203938
Semenza GL (2013) HIF-1 mediates metabolic responses to intratumoral hypoxia and oncogenic mutations. J Clin Invest 123:3664–3671. doi:10.1172/JCI67230
Hubbi ME, Kshitiz, Gilkes DM, et al. (2013) A nontranscriptional role for HIF-1α as a direct inhibitor of DNA replication. Sci Signal 6:ra10. doi:10.1126/scisignal.2003417
Kaidi A, Williams AC, Paraskeva C (2007) Interaction between beta-catenin and HIF-1 promotes cellular adaptation to hypoxia. Nat Cell Biol 9:210–217. doi:10.1038/ncb1534
Corn PG, Ricci MS, Scata KA, et al. (2005) Mxi1 is induced by hypoxia in a HIF-1-dependent manner and protects cells from c-Myc-induced apoptosis. Cancer Biol Ther 4:1285–1294
Conesa-Zamora P, García-Solano J, García-García F, et al. (2013) Expression profiling shows differential molecular pathways and provides potential new diagnostic biomarkers for colorectal serrated adenocarcinoma. Int J Cancer 132:297–307. doi:10.1002/ijc.27674
Conesa-Zamora P, García-Solano J, Turpin MDC, et al. (2015) Methylome profiling reveals functions and genes which are differentially methylated in serrated compared to conventional colorectal carcinoma. Clin Epigenetics 7:101. doi:10.1186/s13148-015-0128-7
Koukourakis MI, Giatromanolaki A, Sivridis E, et al. (2006) Lactate dehydrogenase 5 expression in operable colorectal cancer: strong association with survival and activated vascular endothelial growth factor pathway—a report of the tumour angiogenesis research group. J Clin Oncol 24:4301–4308. doi:10.1200/JCO.2006.05.9501
Righi A, Sarotto I, Casorzo L, et al. (2014) Tumour budding is associated with hypoxia at the advancing front of colorectal cancer. Histopathology. doi:10.1111/his.12602
Saponaro C, Malfettone A, Ranieri G, et al. (2013) VEGF, HIF-1α expression and MVD as an angiogenic network in familial breast cancer. PLoS One 8:e53070. doi:10.1371/journal.pone.0053070
Kuwai T, Kitadai Y, Tanaka S, et al. (2004) Mutation of the von Hippel-Lindau (VHL) gene in human colorectal carcinoma: association with cytoplasmic accumulation of hypoxia-inducible factor (HIF)-1alpha. Cancer Sci 95:149–153
Miyakis S, Sourvinos G, Liloglou TL, et al. (2000) The von Hippel-Lindau (VHL) tumor-suppressor gene is not mutated in sporadic human colon adenocarcinomas. Int J Cancer 88:503–505
Morimoto T, Mitomi H, Saito T, et al. (2014) Distinct profile of HIF1α, PTCH, EphB2, or DNA repair protein expression and BRAF mutation in colorectal serrated adenoma. J Gastroenterol Hepatol 29:1192–1199. doi:10.1111/jgh.12553
Greijer AE, Delis-van Diemen PM, Fijneman RJA, et al. (2008) Presence of HIF-1 and related genes in normal mucosa, adenomas and carcinomas of the colorectum. Virchows Arch 452:535–544. doi:10.1007/s00428-008-0578-9
Chen Z, He X, Xia W, et al. (2013) Prognostic value and clinicopathological differences of HIFs in colorectal cancer: evidence from meta-analysis. PLoS One 8:e80337. doi:10.1371/journal.pone.0080337
Baba Y, Nosho K, Shima K, et al. (2010) HIF1A overexpression is associated with poor prognosis in a cohort of 731 colorectal cancers. Am J Pathol 176:2292–2301. doi:10.2353/ajpath.2010.090972
Sivridis E (2005) Proliferating fibroblasts at the invading tumour edge of colorectal adenocarcinomas are associated with endogenous markers of hypoxia, acidity, and oxidative stress. J Clin Pathol 58:1033–1038. doi:10.1136/jcp.2005.026260
Kuschel A, Simon P, Tug S (2012) Functional regulation of HIF-1α under normoxia—is there more than post-translational regulation? J Cell Physiol 227:514–524. doi:10.1002/jcp.22798
Dimova EY, Kietzmann T (2010) Transcription factors. Methods Mol Biol. doi:10.1007/978-1-60761-738-9
Kumar SM, Yu H, Edwards R, et al. (2007) Mutant V600E BRAF increases hypoxia inducible factor-1alpha expression in melanoma. Cancer Res 67:3177–3184. doi:10.1158/0008-5472.CAN-06-3312
Semenza GL (2012) Hypoxia-inducible factors: mediators of cancer progression and targets for cancer therapy. Trends Pharmacol Sci 33:207–214. doi:10.1016/j.tips.2012.01.005
Staton CA, Chetwood ASA, Cameron IC, et al. (2007) The angiogenic switch occurs at the adenoma stage of the adenoma carcinoma sequence in colorectal cancer. Gut 56:1426–1432. doi:10.1136/gut.2007.125286
Acknowledgments
We are grateful to Fundación para la Formación e Investigación Sanitarias from Healthcare Council of Murcia Region, Spain. We are grateful to Prof. Tuomo Karttunen for the fruitful discussions and critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Sources of funding
This work was supported by two grants from Instituto de Salud Carlos III, Ministerio de Sanidad, Spain (references PI08–1210 and PI12–1232) and grants from Academy of Finland (24,300,234), Emil Aaltonen Foundation, Finnish Cancer Foundation, and Northern Finland Cancer Foundation.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Anne Tuomisto and José García-Solano contributed equally to this work
Rights and permissions
About this article
Cite this article
Tuomisto, A., García-Solano, J., Sirniö, P. et al. HIF-1α expression and high microvessel density are characteristic features in serrated colorectal cancer. Virchows Arch 469, 395–404 (2016). https://doi.org/10.1007/s00428-016-1988-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-016-1988-8