Abstract
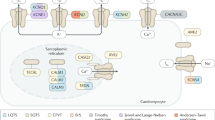
Inherited long QT syndrome (LQTS) is characterized by a prolonged ventricular repolarization (QTc interval) and symptoms (syncope, sudden cardiac arrest) due to polymorphic ventricular arrhythmias. As of today, 13 different cardiac ion channel genes have been associated with congenital LQTS. The most common ones are due to KCNQ1 (LQT-1), KCNH2 (LQT-2), and SCN5A (LQT-3) gene mutations and account for up to 75 % of cases. Typical clinical findings are an increased QT interval on the surface electrocardiogram, specifically altered T wave morphologies, polymorphic ventricular arrhythmias, or an indicative family history. Recently, in the HRS/EHRA expert consensus statement, comprehensive genetic testing of major LQTS genes was recommended for index patients for whom there is a strong clinical suspicion of LQTS. Overall, antiadrenergic therapy, in particular β-receptor blockers, has been the mainstay of therapy and has significantly reduced cardiac events. For high-risk patients, an implantable cardioverter defibrillator (ICD) is recommended. Importantly, lifestyle modification and avoidance of arrhythmia triggers are additional important approaches.
Zusammenfassung
Das angeborene lange QT-Syndrom (LQTS) ist charakterisiert durch eine verlängerte Repolarisation und ein erhöhtes Risiko für das Auftreten eines plötzlichen Herztods. Bislang sind 13 Krankheitsgene bekannt, die für das angeborene LQTS verantwortlich sind; die Ionenkanalgene KCNQ1 (LQT-1), KCNH2 (LQT-2) und SCN5A (LQT-3) sind die häufigsten und für ca. 75 % der Fälle verantwortlich. Typische klinische Zeichen sind neben dem verlängerten QT-Intervall spezifische T-Wellen-Morphologien, polymorphe, ventrikuläre Rhythmusstörungen oder eine hinweisende Familienanamnese. Kürzlich wurde eine internationale Expertenmeinung (HRS/EHRA) verfasst, die bei allen Patienten, bei denen ein hochgradiger, klinischer Verdacht auf ein LQTS besteht, eine genetische Testung empfiehlt. In therapeutischer Hinsicht ist die β-Rezeptor-Blocker-Therapie Mittel der ersten Wahl, da diese signifikant das Auftreten von kardialen Ereignissen verringert. Bei LQTS-Patienten mit einem hohen, kardialen Risiko, besteht die Indikation für einen implantierbaren Kardioverter-Defibrillator (ICD). Unabhängig hiervon haben die Anpassung des Lebensstils und das Vermeiden von Arrhythmietriggern einen wichtigen Stellenwert in der Behandlung der Patienten.


Similar content being viewed by others
References
Ackerman MJ, Priori SG, Willems S et al (2011) HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies: this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Europace 13:1077–1109
Amin AS, Giudicessi JR, Tijsen AJ et al. (2012) Variants in the 3′ untranslated region of the KCNQ1-encoded Kv7.1 potassium channel modify disease severity in patients with type 1 long QT syndrome in an allele-specific manner. Eur Heart J 33:714–723
Anderson CL, Delisle BP, Anson BD et al (2006) Most LQT2 mutations reduce Kv11.1 (hERG) current by a class 2 (trafficking-deficient) mechanism. Circulation 113:365–373
Bartos DC, Duchatelet S, Burgess DE et al (2011) R231C mutation in KCNQ1 causes long QT syndrome type 1 and familial atrial fibrillation. Heart Rhythm 8:48–55
Bellocq C, Van Ginneken AC, Bezzina CR et al (2004) Mutation in the KCNQ1 gene leading to the short QT-interval syndrome. Circulation 109:2394–2397
Bhuiyan ZA, Momenah TS, Gong Q et al (2008) Recurrent intrauterine fetal loss due to near absence of HERG: clinical and functional characterization of a homozygous nonsense HERG Q1070X mutation. Heart Rhythm 5:553–561
Chen S, Zhang L, Bryant RM et al (2003) KCNQ1 mutations in patients with a family history of lethal cardiac arrhythmias and sudden death. Clin Genet 63:273–282
Crotti L, Celano G, Dagradi F et al (2008) Congenital long QT syndrome. Orphanet J Rare Dis 3:18
Etheridge SP, Bowles NE, Arrington CB et al (2011) Somatic mosaicism contributes to phenotypic variation in Timothy syndrome. Am J Med Genet A 155A:2578–2583
Gemma LW, Ward GM, Dettmer MM et al (2011) beta-blockers protect against dispersion of repolarization during exercise in congenital long-QT syndrome type 1. J Cardiovasc Electrophysiol 22:1141–1146
Gillis J, Burashnikov E, Antzelevitch C et al (2011) Long QT, syndactyly, joint contractures, stroke and novel CACNA1C mutation: Expanding the spectrum of Timothy syndrome. Am J Med Genet A [Epub ahead of print]
Goldenberg I, Moss AJ, Zareba W (2006) QT interval: how to measure it and what is “normal”. J Cardiovasc Electrophysiol 17:333–336
Goldenberg I, Bradley J, Moss A et al (2010) Beta-blocker efficacy in high-risk patients with the congenital long-QT syndrome types 1 and 2: implications for patient management. J Cardiovasc Electrophysiol 21:893–901
Goldenberg I, Horr S, Moss AJ et al (2011) Risk for life-threatening cardiac events in patients with genotype-confirmed long-QT syndrome and normal-range corrected QT intervals. J Am Coll Cardiol 57:51–59
Goldenberg I, Thottathil P, Lopes CM et al (2012) Trigger-specific ion-channel mechanisms, risk factors, and response to therapy in type 1 long QT syndrome. Heart Rhythm 9:49–56
Hobbs JB, Peterson DR, Moss AJ et al (2006) Risk of aborted cardiac arrest or sudden cardiac death during adolescence in the long-QT syndrome. JAMA 296:1249–1254
Hofman N, Wilde AA, Kaab S et al (2007) Diagnostic criteria for congenital long QT syndrome in the era of molecular genetics: do we need a scoring system? Eur Heart J 28:575–580
Johnson WH, Jr, Yang P, Yang T et al (2003) Clinical, genetic, and biophysical characterization of a homozygous HERG mutation causing severe neonatal long QT syndrome. Pediatr Res 53:744–748
Kaufman ES, Mcnitt S, Moss AJ et al (2008) Risk of death in the long QT syndrome when a sibling has died. Heart Rhythm 5:831–836
Khan IA, Gowda RM (2004) Novel therapeutics for treatment of long-QT syndrome and torsade de pointes. Int J Cardiol 95:1–6
Kim JA, Lopes CM, Moss AJ et al (2010) Trigger-specific risk factors and response to therapy in long QT syndrome type 2. Heart Rhythm 7:1797–1805
Kurokawa J, Tamagawa M, Harada N et al (2008) Acute effects of oestrogen on the guinea pig and human IKr channels and drug-induced prolongation of cardiac repolarization. J Physiol 586:2961–2973
Migdalovich D, Moss AJ, Lopes CM et al (2011) Mutation and gender-specific risk in type 2 long QT syndrome: implications for risk stratification for life-threatening cardiac events in patients with long QT syndrome. Heart Rhythm 8:1537–1543
Mihic A, Chauhan VS, Gao X et al (2011) Trafficking defect and proteasomal degradation contribute to the phenotype of a novel KCNH2 long QT syndrome mutation. PLoS One 6:e18273
Mohler PJ, Schott JJ, Gramolini AO et al (2003) Ankyrin-B mutation causes type 4 long-QT cardiac arrhythmia and sudden cardiac death. Nature 421:634–639
Moss AJ, Zareba W, Benhorin J et al (1995) ECG T-wave patterns in genetically distinct forms of the hereditary long QT syndrome. Circulation 92:2929–2934
Moss AJ, Zareba W, Hall WJ et al (2000) Effectiveness and limitations of beta-blocker therapy in congenital long-QT syndrome. Circulation 101:616–623
Moss AJ, Zareba W, Schwarz KQ et al (2008) Ranolazine shortens repolarization in patients with sustained inward sodium current due to type-3 long-QT syndrome. J Cardiovasc Electrophysiol 19:1289–1293
Obeyesekere MN, Klein GJ, Modi S et al (2011) How to perform and interpret provocative testing for the diagnosis of Brugada syndrome, long-QT syndrome, and catecholaminergic polymorphic ventricular tachycardia. Circ Arrhythm Electrophysiol 4:958–964
Paulussen AD, Gilissen RA, Armstrong M et al (2004) Genetic variations of KCNQ1, KCNH2, SCN5A, KCNE1, and KCNE2 in drug-induced long QT syndrome patients. J Mol Med (Berl) 82:182–188
Plaster NM, Tawil R, Tristani-Firouzi M et al (2001) Mutations in Kir2.1 cause the developmental and episodic electrical phenotypes of Andersen’s syndrome. Cell 105:511–519
Priori SG, Napolitano C, Schwartz PJ et al (2004) Association of long QT syndrome loci and cardiac events among patients treated with beta-blockers. JAMA 292:1341–1344
Priori SG, Pandit SV, Rivolta I et al (2005) A novel form of short QT syndrome (SQT3) is caused by a mutation in the KCNJ2 gene. Circ Res 96:800–807
Rautaharju PM, Zhou SH, Wong S et al (1992) Sex differences in the evolution of the electrocardiographic QT interval with age. Can J Cardiol 8:690–695
Rautaharju PM, Surawicz B, Gettes LS et al (2009) AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: the ST segment, T and U waves, and the QT interval: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol 53:982–991
Ruan Y, Denegri M, Liu N et al (2010) Trafficking defects and gating abnormalities of a novel SCN5A mutation question gene-specific therapy in long QT syndrome type 3. Circ Res 106:1374–1383
Schwartz PJ (1985) Idiopathic long QT syndrome: progress and questions. Am Heart J 109:399–411
Schwartz PJ (2005) Management of long QT syndrome. Nat Clin Pract Cardiovasc Med 2:346–351
Schwartz PJ (2006) The congenital long QT syndromes from genotype to phenotype: clinical implications. J Intern Med 259:39–47
Schwartz PJ, Stramba-Badiale M (2010) Repolarization abnormalities in the newborn. J Cardiovasc Pharmacol 55:539–543
Schwartz PJ, Spazzolini C, Crotti L et al (2006) The Jervell and Lange-Nielsen syndrome: natural history, molecular basis, and clinical outcome. Circulation 113:783–790
Schwartz PJ, Stramba-Badiale M, Crotti L et al (2009) Prevalence of the congenital long-QT syndrome. Circulation 120:1761–1767
Schwartz PJ, Spazzolini C, Priori SG et al (2010) Who are the long-QT syndrome patients who receive an implantable cardioverter-defibrillator and what happens to them?: data from the European Long-QT Syndrome Implantable Cardioverter-Defibrillator (LQTS ICD) Registry. Circulation 122:1272–1282
Shimizu W, Noda T, Takaki H et al (2003) Epinephrine unmasks latent mutation carriers with LQT1 form of congenital long-QT syndrome. J Am Coll Cardiol 41:633–642
Stramba-Badiale M, Spagnolo D, Bosi G et al (1995) Are gender differences in QTc present at birth? MISNES Investigators. Multicenter Italian Study on Neonatal Electrocardiography and Sudden Infant Death Syndrome. Am J Cardiol 75:1277–1278
Tester DJ, Will ML, Haglund CM et al (2005) Compendium of cardiac channel mutations in 541 consecutive unrelated patients referred for long QT syndrome genetic testing. Heart Rhythm 2:507–517
Tristani-Firouzi M, Jensen JL, Donaldson MR et al (2002) Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J Clin Invest 110:381–388
Wang Q, Shen J, Splawski I et al (1995) SCN5A mutations associated with an inherited cardiac arrhythmia, long QT syndrome. Cell 80:805–811
Yang Y, Liang B, Liu J et al (2010) Identification of a Kir3.4 mutation in congenital long QT syndrome. Am J Hum Genet 86:872–880
Zareba W (2006) Genotype-specific ECG patterns in long QT syndrome. J Electrocardiol 39:S101–S106
Zareba W, Moss AJ, Locati EH et al (2003) Modulating effects of age and gender on the clinical course of long QT syndrome by genotype. J Am Coll Cardiol 42:103–109
Zhang S, Yin K, Ren X et al (2008) Identification of a novel KCNQ1 mutation associated with both Jervell and Lange-Nielsen and Romano-Ward forms of long QT syndrome in a Chinese family. BMC Med Genet 9:24
Zhang X, Chen S, Zhang L et al (2008) Protective effect of KCNH2 single nucleotide polymorphism K897T in LQTS families and identification of novel KCNQ1 and KCNH2 mutations. BMC Med Genet 9:87
Conflict of interest
On behalf of all authors, the corresponding author states that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zumhagen, S., Stallmeyer, B., Friedrich, C. et al. Inherited long QT syndrome. Herzschr Elektrophys 23, 211–219 (2012). https://doi.org/10.1007/s00399-012-0232-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00399-012-0232-8