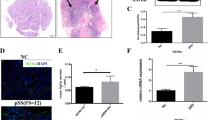
Abstract
The tumor-necrosis-factor-converting-enzyme (TACE)-TNF-α-Amphiregulin (AREG) axis plays an important pathogenic role in inflammatory and autoimmune disorders. However, the pathological roles of these proteins in the chronic autoimmune disease Sjögren’s syndrome (SS) remain to be elucidated. It is known that the TACE–AREG axis is clearly part of a larger cascade of signals that starts with the activation of Furin, responsible for maturation of TACE that, in turn, determines the production of active TNF-α, directly involved in the up-regulation of AREG expression. This study showed that Furin, TACE, TNF-α, and AREG proteins, detected in acinar and ductal cells of human salivary glands from SS patients, increased remarkably in comparison with biopsies of labial salivary glands from healthy controls. The changes in Furin, TACE, TNF- α, and AREG proteins’ level detected in salivary glands biopsies of SS patients could be responsible for pro-inflammatory cytokines overexpression characterizing Sjögren’s syndrome.




Similar content being viewed by others
References
Amrani Y, Ammit AJ, Panettieri RA Jr (2001) Tumor necrosis factor receptor (TNFR) 1, but not TNFR2, mediates tumor necrosis factor-α-induced interleukin-6 and RANTES in human airway smooth muscle cells: role of p38 and p42/44 mitogen-activated protein kinases. Mol Pharmacol 60:646–655
Apostolaki M, Armaka M, Victoratos P, Kollias G (2010) Cellular mechanisms of TNF function in models of inflammation and autoimmunity. Curr Dir Autoimmun 11:1–26
Bashir MM, Sharma MR, Werth VP (2009) TNF-alpha production in the skin. Arch Dermatol Res 301:87–91
Black RA, Rauch CT, Kozlosky CJ, Peschon JJ, Slack JL, Wolfson MF, Castner BJ, Stocking KL, Reddy P, Srinivasan S, Nelson N, Boiani N, Schooley KA, Gerhart M, Davis R, Fitzner JN, Johnson RS, Paxton RJ, March CJ, Cerretti DP (1997) A metalloproteinase disintegrin that releases tumour-necrosis factor-α from cells. Nature 385:729
Boras VV, Cikes N, Lukac J, Cekic-Arambasin A, Virag M, Bosnjak A (2004) The significance of salivary and serum interleukin 6 and basic fibroblast growth factor levels in patients with Sjogren’s syndrome. Coll Antropol 28:305–309
Brown CL, Meise KS, Plowman GD, Coffey RJ, Dempsey PJ (1988) Cell surface ectodomain cleavage of human amphiregulin precursor is sensitive to a metalloprotease inhibitor. Release of a predominant N-glycosylated 43-kDa soluble form. J Biol Chem 273:17258–17268
Chen N, Ma WY, She QB, Wu E, Liu G, Bode AM, Dong ZV (2001) Transactivation of the epidermal growth factor receptor is involved in 12-O-tetradecanoylphorbol-13-acetate-induced signal transduction. J Biol Chem 276:46722–46728
Chokki M, Mitsuhashi H, Kamimura T (2006) Metalloprotease-dependent amphiregulin release mediates tumor necrosis factor-alpha-induced IL-8 secretion in the human airway epithelial cell line NCI-H292. Life Sci 78:3051–3057
Cuello C, Palladinetti P, Tedla N, Di Girolamo N, Lloyd AR, McCluskey PJ, Wakefield D (1998) Chemokine expression and leucocyte infiltration in Sjögren’s syndrome. Br J Rheumatol 37:779–783
Davies MR, Harding CJ, Raines S, Tolley K, Parker AE, Downey-Jones M, Needham MR (2005) Nurr1 dependent regulation of pro-inflammatory mediators in immortalised synovial fibroblasts. J Inflamm 25:2–15
Fox RI, Kang HI, Ando D, Abrams J, Pisa E (1994) Cytokine mRNA expression in salivary gland biopsies of Sjogren’s syndrome. J Immunol 152:5532–5539
Hinkle CL, Sunnarborg SW, Loiselle D, Parker CE, Stevenson M, Russell WE, Lee DC (2004) Selective roles for tumor necrosis factor alpha-converting enzyme/ADAM17 in the shedding of the epidermal growth factor receptor ligand family: the juxtamembrane stalk determines cleavage efficiency. J Biol Chem 279:24179–24188
Ishii T, Onda H, Tanigawa A, Ohshima S, Fujiwara H, Mima T, Katada Y, Deguchi H, Suemura M, Miyake T, Miyatake K, Kawase I, Zhao H, Tomiyama Y, Saeki Y, Nojima H (2006) Isolation and expression profiling of genes upregulated in the peripheral blood cells of systemic lupus erythematosus patients. DNA Res 112:1–11
Kamachi M, Kawakami A, Yamasaki S, Hida A, Nakashima T, Nakamura H, Ida H, Furuyama M, Nakashima K, Shibatomi K, Miyashita T, Migita K, Eguchi K (2002) Regulation of apoptotic cell death by cytokines in a human salivary gland cell line: distinct and synergistic mechanisms in apoptosis induced by tumor necrosis factor alpha and interferon gamma. J Lab Clin Med 139:13–19
Kollias G (2005) TNF pathophysiology in murine models of chronic inflammation and autoimmunity. Semin Arthritis Rheum 34:3–6
Koski H, Janin A, Humphreys-Beher MG, Sorsa T, Malmström M, Konttinen YT (2001) Tumor necrosis factor-alpha and receptors for it in labial salivary glands in Sjögren’s syndrome. Clin Exp Rheumatol 19:131–137
Kulkarni K, Selesniemi K, Brown TL (2006) Interferon-gamma sensitizes the human salivary gland cell line, HSG, to tumor necrosis factor-alpha induced activation of dual apoptotic pathways. Apoptosis 11:2205–2215
Lisi S, Sisto M (2010) Effects of biological drug adalimumab on tumour necrosis factor-alpha-converting enzyme activation. Immunol Cell Biol 88:297–304
McArthur C, Wang Y, Veno P, Zhang J, Fiorella Rv (2002) Intracellular trafficking and surface expression of SS-A (Ro), SS-B (La), poly(ADP-ribose) polymerase and alpha-fodrin autoantigens during apoptosis in human salivary gland cells induced by tumour necrosis factor-alpha. Arch Oral Biol 47:443–448
Moss ML, Jin SL, Becherer JD, Bickett DM, Burkhart W, Chen WJ, Clay WC, Didsbury JR, Hassler D, Hoffman CR, Kost TA, Lambert MH, Leesnitzer MA, McCauley P, McGeehan G, Mitchell J, Moyer M, Pahel G, Rocque W, Overton LK, Schoenen F, Seaton T, Su JL, Becherer JD et al (1997) Cloning of a disintegrin metalloproteinase that processes precursor tumour-necrosis factor-α. Nature 385:733
Oxholm P, Daniels TE, Bendtzen K (1992) Cytokine expression in labial salivary glands from patients with primary Sjögren’s syndrome. Autoimmunity 12:185–191
Piepkorn M, Pittelkow MR, Cook PW (1998) Autocrine regulation of keratinocytes: the emerging role of heparin-binding, epidermal growth factor-related growth factors. J Invest Dermatol 111:715–721
Polihronis M, Tapinos NI, Theocharis SE, Economou A, Kittas C, Moutsopoulos HM (1998) Models of epithelial cell death and repair in Sjögren’s syndrome (SS). Clin Exp Immunol 114:485–490
Sahin U, Weskamp G, Kelly K, Zhou HM, Higashiyama S, Peschon J, Hartmann D, Saftig P, Blobel CP (2004) Distinct roles for ADAM10 and ADAM17 in ectodomain shedding of six EGFR ligands. J Cell Biol 164:769–779
Schmiegel W, Roeder C, Schmielau J, Rodeck U, Kalthoff H (1993) Tumor necrosis factor alpha induces the expression of transforming growth factor alpha and the epidermal growth factor receptor in human pancreatic cancer cells. Proc Natl Acad Sci USA 90:863–867
Sens DA, Hintz DS, Rudisill MT, Sens MA, Spicer SS (1985) Explant culture of human submandibular gland epithelial cells: evidence for ductal origin. Lab Invest 52:559–567
Shoyab M, McDonald VL, Bradley JG, Todaro GJ (1988) Amphiregulin: a bifunctional growth-modulating glycoprotein produced by the phorbol 12-myristate 13-acetate-treated human breast adenocarcinoma cell line MCF-7. Proc Natl Acad Sci USA 85:6528–6532
Sisto M, Lisi S, D’Amore M, Mitolo V, Scagliusi P (2007) Anti-Ro and anti-La autoantibodies induce TNF-alpha production by human salivary gland cells: an in vitro study. Reumatismo 59:221–226
Sisto M, D’Amore M, Scagliusi P, Mitolo V, Lisi S (2008) Selective TNF-alpha gene silencing attenuates apoptosis in human salivary gland epithelial cells. Int J Immunopathol Pharmacol 21:1045–1047
Sisto M, D’Amore M, Caprio S, Mitolo V, Scagliusi P, Lisi S (2009a) Tumor necrosis factor inhibitors block apoptosis of human epithelial cells of the salivary glands. Ann N Y Acad Sci 1171:407–414
Sisto M, Lisi S, Lofrumento DD, Caprio S, Mitolo V, D’Amore M (2009b) TNF blocker drugs modulate human TNF-alpha-converting enzyme pro-domain shedding induced by autoantibodies. Immunobiology (in press)
Sisto M, Lisi S, Lofrumento DD, Frassanito MA, Cucci L, D’Amore S, Mitolo V, D’Amore M (2009c) Induction of TNF-alpha-converting enzyme-ectodomain shedding by pathogenic autoantibodies. Int Immunol 21:1341–1349
Soliotis FC, Moutsopoulos HM (2004) Sjogren’s syndrome. Autoimmunity 37:305–307
Srour N, Lebel A, McMahon S, Fournier I, Fugère M, Day R, Dubois CM (2003) TACE/ADAM-17 maturation and activation of sheddase activity require proprotein convertase activity. FEBS Lett 554:275–283
Sternlicht MD, Sunnarborg SW (2008) The ADAM17-amphiregulin-EGFR axis in mammary development and cancer. J Mammary Gland Biol Neoplasia 13:181–194
Sunnarborg SW, Hinkle CL, Stevenson M, Russell WE, Raska CS, Peschon JJ, Castner BJ, Gerhart MJ, Paxton RJ, Black RA, Lee DC (2002) Tumor necrosis factor-alpha converting enzyme (TACE) regulates epidermal growth factor receptor ligand availability. J Biol Chem 277:12838–12845
Vitali C, Bombardieri S, Jonsson R, Moutsopoulos HM, Alexander EL, Carsons SE, Daniels TE, Fox PC, Fox RI, Kassan SS, Pillemer SR, Talal N, Weisman MH (2002) Classification criteria for Sjögren’s syndrome: a revised version of the European criteria proposed by the American-European consensus group. European study group on classification criteria for Sjögren’s syndrome. Ann Rheum Dis 61:554–558
Willeke P, Schotte H, Schlüter B, Erren M, Becker H, Dyong A, Mickholz E, Domschke W, Gaubitz M (2003) Interleukin 1beta and tumour necrosis factor alpha secreting cells are increased in the peripheral blood of patients with primary Sjögren’s syndrome. Ann Rheum Dis 62:359–362
Woodworth CD, McMullin E, Iglesias M, Plowman GD (1995) Interleukin 1 alpha and tumor necrosis factor alpha stimulate autocrine amphiregulin expression and proliferation of human papillomavirus-immortalized and carcinoma-derived cervical epithelial cells. Proc Natl Acad Sci USA 92:2840–2844
Yamane S, Ishida S, Hanamoto Y, Kumagai K, Masuda R, Tanaka K, Shiobara N, Yamane N, Mori T, Juji T, Fukui N, Itoh T, Ochi T, Suzuki R (2008) Proinflammatory role of amphiregulin, an epidermal growth factor family member whose expression is augmented in rheumatoid arthritis patients. J Inflamm 5:5
Yamashita Y, Kawashima I, Gunji Y, Hishinuma M, Shimada M (2010) Progesterone is essential for maintenance of Tace/Adam17 mRNA expression, but not EGF-like factor, in cumulus cells, which enhances the EGF receptor signaling pathway during in vitro maturation of porcine COCs. J Reprod Dev 56:315–323
Acknowledgments
This work was supported by grant (No: 20216000056) from the Italian Ministry for Universities and Research. We are grateful to M.V.C. Pragnell, B.A., for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Margherita Sisto and Sabrina Lisi have equally contributed to this work and both are equally considered as “first author”.
Rights and permissions
About this article
Cite this article
Sisto, M., Lisi, S., Lofrumento, D.D. et al. Expression of pro-inflammatory TACE-TNF-α-amphiregulin axis in Sjögren’s syndrome salivary glands. Histochem Cell Biol 134, 345–353 (2010). https://doi.org/10.1007/s00418-010-0735-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-010-0735-5