Abstract
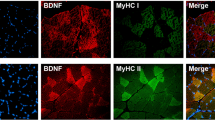
The present study was aimed to localise lymphatic vessels and their growth factors in human and mouse skeletal muscle with immunohistochemistry and specific antibodies (VEGFR-3, LYVE-1, VEGF-C and VEGF-D). The largest lymphatic vessels were found in perimysial connective tissue next to the arteries and veins, as has been shown earlier with electron microscopy. As a new finding, we also found small LYVE-1 positive vessels in the capillary bed between muscle fibres. These vessels were located next to CD31 positive blood capillaries and were of the same size, but fewer in number. In addition, we described the localisation of the two main lymphangiogenic growth factor proteins, vascular endothelial growth factor-C and -D. Both proteins were expressed in skeletal muscle at mRNA and protein levels. VEGF-D was located under the sarcolemma in some of the muscle fibres, in the endothelia of larger blood vessels and in fibroblasts. VEGF-C protein was localised to the nerves and muscle spindles, to fibroblasts and surrounding connective tissue, but was not found in muscle fibres or endothelial cells. Our results are the first to suggest the presence of lymphatic capillaries throughout the skeletal muscle, and to present the localisation of VEGF-C and -D in the muscles.




Similar content being viewed by others
References
Achen MG, Jeltsch M, Kukk E, Makinen T, Vitali A, Wilks AF, Alitalo K, Stacker SA (1998) Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk1) and VEGF receptor 3 (Flt4). Proc Natl Acad Sci USA 95:548–553
Aukland K, Reed R (1993) Interstitial-lymphatic mechanisms in the control of extracellular fluid volume. Physiol Rev 73:1–78
Bach C, Lewis GP (1973) Lymph flow and lymph protein concentration in the skin and muscle of the rabbit hind limb. J Physiol (Lond) 235:477–492
Banerji S, Ni J, Wang S-X, Clasper S, Su J, Tammi R, Jones M, Jackson DG (1999) LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J Cell Biol 144:789–801
Breiteneder-Geleff S, Soleiman A, Kowalski H, Horvet R, Amann G, Kriehuber E, Diem K, Weninger W, Tchachler E, Alitalo K, Kerjaschki D (1999) Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: podoplanin as a specific marker for lymphatic endothelium. Am J Pathol 154:385–394
Byzova TV, Goldman CK, Jankau J, Chen J, Cabrera G, Achen MG, Stacker SA, Carnevale KA, Siemionow M, Deitcher SR, DiCorleto PE (2002) Adenovirus encoding vascular endothelial growth factor-D induces tissue-specific vascular patterns in vivo. Blood 99:4434–4442
Coates G, O’Brodovich H, Goeree G (1993) Hindlimb and lung lymph flows during prolonged exercise. J Appl Physiol 75:633–638
Havas E, Parviainen T, Vuorela J, Toivanen J, Nikula T, Vihko V (1997) Lymph flow dynamics in exercising human skeletal muscle as detected by scintography. J Physiol (Lond) 504:233–239
Havas E, Lehtonen M, Vuorela J, Parviainen T, Vihko V (2000) Albumin clearance from human skeletal muscle during prolonged steady-state running. Exp Physiol 85:863–868
Hirakawa S, Young-Kwon H, Harvey N, Schacht V, Matsuda K, Liberman T, Detmar M (2003) Identification of vascular lineage-specific genes by transcriptional profiling of isolated blood vascular and lymphatic endothelial cells. Am J Pathol 162:575–586
Irjala H, Johansson EL, Grenman R, Alanen K, Salmi M, Jalkanen S (2001) Mannose receptor is a novel ligand for L-selectin and mediates lymphocyte binding to lymphatic endothelium. J Exp Med 194:1033–1042
Jackson DG (2003) The lymphatics revisited: new perspectives from the hyaluronan receptor LYVE-1. Trends Cardiovasc Med 13:1–7
Jackson DG, Prevo R, Clasper S, Banerji S (2001) LYVE-1, the lymphatic system and tumor lymphangiogenesis. Trends Immunol 22:317–321
Jakobsson S, Kjellmer I (1964) Flow and protein content of lymph in resting and exercising skeletal muscle. Acta physiol Scand 60:278–285
Jeltsch M, Kaipainen A, Joukov V, Meng X, Lakso M, Rauvala H, Swartz M, Fukumura D, Jain RK, Alitalo K (1997) Hyperplasia of lymphatic vessels in VEGF-C transgenic mice. Science 276:1423–1425
Jussila L, Alitalo K (2002) Vascular growth factors and lymphangiogenesis. Physiol Rev 82:673–700
Jussila L, Valtola R, Partanen TA, Salven P, Heikkilä P, Matikainen MT, Renkonen R, Kaipainen A, Detmar M, Tschachler E, Alitalo R, Alitalo K (1998) Lymphatic endothelium and Kaposi’s sarcoma spindle cells detected by antibodies against the vascular endothelial growth factor receptor-3. Cancer Res 58:1599–1604
Kaipainen A, Korhonen J, Mustonen T, van Hinsberg VWM, Fang G-H, Dumont D, Breitman M, Alitalo K (1995) Expression of the fms-like tyrosine kinase FLT4 gene becomes restricted to lymphatic endothelium during development. Proc Natl Acad Sci USA 92:3566–3570
Kivelä R, Silvennoinen M, Touvra AM, Lehti TM, Kainulainen H, Vihko V (2006) Effects of experimental type 1 diabetes and exercise training on angiogenic gene expression and capillarization in skeletal muscle. FASEB J 20:1570–1572 (E921–E930)
Kukk E, Lymboussaki A, Taira S, Kaipainen A, Jeltsch M, Joukov V, Alitalo K (1996) VEGF-C receptor binding and pattern of expression with VEGFR-3 suggests a role in lymphatic vascular development. Development 122:3829–3837
Le Bras B, Barallobre MJ, Homman-Ludiye J, Ny A, Wyns S, Tammela T, Haiko P, Kärkkäinen MJ, Yuan L, Muriel MP, Chatzopoulou E, Breant C, Zalc B, Carmeliet P, Alitalo K, Eichmann A, Thomas JL (2006) VEGF-C is a trophic factor for neural progenitors in the vertebrate embryonic brain. Nat Neurosci 9:340–348
Mazzoni MC, Skalak TC, Schmid-Schönbein GW (1990) Effects of skeletal muscle fibre deformation on lymphatic volumes. Am J Physiol 259:H1860–H1868
Oliver G, Detmar M (2002) The rediscovery of the lymphatic system: old and new insights into the development and biological function of the lymphatic vasculature. Genet Dev 16:773–783
Olszewski WL, Engeset A, Sokolowski J (1977) Lymph flow and protein in the normal male leg during lying, getting up, and walking. Lymphology 10:178–183
Paavonen K, Puolakkainen P, Jussila L, Jahkola T, Alitalo K (2000) Vascular endothelial growth factor receptor-3 in lymphangiogenesis in wound healing. Am J Pathol 156:1499–1504
Pajusola K, Aprelikova O, Korhonen J, Kaipainen A, Pertovaara L, Alitalo R, Alitalo K (1992) FLT4 receptor tyrosine kinase contains seven immunoglobulin-like loops and is expressed in multiple human tissues and cell lines. Cancer Res 52:5738–5743
Podgrabinska S, Braun P, Velasco P, Kloos B, Pepper MS, Jackson DG, Skobe M (2002) Molecular characterization of lymphatic endothelial cells. PNAS 99:16069–16074
Reed RK, Johansen S, Noddeland H (1985) Turnover rate of interstitial albumin in rat skin and skeletal muscle. Effects of limb movements and motor activity. Acta Physiol Scand 125:711–718
Rissanen TT, Markkanen JE, Gruchala M, Heikura T, Puranen A, Kettunen MI, Kholova I, Kauppinen RA, Achen MG, Stacker SA, Alitalo K, Ylä-Herttuala S (2003) VEGF-D is the strongest angiogenic and lymphangiogenic effector among VEGFs delivered into skeletal muscle via adenoviruses. Circ Res 92:1098–1106
Ristimäki A, Narko K, Enholm B, Joukov V, Alitalo K (1998) Proinflammatory cytokines regulate expression of the lymphatic endothelial mitogen vascular endothelial growth factor-C. J Biol Chem 273:8413–8418
Rutanen J, Leppanen P, Tuomisto TT, Rissanen TT, Hiltunen MO, Vajanto I, Niemi M, Hakkinen T, Karkola K, Stacker SA, Achen MG, Alitalo K, Yla-Herttuala S (2003) Vascular endothelial growth factor-D expression in human atherosclerotic lesions. Cardiovasc Res 59:971–979
Ryan TJ, Mortimer PS, Jones RL (1986) Lymphatics of the skin. Int J Dermatol 25:411–419
Schlingemann RO, Dingjan GM, Emeis JJ, Blok J, Warnaar SO, Ruiter DJ (1985) Monoclonal antibody PAL-E specific for endothelium. Lab Invest 52:71–76
Schmid-Schönbein GW (1990) Microlymphatics and lymph flow. Physiol Rev 70:987–1028
Skalak TC, Schmid-Schönbein GW, Zweifach BW (1984) New morphological evidence for a mechanism of lymph formation in skeletal muscle. Microvasc Res 28:95–112
Sleeman JP, Krishnan J, Kirkin V, Bauman P (2001) Markers for the lymphatic endothelium: in search of the holy grail? Microsc Res Tech 55:61–69
Stingl J, Stembera O (1974) Distribution and ultrastructure of the initial lymphatics of some skeletal muscles in the rat. Lymphology 7:160–168
Valtola R, Salven P, Heikkilä P, Taipale J, Joensuu H, Rehn M, Pihlajaniemi T, Weich H, deWaal R, Alitalo K (1999) VEGFR-3 and its ligand VEGF-C are associated in angiogenesis in breast cancer. Am J Pathol 154:1381–1390
Veikkola T, Jussila L, Makinen T, Karpanen T, Jeltsch M, Petrova TV, Kubo H, Thurston G, McDonald DM, Achen MG, Stacker SA, Alitalo K (2001) Signalling via vascular endothelial growth factor receptor-3 is sufficient for lymphangiogenesis in transgenic mice. EMBO J 20:1223–1231
Wigle JT, Oliver G (1999) Prox1 function is required for the development of the murine lymphatic system. Cell 98:769–778
Witzenbihler B, Asahara T, Murohara T, Silver M, Spyridopoulos I, Magner M, Principe N, Kearney M, Hu JS, Isner JM (1998) Vascular endothelial growth factor-C (VEGF-C/VEGF-2) promotes angiogenesis in the settings of tissue ischemia. Am J Pathol 153:381–394
Acknowledgments
The present study was financially supported by the LIKES Foundation for Sport and Health Sciences, the Finnish Ministry of Education and the Finnish Graduate School in Musculo-Skeletal Problems. Professor Kari Alitalo (Molecular/Cancer Biology Laboratory, University of Helsinki, Finland) generously provided the VEGFR-3 antibodies. Drs. Lotta Jussila and Marika Kärkkäinen (Molecular/Cancer Biology Laboratory, University of Helsinki, Finland) are acknowledged for their valuable comments and help. Dr. Reino Pitkänen (Central Hospital of Central Finland, Jyväskylä, Finland) is thanked for providing the human skin samples.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kivelä, R., Havas, E. & Vihko, V. Localisation of lymphatic vessels and vascular endothelial growth factors-C and -D in human and mouse skeletal muscle with immunohistochemistry. Histochem Cell Biol 127, 31–40 (2007). https://doi.org/10.1007/s00418-006-0226-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-006-0226-x