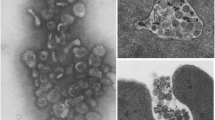
Abstract
Trypanosoma cruzi epimastigote forms concentrate their major protease, cruzipain, in the same compartment where these parasites store macromolecules obtained from medium and for this ability these organelles were named as reservosomes. Intracellular digestion occurs mainly inside reservosomes and seems to be modulated by cruzipain and its natural inhibitor chagasin that also concentrates in reservosomes. T. cruzi mammalian forms, trypomastigotes and amastigotes, are unable to capture macromolecules by endocytosis, but also express cruzipain and chagasin, whose role in infectivity has been described. In this paper, we demonstrate that trypomastigotes and amastigotes also concentrate cruzipain, chagasin as well as serine carboxypeptidase in hydrolase-rich compartments of acidic nature. The presence of P-type proton ATPase indicates that this compartment is acidified by the same enzyme as epimastigote endocytic compartments. Electron microscopy analyzes showed that these organelles are placed at the posterior region of the parasite body, are single membrane bound and possess an electron-dense matrix with electronlucent inclusions. Three-dimensional reconstruction showed that these compartments have different size and shape in trypomastigotes and amastigotes. Based on these evidences, we suggest that all T. cruzi developmental stages present lysosome-related organelles that in epimastigotes have the additional and unique ability of storing cargo.





Similar content being viewed by others
References
Adade CM, de Castro SL, Soares MJ (2007) Ultrastructural localization of Trypanosoma cruzi lysosomes by aryl sulphatase cytochemistry. Micron 38:252–256
Aparicio IM, Scharfstein J, Lima APCA (2004) A new cruzipain dependent pathway of human cell invasion by Trypanosoma cruzi requires trypomastigote membranes. Infect Immun 72:5892–5902
Besteiro S, Williams RAM, Coombs GH, Mottram JC (2007) Protein turnover and differentiation in Leishmania. Int J Parasitol 37:1063–1075
Beyenbach KW, Wieczorek H (2006) The V-type H+ ATPase: molecular structure and function, physiological roles and regulation. J Exp Biol 209:577–589
Brooks DR, Tetley L, Coombs GH, Mottram JC (2000) Processing and trafficking of cysteine proteases in Leishmania mexicana. J Cell Sci 113:4035–4041
Camargo EP (1964) Growth and differentiation of Trypanosoma cruzi. I. Origin of metacyclic trypanosomes in liquid media. Rev Inst Med Trop São Paulo 6:93–100
Campetella O, Henriksson J, Åslund L, Frasch ACC, Pettersson U, Cazzulo JJ (1992) The major cysteine proteinase (Cruzipain) from Trypanosoma cruzi is encoded by multiple polymorphic tandemly organized genes located on different chromosomes. Mol Biochem Parasitol 50:225–234
Carvalho TMU, De Souza W (1983) Separation of amastigotes and trypomastigotes of Trypanosoma cruzi from cultured cells. Z Parasitenkd 69:571–575
Cazzulo JJ, Cazzulo Franke MC, Martinez J, Franke De Cazzulo BM (1990) Some kinetic properties of a cysteine proteinase (cruzipain) from Trypanosoma cruzi. Biochim Biophys Acta. 1037:186–191
Cazzulo JJ, Stoka V, Turk V (2001) The major cysteine proteinase of Trypanosoma cruzi: a valid target for chemotherapy of Chagas disease. Current Pharm Des 7:1143–1156
Cuevas IC, Rohloff P, Sanchez DO, Docampo R (2005) Characterization of farnesylated protein tyrosine phosphatase TcPRL-1 from Trypanosoma cruzi. Eukaryot Cell 4:1550–1561
Cunha-e-Silva NL, Atella GC, Porto-Carreiro IA, Morgado-Diaz JA, Pereira MG, De Souza W (2002) Isolation and characterization of a reservosome fraction from Trypanosoma cruzi. FEMS Microbiol Lett 214:7–12
Cunha-e-Silva NL, Sant’Anna C, Pereira MG, Porto-Carreiro I, Jeovanio AL, De Souza W (2006) Reservosomes: multipurpose organelles? Parasitol Res 99:325–327
De Duve C (1973) The lysosome in retrospect. In: Dingle JT, Fell HB (eds) Lysosomes in biology and pathology, vol 1. North-Holland, Amsterdam, pp 3–40
De Souza W (2002) Basic cell biology of Trypanosoma cruzi. Curr Pharm Des 8:269–285
Eakin AE, Mills AA, Harth G, McKerrow JH, Craik CS (1992) The sequence, organization, and expression of the major cysteine protease (cruzain) from Trypanosoma cruzi. J Biol Chem 267:7411–7420
Engel JC, Doyle PS, Palmer J, Hsieh I, Bainton DF, Mckerrow JH (1998) Cysteine protease inhibitors alter Golgi complex ultrastructure and function in Trypanosoma cruzi. J Cell Sci 111:597–606
Engel JC, García CT, Hsieh I, Doyle PS, Mckerrow JH (2000) Upregulation of the secretory pathway in cysteine protease inhibitor-resistant Trypanosoma cruzi. J Cell Sci 113:1345–1354
Hall BS, Pal A, Goulding D, Field MC (2004) Rab4 is an essential regulator of lysosomal trafficking in trypanosomes. J Biol Chem 279:45047–45056
Hessler D, Young SJ, Carragher BO, Martone ME, Hinshaw JE, Milligan RA, Masliah E, Whittaker M, Lamont S, Ellisman MH (1992) SYNU: software for visualization of three-dimensional biological structures. Microscopy 22:72–82
Meirelles MN, Juliano L, Carmona E, Silva SG, Costa EM, Murta AC, Scharfstein J (1992) Inhibitors of the major cysteinyl proteinase (GP57/51) impair host cell invasion and arrest the intracellular development of Trypanosoma cruzi in vitro. Mol Biochem Parasitol 52:175–184
Monteiro AC, Abrahamson M, Lima AP, Vannier-Santos MA, Scharfstein J (2001) Identification, characterization and localization of chagasin, a tight-binding cysteine protease inhibitor in Trypanosoma cruzi. J Cell Sci 114:3933–3942
Mullin KA, Foth B, Ilgoutz SM, Callaghan J, McFadden GM, McConville MJ (2001) Regulated degradation of ER membrane proteins in a novel tubular lysosome in Leishmania mexicana. Mol Biol Cell 12:2364–2377
Murta AC, Persechini PM, Souto Padron T, De Souza W, Guimarães JA, Scharfstein J (1990) Structural and functional identification of GP57/51 antigen of Trypanosoma cruzi as a cysteine proteinase. Mol Biochem Parasitol 43:27–38
Parsons M, Ruben L (2000) Pathways involved in environmental sensing in trypanosomatids. Parasitol Today 16:56–62
Parussini F, Garcia M, Mucci J, Aguero F, Sanchez D, Hellman U, Aslund L, Cazzulo JJ (2003) Characterization of a lysosomal serine carboxypeptidase from Trypanosoma cruzi. Mol Biochem Parasitol 131:11–23
Raposo G, Marks MS, Cutler DF (2007) Lysosome related organelles: driving post-Golgi compartments into specialization. Curr Opin Cell Biol 19:394–401
Sant’Anna C, Pereira MG, Lemgruber L, De Souza W, Cunha-e-Silva N (2008) New insights into the morphology of Trypanosoma cruzi reservosome. Microsc Res Tech 71(8):599–605
Santos CC, Sant’Anna C, Terres A, Cunha-e-Silva NL, Scharfstein J, Lima AP (2005) Chagasin, the endogenous cysteine-protease inhibitor of Trypanosoma cruzi, modulates parasite differentiation and invasion of mammalian cells. J Cell Sci 118:901–915
Scharfstein J, Schmitz V, Morandi V, Capella MM, Lima APCA, Morrot A, Juliano L, Muller-Esterl W (2000) Host cell invasion by Trypanosoma cruzi is potentiated by activation of bradykinin B2 receptors. J Exp Med 192:1289–1330
Serrano R, Villalba JM, Palmgren MG, Portillo F, Parets-Soler A, Roldan M, Ferguson C, Montesinos C (1992) Studies of the plasma membrane H(+)-ATPase of yeast and plants. Biochem Soc Trans 20:562–566
Shimamura M, Hager KM, Hajduk SL (2001) The lysosomal targeting and intracellular metabolism of trypanosome lytic factor by Trypanosoma brucei brucei. Mol Biochem Parasitol 115:227–237
Soares MJ, De Souza W (1991) Endocytosis of gold-labeled proteins and LDL by Trypanosoma cruzi. Parasitol Res 77:461–469
Soares MJ, De Souza W (1988) Cytoplasmic organelles of trypanosomatids. A cytochemical and stereological stereological study. J Submicrosc Cytol Pathol 20:349–363
Soares MJ, Souto-Padrón T, Bonaldo MC, Goldenberg S, de Souza W (1989) A stereological study of the differentiation process in Trypanosoma cruzi. Parasitol Res 75:522–527
Soares MJ, Souto-Padrón T, De Souza W (1992) Identification of a large pre-lysosomal compartment in the patogenic protozoan Trypanosoma cruzi. J Cell Sci 102:157–167
Souto-Padrón T, Campetella OE, Cazzulo JJ, De Souza W (1990) Cysteine proteinase in Trypanosoma cruzi: immunocytochemical localization and involvement in parasite-host cell interation. J Cell Sci 96:485–490
Todorov AG, Andrade DS, Pesquero JB, Araújo RC, Bader M, Stewart J, Gera L, Muller-Esterl W, Morandi V, Goldenberg RC, Neto HC, Scharfstein J (2003) Trypanosoma cruzi induces edematogenic responses in mice and invades cardiomyocytes and endothelial cells in vitro by activating distinct kinin receptor (B1/B2) subtypes. FASEB J 7:73–75
Ueda-Nakamura T, a Sampaio M, Cunha-e-Silva N, Traub-Cseko YM, De Souza W (2002) Expression and processing of megasome cysteine proteinases during Leishmania amazonensis differentiation. Parasitol Res 88:332–337
Vieira M, Rohloff P, Luo S, Cunha-e-Silva NL, De Souza W, Docampo R (2005) Role for a P-type H+-ATPase in the acidification of the endocytic pathway of Trypanosoma cruzi. Biochem J 392:467–474
Waller RF, McConville MJ (2002) Developmental changes in lysosome morphology and function Leishmania parasites. Int J Parasitol 32:1435–1445
Young SJ, Royer SM, Groves PM, Kinnamon JC (1987) Three-dimensional reconstruction from serial micrographs using an IBM PC. J Elect Microsc Tech 6:207–215
Acknowledgments
The authors are grateful to Dr. Ana Paula C. A. Lima and Dr. Roberto Docampo for kindly providing the antibodies against chagasin and P-type +H-ATPase, respectively. Also, we would like to thank Mr. Antonio Bosco Carlos for technical support. This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) e Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sant’Anna, C., Parussini, F., Lourenço, D. et al. All Trypanosoma cruzi developmental forms present lysosome-related organelles. Histochem Cell Biol 130, 1187–1198 (2008). https://doi.org/10.1007/s00418-008-0486-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-008-0486-8