Abstract
Purpose
Exercise-induced muscle pain is a self-limiting condition which impacts physical activity habits. Transcutaneous electrical nerve stimulation (TENS) promotes pain reduction and functional improvement in different pain conditions. We propose that applying TENS during exercise might reduce pain and improve physical performance. Thus, we aimed to investigate immediate effects of TENS applied during resistance exercise.
Methods
Healthy subjects of both sexes, irregularly active or sedentary were assigned into two groups: active (n = 24) or placebo (n = 22) TENS. The study was conducted over five moments: on day 0, subjects were recruited, on day 1 subjects performed the one-repetition maximum test (1RM); 72 h later, on day 2, 1RM was retested; 48 h later, on day 3, TENS was applied during a functional-resisted exercise protocol for upper limbs (bench press and rowing), with an intensity of 80% of 1RM; and 24 h after, on day 4, subjects were reevaluated. Assessment included pain intensity at rest and with movement, pressure pain thresholds, and muscle fatigue.
Results
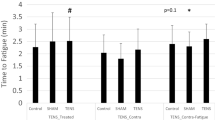
TENS did not reduce pain intensity when compared to placebo (p > 0.05). TENS reduce PPT in the latissmus dorsi: p = 0.02 and anterior tibialis: p = 0.04 in immediate reassessment. Immediate effects of TENS were significant for fatigue perception at rest (p = 0.01) and number of maximum repetitions during exercise sets, starting from the 5th set of rowing exercise (p = 0.002).
Conclusion
Our results show that TENS did not reduce pain perception in healthy individuals, but its use induced increased muscle action, contributing to a greater fatigue perception.







Similar content being viewed by others
Abbreviations
- 1RM:
-
1 maximum repetition test
- BMI:
-
Body mass index
- CPM:
-
Conditioned pain modulation
- DASH:
-
Disabilities of the arm, shoulder and hand questionnaire
- DBP:
-
Diastolic blood pressure
- DOMS:
-
Delayed-onset muscle soreness
- HR:
-
Heart rate
- IPAQ:
-
Physical activity questionnaire
- ME:
-
Muscle endurance
- MF:
-
Muscle fatigue
- PAR-Q:
-
Physical activity readiness questionnaire
- PI:
-
Pain intensity
- PO:
-
Power output
- PPT:
-
Pressure pain threshold
- ReBEC:
-
Brazilian registry of clinical trials
- RR:
-
Respiratory rate
- SBP:
-
Systolic blood pressure
- SpO2 :
-
Peripheral oxygen saturation
- ST:
-
Temporal summation
- TENS:
-
Transcutaneous electrical nerve stimulation
- TR:
-
Thermoregulation
- NMDA:
-
N-metil-d-aspartato
- NES:
-
Neuromuscular electrical stimulation
References
Al-Nakhli HH et al (2012) The use of thermal infra-red imaging to detect delayed onset muscle soreness. J Vis Exp JoVE. https://doi.org/10.3791/3551
Amann M et al (2007) Severity of arterial hypoxaemia affects the relative contributions of peripheral muscle fatigue to exercise performance in healthy humans. J Physiol 581(Pt 1):389–403
Astokorki AHY, Mauger AR (2017) Transcutaneous electrical nerve stimulation reduces exercise-induced perceived pain and improves endurance exercise performance. Eur J Appl Physiol 117(3):483–492
Benedetti F, Pollo A, Colloca L (2007) Opioid-mediated placebo responses boost pain endurance and physical performance: is it doping in sport competitions? J Neurosci Off J Soc Neurosci 27(44):11934–11939
Bickel CS, Gregory CM, Dean JC (2011) Motor unit recruitment during neuromuscular electrical stimulation: a critical appraisal. Eur J Appl Physiol 111(10):2399–2407
Bjordal JM, Johnson MI, Ljunggreen AE (2003) Transcutaneous electrical nerve stimulation (TENS) can reduce postoperative analgesic consumption. A meta-analysis with assessment of optimal treatment parameters for postoperative pain. Eur J Pain 7(2):181–188
Blanchfield A, Hardy J, Marcora S (2014) Non-conscious visual cues related to affect and action alter perception of effort and endurance performance. Front Hum Neurosci 8:967
Bouhassira D et al (2003) Comparison of the pain suppressive effects of clinical and experimental painful conditioning stimuli. Brain J Neurol 126(Pt 5):1068–1078
Boushel R (2010) Muscle metaboreflex control of the circulation during exercise. Acta Physiologica (Oxford, England) 199(4):367–383
Butterfield TA (2010) Eccentric exercise in vivo: strain-induced muscle damage and adaptation in a stable system. Exerc Sport Sci Rev 38(2):51–60
Cheung K, Hume P, Maxwell L (2003) Delayed onset muscle soreness: treatment strategies and performance factors. Sports Med (Auckland, NZ) 33(2):145–164
Connolly DAJ, Sayers SP, Mchugh MP (2003) Treatment and prevention of delayed onset muscle soreness. J Strength Cond Res 17(1):197–208
Corrêa JB et al (2013) Effects of the carrier frequency of interferential current on pain modulation in patients with chronic nonspecific low back pain: a protocol of a randomised controlled trial. BMC Musculoskelet Disord 14:195
Dailey DL et al (2013) Transcutaneous electrical nerve stimulation reduces pain, fatigue and hyperalgesia while restoring central inhibition in primary fibromyalgia. Pain 154(11):2554–2562
Dannecker EA, Koltyn KF (2014) Pain during and within hours after exercise in healthy adults. Sports Med (Auckland, NZ) 44(7):921–942
Dannecker EA, Sluka KA (2011) Pressure and activity-related allodynia in delayed-onset muscle pain. Clin J Pain 27(1):42–47
Denegar CR, Perrin DH (1992) Effect of transcutaneous electrical nerve stimulation, cold, and a combination treatment on pain, decreased range of motion, and strength loss associated with delayed onset muscle soreness. J Athl Train 27(3):200–206
DeSantana JM et al (2008a) Effectiveness of transcutaneous electrical nerve stimulation for treatment of hyperalgesia and pain. Curr Rheumatol Rep 10(6):492–499
DeSantana JM, Santana-Filho VJ, Sluka KA (2008b) Modulation between high- and low-frequency transcutaneous electric nerve stimulation delays the development of analgesic tolerance in arthritic rats. Arch Phys Med Rehabil 89(4):754–760
Dworkin RH et al (2009) Interpreting the clinical importance of group differences in chronic pain clinical trials: IMMPACT recommendations. Pain 146(3):238–244
Eckert NR et al (2017) Testing assumptions in human pain models: psychophysical differences between first and second pain. J Pain Off J Am Pain Soc 18(3):266–273
Graven-Nielsen T et al (2002) Inhibition of maximal voluntary contraction force by experimental muscle pain: a centrally mediated mechanism. Muscle Nerve 26(5):708–712
Herrero JF, Laird JM, López-García JA (2000) Wind-up of spinal cord neurones and pain sensation: much ado about something? Prog Neurobiol 61(2):169–203
Hibbert AW et al (2017) No influence of transcutaneous electrical nerve stimulation on exercise-induced pain and 5-km cycling time-trial performance. Front Physiol 8:7
Hopkins J et al (2002) Cryotherapy and transcutaneous electric neuromuscular stimulation decrease arthrogenic muscle inhibition of the vastus medialis after knee joint effusion. J Athl Train 37(1):25–31
Kahl C, Cleland JA (2005) Visual analogue scale, numeric pain rating scale and the McGill pain Questionnaire: an overview of psychometric properties. Phys Therapy Rev 10(2):123–128
Kalra A, Urban MO, Sluka KA (2001) Blockade of opioid receptors in rostral ventral medulla prevents antihyperalgesia produced by transcutaneous electrical nerve stimulation (TENS). J Pharmacol Exp Ther 298(1):257–263
Kennedy DS et al (2013) Firing of antagonist small-diameter muscle afferents reduces voluntary activation and torque of elbow flexors. J Physiol 591(14):3591–3604
Kenny GP, Mcginn R (2017) Restoration of thermoregulation after exercise. J Appl Physiol (Bethesda, Md.: 1985) 122(4):933–944
King EW et al (2005) Transcutaneous electrical nerve stimulation activates peripherally located alpha-2A adrenergic receptors. Pain 115(3):364–373
Kosek E, Ekholm J, Hansson P (1999) Pressure pain thresholds in different tissues in one body region. The influence of skin sensitivity in pressure algometry. Scand J Rehabil Med 31(2):89–93
Lauver JD, Cayot TE, Scheuermann BW (2016) Influence of bench angle on upper extremity muscular activation during bench press exercise. Eur J Sport Sci 16(3):309–316
Lavender AP, Nosaka K (2006) Comparison between old and young men for changes in makers of muscle damage following voluntary eccentric exercise of the elbow flexors. Appl Physiol Nutr Metab Physiologie Appliquee Nutrition Et Metabolisme 31(3):218–225
Liebano RE et al (2011) An investigation of the development of analgesic tolerance to TENS in humans. Pain 152(2):335–342
Liebano RE et al (2013) Transcutaneous electrical nerve stimulation and conditioned pain modulation influence the perception of pain in humans: TENS and CPM influence pain in humans. Eur J Pain 17:1539–1546
Lima LV et al (2015) Associating high intensity and modulated frequency of TENS delays analgesic tolerance in rats. Eur J Pain (London, England) 19(3):369–376
Maeda Y et al (2007) Release of GABA and activation of GABA(A) in the spinal cord mediates the effects of TENS in rats. Brain Res 1136(1):43–50
Mankovsky-Arnold T et al (2013) TENS attenuates repetition-induced summation of activity-related pain following experimentally induced muscle soreness. J Pain Off J Am Pain Soc 14(11):1416–1424
Matsudo S et al (2012) Questionário Internacional de atividade física (IPAQ): Estudo de validade e reprodutibilidade no Brasil. Revista Brasileira de Atividade Física Saúde 6(2):5–18
Matuszak ME et al (2003) Effect of rest interval length on repeated 1 repetition maximum back squats. J Strength Cond Res 17(4):634–637
Mauger AR (2014) Factors affecting the regulation of pacing: current perspectives. Open Access J Sports Med 5:209–214
Moran F et al (2011) Hypoalgesia in response to transcutaneous electrical nerve stimulation (TENS) depends on stimulation intensity. J Pain Off J Am Pain Soc 12(8):929–935
Nampo FK et al (2016) Effect of low-level phototherapy on delayed onset muscle soreness: a systematic review and meta-analysis. Lasers Med Sci 31(1):165–177
Nelson ME et al (2007) Physical activity and public health in older adults: recommendation from the American College of Sports Medicine and the American Heart Association. Circulation 116(9):1094–1105
Nie H et al (2006) Enhanced temporal summation of pressure pain in the trapezius muscle after delayed onset muscle soreness. Exp Brain Res 170(2):182–190
Orfale AG et al (2005) Translation into Brazilian Portuguese, cultural adaptation and evaluation of the reliability of the Disabilities of the Arm, Shoulder and Hand Questionnaire. Braz J Med Biol Res Revista Brasileira De Pesquisas Medicas E Biologicas 38:2, p. 293–302 n.
Osiri M et al (2000) Transcutaneous electrical nerve stimulation for knee osteoarthritis. Cochrane Database Syst Rev 4:CD002823
Pantaleão MA et al (2011) Adjusting pulse amplitude during transcutaneous electrical nerve stimulation (TENS) application produces greater hypoalgesia. J Pain Off J Am Pain Soc 12(5):581–590
Petrofsky J et al (2011) The contribution of skin blood flow in warming the skin after the application of local heat; the duality of the Pennes heat equation. Med Eng Phys 33(3):325–329
Pietrosimone BG et al (2009) Immediate effects of transcutaneous electrical nerve stimulation and focal knee joint cooling on quadriceps activation. Med Sci Sports Exerc 41(6):1175–1181
Platon B et al (2010) High-frequency, high-intensity transcutaneous electrical nerve stimulation as treatment of pain after surgical abortion. Pain 148(1):114–119
Pud D, Granovsky Y, Yarnitsky D (2009) The methodology of experimentally induced diffuse noxious inhibitory control (DNIC)-like effect in humans. Pain 144(1–2):16–19
Radhakrishnan R, Sluka KA (2003) Spinal muscarinic receptors are activated during low or high frequency TENS-induced antihyperalgesia in rats. Neuropharmacology 45(8):1111–1119
Radhakrishnan R et al (2003) Spinal 5-HT(2) and 5-HT(3) receptors mediate low, but not high, frequency TENS-induced antihyperalgesia in rats. Pain 105(1–2):205–213
Rahnama N, Rahmani-Nia F, Ebrahim K (2005) The isolated and combined effects of selected physical activity and ibuprofen on delayed-onset muscle soreness. J Sports Sci 23(8):843–850
Rakel B, Frantz R (2003) Effectiveness of transcutaneous electrical nerve stimulation on postoperative pain with movement. J Pain Off J Am Pain Soc 4(8):455–464
Rakel B et al (2010) A new transient sham TENS device allows for investigator blinding while delivering a true placebo treatment. J Pain Off J Am Pain Soc 11(3):230–238
Ritti-Dias RM et al (2011) Influence of previous experience on resistance training on reliability of one-repetition maximum test. J Strength Cond Res 25(5):1418–1422
Röhrig B et al (2010) Sample size calculation in clinical trials. Deutsches Ärzteblatt Int 107(31–32):552–556 (ago)
Rokugo T, Takeuchi T, Ito H (2002) A histochemical study of substance P in the rat spinal cord: effect of transcutaneous electrical nerve stimulation. J Nippon Med Sch Nippon Ika Daigaku Zasshi v 69(5):428–433
Sandberg ML, Sandberg MK, Dahl J (2007) Blood flow changes in the trapezius muscle and overlying skin following transcutaneous electrical nerve stimulation. Phys Therapy 87(8):1047–1055
Sato KL et al (2012) Increasing intensity of TENS prevents analgesic tolerance in rats. J Pain Off J Am Pain Soc 13(9):884–890
Sayers SP, Dannecker EA (2004) How to prevent delayed onset muscle soreness (DOMS) after eccentric exercise. Int SportMed J 5(2):84–97
Schlader ZJ et al (2011) Skin temperature as a thermal controller of exercise intensity. Eur J Appl Physiol 111(8):1631–1639
Sluka KA et al (1998) Treatment with either high or low frequency TENS reduces the secondary hyperalgesia observed after injection of kaolin and carrageenan into the knee joint. Pain 77(1):97–102
Sluka KA et al (1999) Spinal blockade of opioid receptors prevents the analgesia produced by TENS in arthritic rats. J Pharmacol Exp Ther 289(2):840–846
Sluka KA, Vance CGT, Lisi TL (2005) High-frequency, but not low-frequency, transcutaneous electrical nerve stimulation reduces aspartate and glutamate release in the spinal cord dorsal horn. J Neurochem 95(6):1794–1801
Sowman PF et al (2011) Diffuse noxious inhibitory control evoked by tonic craniofacial pain in humans. Eur J Pain 15(2):139–145
Strafella AP, Ko JH, Monchi O (2006) Therapeutic application of transcranial magnetic stimulation in Parkinson’s disease: the contribution of expectation. NeuroImage 31(4):1666–1672
Thomas S, Reading J, Shephard RJ (1992) Revision of the physical activity readiness questionnaire (PAR-Q). Can J Sport Sci Journal Canadien Des Sciences Du Sport v 17(4):338–345
Tomasi F et al (2015) Transcutaneous electrical nerve stimulation improves exercise tolerance in healthy subjects. Int J Sports Med 36(08):661–665
Tourville TW, Connolly DAJ, Reed BV (2006) Effects of sensory-level high-volt pulsed electrical current on delayed-onset muscle soreness. J Sports Sci 24(9):941–949
Tous-Fajardo J et al (2016) W5″ test: a simple method for measuring mean power output in the bench press exercise. Eur J Sport Sci 16(8):940–947 n.
Trebs AA, Brandenburg JP, Pitney WA (2010) An electromyography analysis of 3 muscles surrounding the shoulder joint during the performance of a chest press exercise at several angles. J Strength Cond Res 24(7):1925–1930
Tucker R (2009) The anticipatory regulation of performance: the physiological basis for pacing strategies and the development of a perception-based model for exercise performance. Br J Sports Med 43(6):392–400
Vieira PJC et al (2012) Effect of transcutaneous electrical nerve stimulation on muscle metaboreflex in healthy young and older subjects. Eur J Appl Physiol 112(4):1327–1334
Wager TD, Scott DJ, Zubieta J-K (2007) Placebo effects on human -opioid activity during pain. Proc Natl Acad Sci 104(26):11056–11061
Willis WD (2001) Role of neurotransmitters in sensitization of pain responses. Ann NY Acad Sci 933:142–156
Zimmermann K et al (2012) Central projection of pain arising from delayed onset muscle soreness (DOMS) in human subjects. PloS One 7(10):e47230
Zubieta J-K et al (2005) Placebo effects mediated by endogenous opioid activity on mu-opioid receptors. J Neurosci Off J Soc Neurosci 25(34):7754–7762
Butera, KA et al (2018) Prolonged reduction in shoulder strength after transcutaneous electrical nerve stimulation treatment of exercise-induced acute muscle pain. Pain Pract. https://doi.org/10.1111/papr.12690
Acknowledgements
MAM, TABP, AGRN and MEDSG conceived and designed the study. LMT, BTQL, LMSC conducted the experiment. MAM and JMDS performed data analysis. MAM wrote the manuscript. LVL and JMDS reviewed the manuscript. All authors agree with the final version of the manuscript.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
MAM, TABP, AGRN and MEDSG conceived and designed the study. LMT, BTQL, LMSC conducted the experiment. MAM and JMDS performed data analysis. MAM wrote the manuscript. LVL and JMDS reviewed the manuscript. All authors agree with the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Communicated by Bénédicte Schepens.
Rights and permissions
About this article
Cite this article
Menezes, M.A., Pereira, T.A.B., Tavares, L.M. et al. Immediate effects of transcutaneous electrical nerve stimulation (TENS) administered during resistance exercise on pain intensity and physical performance of healthy subjects: a randomized clinical trial. Eur J Appl Physiol 118, 1941–1958 (2018). https://doi.org/10.1007/s00421-018-3919-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-018-3919-7