Abstract
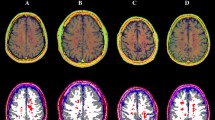
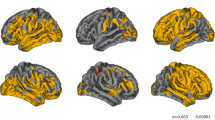
Despite that cognitive impairment is a known early feature present in multiple sclerosis (MS) patients, the biological substrate of cognitive deficits in MS remains elusive. In this study, we assessed whether T1 relaxometry, as obtained in clinically acceptable scan times by the recent Magnetization Prepared 2 Rapid Acquisition Gradient Echoes (MP2RAGE) sequence, may help identifying the structural correlate of cognitive deficits in relapsing-remitting MS patients (RRMS). Twenty-nine healthy controls (HC) and forty-nine RRMS patients underwent high-resolution 3T magnetic resonance imaging to obtain optimal cortical lesion (CL) and white matter lesion (WML) count/volume and T1 relaxation times. T1 z scores were then obtained between T1 relaxation times in lesion and the corresponding HC tissue. Patient cognitive performance was tested using the Brief Repeatable Battery of Neuro-psychological Tests. Multivariate analysis was applied to assess the contribution of MRI variables (T1 z scores, lesion count/volume) to cognition in patients and Bonferroni correction was applied for multiple comparison. T1 z scores were higher in WML (p < 0.001) and CL-I (p < 0.01) than in the corresponding normal-appearing tissue in patients, indicating relative microstructural loss. (1) T1 z scores in CL-I (p = 0.01) and the number of CL-II (p = 0.04) were predictors of long-term memory; (2) T1 z scores in CL-I (β = 0.3; p = 0.03) were independent determinants of long-term memory storage, and (3) lesion volume did not significantly influenced cognitive performances in patients. Our study supports evidence that T1 relaxometry from MP2RAGE provides information about microstructural properties in CL and WML and improves correlation with cognition in RRMS patients, compared to conventional measures of disease burden.

Similar content being viewed by others
References
Peyser JM et al (1990) Guidelines for neuropsychological research in multiple sclerosis. Arch Neurol 47(1):94–97
Rao SM et al (1991) Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41(5):685–691
Chiaravalloti ND, DeLuca J (2008) Cognitive impairment in multiple sclerosis. Lancet Neurol 7(12):1139–1151
Amato MP, Zipoli V, Portaccio E (2008) Cognitive changes in multiple sclerosis. Expert Rev Neurother 8(10):1585–1596
Amato MP, Zipoli V, Portaccio E (2006) Multiple sclerosis-related cognitive changes: a review of cross-sectional and longitudinal studies. J Neurol Sci 245(1–2):41–46
Rovaris M, Comi G, Filippi M (2006) MRI markers of destructive pathology in multiple sclerosis-related cognitive dysfunction. J Neurol Sci 245(1–2):111–116
Berg D et al (2000) Lesion pattern in patients with multiple sclerosis and depression. Mult Scler 6(3):156–162
Calabrese M et al (2009) Cortical lesions and atrophy associated with cognitive impairment in relapsing-remitting multiple sclerosis. Arch Neurol 66(9):1144–1150
Mainero C et al (2004) fMRI evidence of brain reorganization during attention and memory tasks in multiple sclerosis. Neuroimage 21(3):858–867
Rao SM et al (1989) Correlation of magnetic resonance imaging with neuropsychological testing in multiple sclerosis. Neurology 39(2 Pt 1):161–166
Swirsky-Sacchetti T et al (1992) Neuropsychological and structural brain lesions in multiple sclerosis: a regional analysis. Neurology 42(7):1291–1295
Geurts JJ et al (2005) Intracortical lesions in multiple sclerosis: improved detection with 3D double inversion-recovery MR imaging. Radiology 236(1):254–260
Calabrese M, Filippi M, Gallo P (2010) Cortical lesions in multiple sclerosis. Nat Rev Neurol 6(8):438–444
Roosendaal SD et al (2009) Accumulation of cortical lesions in MS: relation with cognitive impairment. Mult Scler 15(6):708–714
Parry A et al (2002) White matter and lesion T1 relaxation times increase in parallel and correlate with disability in multiple sclerosis. J Neurol 249(9):1279–1286
Nielsen AS et al (2013) Contribution of cortical lesion subtypes at 7T MRI to physical and cognitive performance in MS. Neurology 81(7):641–649
Bakshi R et al (2005) Imaging of multiple sclerosis: role in neurotherapeutics. NeuroRx 2(2):277–303
Castriota-Scanderbeg A et al (2004) T1 relaxation maps allow differentiation between pathologic tissue subsets in relapsing-remitting and secondary progressive multiple sclerosis. Mult Scler 10(5):556–561
McDonald WI et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50(1):121–127
Deoni SC (2010) Quantitative relaxometry of the brain. Top Magn Reson Imaging 21(2):101–113
Marques JP et al (2010) MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage 49(2):1271–1281
Kober T et al (2012) MP2RAGE multiple sclerosis magnetic resonance imaging at 3 T. Invest Radiol 47(6):346–352
Boringa JB et al (2001) The brief repeatable battery of neuropsychological tests: normative values allow application in multiple sclerosis clinical practice. Mult Scler 7(4):263–267
Polman CH et al (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58(6):840–846
Rao SM (1990) A manual for the brief repeatable battery of neuropsychological tests in multiple sclerosis. Medical College of Wisconsin Milwaukee, WI
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67(6):361–370
Penner IK et al (2009) The Fatigue Scale for Motor and Cognitive Functions (FSMC): validation of a new instrument to assess multiple sclerosis-related fatigue. Mult Scler 15(12):1509–1517
Mugler JP 3rd (2014) Optimized three-dimensional fast-spin-echo MRI. J Magn Reson Imaging 39:745–767
Roche A (2011) A four-dimensional registration algorithm with application to joint correction of motion and slice timing in fMRI. IEEE Trans Med Imaging 30(8):1546–1554
Roche A et al (2011) On the convergence of EM-like algorithms for image segmentation using Markov random fields. Med Image Anal 15(6):830–839
Bitsch A et al (2001) A longitudinal MRI study of histopathologically defined hypointense multiple sclerosis lesions. Ann Neurol 49(6):793–796
Larsson HB et al (1989) Assessment of demyelination, edema, and gliosis by in vivo determination of T1 and T2 in the brain of patients with acute attack of multiple sclerosis. Magn Reson Med 11(3):337–348
Laule C et al (2004) Water content and myelin water fraction in multiple sclerosis. A T2 relaxation study. J Neurol 251(3):284–293
Davies GR et al (2007) Normal-appearing grey and white matter T1 abnormality in early relapsing-remitting multiple sclerosis: a longitudinal study. Mult Scler 13(2):169–177
Manfredonia F et al (2007) Normal-appearing brain t1 relaxation time predicts disability in early primary progressive multiple sclerosis. Arch Neurol 64(3):411–415
Papadopoulos K et al (2010) TI-relaxation time changes over five years in relapsing-remitting multiple sclerosis. Mult Scler 16(4):427–433
Vrenken H et al (2006) Whole-brain T1 mapping in multiple sclerosis: global changes of normal-appearing gray and white matter. Radiology 240(3):811–820
Rao SM (1995) Neuropsychology of multiple sclerosis. Curr Opin Neurol 8(3):216–220
Allen G et al (1997) Attentional activation of the cerebellum independent of motor involvement. Science 275(5308):1940–1943
Audoin B et al (2003) Compensatory cortical activation observed by fMRI during a cognitive task at the earliest stage of MS. Hum Brain Mapp 20(2):51–58
Staffen W et al (2002) Cognitive function and fMRI in patients with multiple sclerosis: evidence for compensatory cortical activation during an attention task. Brain 125(Pt 6):1275–1282
Acknowledgments
We thank Jaeseok Park for his kind help with the DIR sequence as well as Georgina Palau for her dedicated work.
This work was supported by the Swiss National Science Foundation under grant PZ00P3_131914/11, the Swiss MS Society and the Societé Académique Vaudoise.
The funding sources had no role in study design, in the collection, analysis, and interpretation of data, in the writing of the report and in the decision to submit the paper for publication.
Conflicts of interest
Dr Krueger and Dr Kober work for Siemens AG. The other authors have no competing interests and nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
S. Simioni and F. Amarù contributed equally and share first authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Simioni, S., Amarù, F., Bonnier, G. et al. MP2RAGE provides new clinically-compatible correlates of mild cognitive deficits in relapsing-remitting multiple sclerosis. J Neurol 261, 1606–1613 (2014). https://doi.org/10.1007/s00415-014-7398-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7398-4