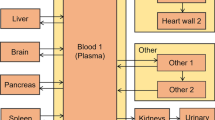
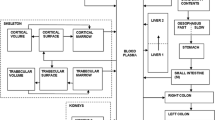
Abstract
The objective of the present work is to apply the plasma clearance parameters to strontium, previously determined in our laboratory, to improve the biokinetic and dosimetric models of strontium-90 (90Sr) used in radiological protection; and also to apply this data for the estimation of the radiation doses from strontium-89 (89Sr) after administration to patients for the treatment of the painful bone metastases. Plasma clearance and urinary excretion of stable strontium tracers of strontium-84 (84Sr) and strontium-86 (86Sr) were measured in GSF-National Research Center for Environment and Health (GSF) in 13 healthy German adult subjects after intravenous injection and oral administration. The biological half-life of strontium in plasma was evaluated from 49 plasma concentration data sets following intravenous injections. This value was used to determine the transfer rates from plasma to other organs and tissues. At the same time, the long-term retention of strontium in soft tissue and whole body was constrained to be consistent with measured values available. A physiological urinary path was integrated into the biokinetic model of strontium. Parameters were estimated using our own measured urinary excretion values. Retention and excretion of strontium were modeled using compartmental transfer rates published by the International Commission on Radiological Protection (ICRP), the SENES Oak Ridge Inc. (SENES), and the Urals Research Center for Radiation Medicine (TBM). The results were compared with values calculated by applying our GSF parameters (GSF). For the dose estimation of 89Sr, a bone metastases model (GSF-M) was developed by adding a compartment, representing the metastases, into the strontium biokinetic model. The related parameters were evaluated based on measured data available in the literature. A set of biokinetic parameters was optimized to represent not only the early plasma kinetics of strontium but also the long-term retention measured in soft tissue and whole body. The ingestion dose coefficients of 90Sr were computed and compared with different biokinetic model parameters. The ingestion dose coefficients were calculated as 2.8 × 10−8, 2.1 × 10−8, 2.5 × 10−8 and 3.8 × 10−8 Sv Bq−1 for ICRP, SENES, TBM and GSF model parameters, respectively. Moreover, organ absorbed dose for the radiopharmaceutical of 89Sr in bone metastases therapy was estimated based on the GSF and ICRP biokinetic model parameters. The effective doses were 3.3, 1.8 and 1.2 mSv MBq−1 by GSF, GSF-M, and ICRP Publication 67 model parameters, respectively, compared to the value of 3.1 mSv MBq−1 reported by ICRP Publication 80. The absorbed doses of red bone marrow and bone surface, 17 and 21 mGy MBq−1 calculated by GSF parameters, and 7.1 and 8.8 mGy MBq−1 by GSF-M parameters, are comparable to the clinical results of 3–19 mGy MBq−1 for bone marrow and 16 mGy MBq−1 for bone surface. Based on the GSF-M model, the absorbed dose of 89Sr to metastases was estimated to be 434 mGy MBq−1. The strontium clearance half-life of 0.25 h from the plasma obtained in the present study is obviously faster than the value of 1.1 h recommended by ICRP. There are no significant changes for ingestion dose coefficients of 90Sr using different model parameters. A model including the metastases was particularly developed for dose estimation of 89Sr treatment for the pain of bone metastases.








Similar content being viewed by others
References
United Nations (2000) Sources and effects of ionizing radiation: 2000 report to the General Assembly, with scientific annexes, vol 1, Annex C. United Nations Scientific Committee on the Effects of Atomic Radiation, New York
Blake GM, Zivanovic MA, McEwan AJ, Ackery DM (1986) Sr-89 therapy: strontium kinetics in disseminated carcinoma of the prostate. Eur J Nucl Med 12:447–454
Pandit-Taskar N, Batraki M, Divgi CR (2004) Radiopharmaceutical therapy for palliation of bone pain from osseous metastases. J Nucl Med 45:1358–1365
Tu SM, Kim J, Pagliaro LC, Vakar-Lopez F, Wong FC, Wen S, General R, Podoloff DA, Lin SH, Logothetis CJ (2005) Therapy tolerance in selected patients with androgen-independent prostate cancer following strontium-89 combined with chemotherapy. J Clin Oncol 23:7904–7910
Gunawardana DH, Lichtenstein M, Better N, Rosenthal M (2004) Results of strontium-89 therapy in patients with prostate cancer resistant to chemotherapy. Clin Nucl Med 29:81–85
Bauman G, Charette M, Reid R, Sathya J (2005) Radiopharmaceuticals for the palliation of painful bone metastases – a systematic review. Radiother Oncol 75:258.e1–258.e13
Storto G, Klain M, Paone G, Liuzzi R, Molino L, Marinelli A, Soricelli A, Pace L, Salvatore M (2006) Combined therapy of Sr-89 and zoledronic acid in patients with painful bone metastases. Bone 39:35–41
Chow E, Harris K, Fan G, Tsao M, Sze WM (2007) Palliative radiotherapy trials for bone metastases: a systematic review. J Clin Oncol 25:1423–1436
Malmud LS, Charkes ND (1975) Bone scanning: principles, technique and interpretation. Clin Orthop Relat Res 107:112–122
Ell PJ, Dash J, Raymond J (1976) Bone scanning: a review on purpose and method. Skeletal Radiol 1:33–45
Wasserman RH (1998) Strontium as a tracer for calcium in biological and clinical research. Clin Chem 44:437–439
International Commission on Radiological Protection (1979) Limits on intakes of radionuclides for workers. Part I. ICRP Publication 30. Ann ICRP 2(3/4)
International Commission on Radiological Protection (2006) Human alimentary tract model for radiological protection. ICRP Publication 100, Ann ICRP 36(1–2)
International Commission on Radiological Protection (1975) Report of the task group on reference man. ICRP Publication 23, Pergamon Press, Oxford
International Commission on Radiological Protection (1987) Radiation doses to patients from radiopharmaceuticals. ICRP Publication 53. Ann ICRP 18(1–4)
International Commission on Radiological Protection (1989) Age-dependent doses to members of the public from intake of radionuclides: Part 1. ICRP Publication 56. Ann ICRP 20(2)
International Commission on Radiological Protection (1993) Age-dependent doses to members of the public from intake of radionuclides: Part 2 Ingestion dose coefficients. ICRP Publication 67. Ann ICRP 23(3–4)
Shagina NB, Tolstykh EI, Degteva MO, Kozheurov VP, Anspaugh LR, Napier BA (2002) Update and improvements in a model for the biokinetics of strontium. Final Report for milestone 3, Part 2 US–Russian Joint Coordinating Committee on Radiation Effects Research, Project 1.1; Chelyabinsk: Urals Research Center for Radiation Medicine
Apostoaei AI (2001) Determination of the uncertainties in radiation doses from ingestion of 90Sr. Dissertation, University of Tennessee, Knoxville, TN
Höllriegl V, Werner E, Roth P, Schramel P, Wendler I (2002) Studies of strontium biokinetics in humans. Part 1: optimisation of intrinsic labelling of foodstuffs with stable isotopes of strontium. Radiat Environ Biophys 41:179–183
Höllriegl V, Louvat P, Werner E, Roth P, Schramel P, Wendler I, Felgenhauer N, Zilker T (2002) Studies of strontium biokinetics in humans. Part 2: uptake of strontium from aqueous solution and labelled foodstuffs. Radiat Environ Biophys 41:281–287
Höllriegl V, Roth P, Werner E, Schramel P, Wendler I, Felgenhauer N, Zilker T (2003) Influence of calcium on the uptake of ingested strontium. Kerntechnik 68:185–188
Höllriegl V, Röhmuß M, Oeh U, Roth P (2004) Strontium biokinetics in humans: influence of alginate on the uptake of ingested strontium. Health Phys 86:193–196
Höllriegl V, Li WB, Oeh U, Roth P (2006) Methods for assessing gastrointestinal absorption of strontium in humans by stable tracer techniques. Health Phys 90:232–240
Li W B, Höllriegl V, Roth P, Oeh U (2006) Human biokinetics of strontium. Part I: intestinal absorption rate and its impact on the dose coefficient of 90Sr after ingestion. Radiat Environ Biophys 45:115–124
Höllriegl V, Li WB, Oeh U (2006) Human biokinetics of strontium-part II: Final data evaluation of intestinal absorption and urinary excretion of strontium in human subjects after stable tracer administration. Radiat Environ Biophys 45:179–185
Smith H, Navani A, Fishman SM (2004) Radiopharmaceuticals for palliation of painful osseous metastases. Am J Hosp Palliat Care 21:303–313
Silberstein EB (2000) Systemic radiopharmaceutical therapy of painful osteoblastic metastases. Semin Radiat Oncol 10:240–249
McEwan AJB (2000) Use of radionuclides for the palliation of bone metastases. Semin Radiat Oncol 10:103–114
Robinson RG, Preston DF, Spicer JA, Baxter KG (1992) Radionuclide therapy of intractable bone pain: emphasis on strontium-89. Semin Nucl Med 22:28–32
Silberstein EB, Williams C (1985) Strontium-89 therapy for the pain of osseous metastases. J Nucl Med 26:345–348
Bialkowski MM, Wierzbicki JG, Porter AT (1997) Modeling of internal dose distributions during Sr-89 treatment of a patient with bone metastases. Cancer Biother Radiopharm 12:355–362
Breen SL, Powe JE, Porter AT (1992) Dose estimation in strontium-89 radiotherapy of metastatic prostatic carcinoma. J Nucl Med 33:1316–1323
DeGrazia JA, Ivanovich P, Fellows H, Rich C (1965) A double isotope method for measurement of intestinal absorption of calcium in man. J Lab Clin Med 66:822–829
Werner E, Roth P, Höllriegl V, Hansen Ch, Kaltwasser JP, Giussani A, Cantone MC, Greim H, Zilker T, Felgenhauer N (2002) Assessment of intestinal absorption of trace metals in humans by means of stable isotopes. Isotopes Environ Health Stud 38:23–31
Firestone RB (1996) Table of isotopes. 8th CD-ROM edn. Wiley, New York
Tsai SC, Hsieh TH, Hsieh BT, Lin WY, Wang SJ, Lee DW, Shen LH, Huang H, Ting G (2000) Biodistribution study of strontium-85–Strontium chloride in rats. Ann Nucl Med Sci 13:37–41 (in Chinese)
Sotogaku N, Endo K, Hirunuma R, Enomoto S, Ambe S, Ambe F (1999) Binding properties of various metals to blood components and serum proteins: a multitracer study. J Trace Elem Med Biol 13:1–6
Olehy DA, Schmitt RA, Bethard WF (1966) Neutron activation analysis of magnesium, calcium, strontium, barium, manganese, cobalt, copper, zinc, sodium, and potassium in human erythrocytes and plasma. J Nucl Med 7:917–927
Höllriegl V, Li WB, Greiter M, Oeh U (2007) Plasma clearance and urinary excretion after intravenous injection of stable 84Sr isotope in humans. Radiat Prot Dosimetry. doi:10.1093/rpd/ncm265
Mraz FR (1962) Calcium and strontium uptake by rat liver and kidney mitochondria. Proc Soc Exp Biol Med 111:429–431
International Commission on Radiological Protection (1980) Limits of intakes radionuclides by workers. ICRP Publication 30 Part 2. Ann ICRP 4(3/4)
Apostoaei AI, Miller LF (2004) Uncertainty in dose coefficients from ingestion of 131I, 137Cs, and 90Sr. Health Phys 86:460–482
Krüger-Thiemer E (1977) Chapter 2. Pharmacokinetics. Kinetic aspects of absorption, distribution, and elimination of drugs. In: van Rossum JM (ed) Kinetics of drug action. Handbook of experimental pharmacology, vol 47, Heffter-Heubner New Series. Springer, Berlin, pp 63–123
Guyton AC, Hall JE (2006) Textbook of medical physiology. 11th edn. Elsevier Saunders, Philadelphia, pp 314–315
Leggett RW (1993) An age-specific kinetic model of lead metabolism in humans. Environ Health Perspect 101:598–616
Bishop M, Harrison GE, Raymond WH, Sutton A, Rundo J (1960) Excretion and retention of radioactive strontium in normal men following a single intravenous injection. Int J Radiat Biol 2:125–142
Harrison GE, Carr TEF, Sutton A (1967) Distribution of radioactive calcium, strontium, barium and radium following intravenous injection into a health man. Int J Radiat Biol 13:235–247
Newton D, Harrison GE, Rundo J, Kang C, Warner AJ (1990) Metabolism of Ca and Sr in late adult life. Health Phys 59:433–442
Carr TEF, Harrison GE, Nolan J (1973) The long-term excretion and retention of an intravenous dose of 45Ca in two health men. Calcif Tiss Res 12:217–226
Bauer GC, Ray RD (1958) Kinetics of strontium metabolism in man. J Bone Joint Surg 40-A:171–186
Likhtarev IA, Dobroskok IA, Ilyin LA, Krasnoschekova GP, Likhtareva M, Smirnov BI, Sobolev EP, Shamov VP, Shapiro EL (1975) A study of certain characteristics of strontium metabolism in a homogeneous group of human subjects. Health Phys 28:49–60
Schulert AR, Peets EA, Laszlo D, Spencer H, Charles M, Samachson J (1959) Comparative metabolism of strontium and calcium in man. Int J Appl Radiat Isot 4:144–153
Mays CW, Lloyd RD, Christensen WR, Atherton DR, Pitchford GS (1963) Radium metabolism in a man. Radiat Res 19:210
Comar CL, Wasserman RH, Ullberg S, Andrews GA (1957) Strontium metabolism and strontium–calcium discrimination in man. Proc Soc Exp Biol Med 95:386–391
Fujita M, Yabe A, Ueno K, Oshino M, Okuyama N (1963) The behavior of strontium-85 in a normal man following a single ingestion—absorption and excretion. Health Phys 9:407–415
Apostoaei AI (2002) Absorption of strontium from the gastrointestinal tract into plasma in healthy human adults. Health Phys 83:56–65
Dickinson CZ, Hendrix NS (1993) Strontium-89 therapy in painful bony metastases. J Nucl Med Technol 21:133–137
Robinson RG, Blake GM, Preston DF, McEwan AJ, Spicer JA, Martin NL, Wegst AV, Ackery DM (1989) Strontium-89: treatment results and kinetics in patients with painful metastatic prostate and breast cancer in bone. Radiographics 9:271–281
International Commission on Radiological Protection (1998) Radiation dose to patients from radiopharmaceuticals (Addendum 2 to ICRP Publication 53). ICRP Publication 80, Ann ICRP 28(3)
International Commission on Radiological Protection (1991) 1990 Recommendations of the International Commission on Radiological Protection. ICRP Publication 60, Ann ICRP 21(1–3)
International Commission on Radiological Protection (1977) Recommendation of the International Commission on Radiological Protection. ICRP Publication 26, Ann ICRP 1(3)
Harrison JD, Leggett RW, Noßke D, Paquet F, Phipps AW, Taylor DM, Métivier H (2001) Reliability of the ICRP’s dose coefficients for members of the public, II. Uncertainties in the absorption of ingested radionuclides and the effect on dose estimates. Radiat Prot Dosimetry 95:295–308
Harrison JD, Khursheed A, Phipps AW, Goossens L, Kraan B, Harper F (1998) Uncertainties in biokinetic parameters and dose coefficients determined by expert judgement. Radiat Prot Dosimetry 79:355–358
Goossens LHJ, Harrison JD, Kraan BCP, Cooke RM, Harper FT, Hora SC (1998) Probabilistic accident consequence uncertainty analysis. Uncertainty assessment for internal dosimetry, vol 1–2, Appendices. NUREG/CR-6571; EUR 16773; SAND98-0119, Office for Official Publications of the European Communities, Luxembourg
Acknowledgments
We thank James E. Turner, Matthias Greiter and Augusto Giussani for a valuable review of the manuscript. We thank Andrei Iulian Apostoaei for providing us his dissertation. Thanks are due to the reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W.B., Höllriegl, V., Roth, P. et al. Influence of human biokinetics of strontium on internal ingestion dose of 90Sr and absorbed dose of 89Sr to organs and metastases. Radiat Environ Biophys 47, 225–239 (2008). https://doi.org/10.1007/s00411-007-0154-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-007-0154-8