Abstract
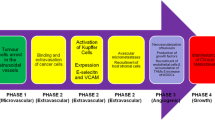
The liver is recognized as the target organ of metastases of almost all prominent malignancies. Its unique biology renders this organ particularly susceptible to circulating disseminated tumour cells (DTCs), and it can be assumed that very early metastasis occurs in the liver. The premetastatic niche concept may explain very early metastasis, as it defines priming of a future target organ of metastasis by factors that may already be secreted from premalignant lesions. This review shows that comprehensive knowledge on mechanisms of premetastatic niche formation in the liver is based on pre-clinical models only and still rather rare, mostly due to the scarcity of mouse liver metastasis models displaying a tumour cell-free period in the liver or lack of liver-tropic syngeneic tumour cells to probe for the niche. Attentive re-assessment of previous studies and reviews was undertaken revealing only two clearly identified tumour-derived secreted factors (TDSFs), both inducing infiltration of the liver by bone marrow-derived cells and increased liver metastasis, namely tissue inhibitor of metalloproteinases-1 (TIMP-1) and macrophage-inducing factor (MIF). Future directions of this research area will comprise elucidation of the impact of TDSFs on regulation and activity of myeloid-derived suppressor cells and/or the specific architecture and homeostasis of the liver, as well as development of prognostic TDSF detection in patients at risk of liver metastasis.



Similar content being viewed by others
References
Stoecklein NH, Klein CA (2010) Genetic disparity between primary tumours, disseminated tumour cells, and manifest metastasis. Int J Cancer 126:589–598
Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F, Reichert M, Beatty GL, Rustgi AK, Vonderheide RH et al (2012) EMT and dissemination precede pancreatic tumor formation. Cell 148:349–361
Polzer B, Klein CA (2013) Metastasis awakening: the challenges of targeting minimal residual cancer. Nat Med 19:274–275
Sceneay J, Smyth MJ, Möller H (2013) The pre-metastatic niche: finding common ground. Cancer Metastasis Rev 32:449–464
Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C, MacDonald DD, Jin DK, Shido K, Kerns SA et al (2005) VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438:820–827
Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H et al (2015) Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol. doi:10.1038/ncb3169
Yachida S, Jones S, Bozic I, Antal T, Leary A, Fu B, Kamiyama M, Hruban RH, Eshleman JR, Nowak MA et al (2010) Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 467:1114–1117
Mauti LA, Le Bitoux MA, Baumer K, Stehle JC, Golshayan D, Provero P, Stamenkovic I (2011) Myeloid-derived suppressor cells are implicated in regulating permissiveness for tumor metastasis during mouse gestation. J Clin Invest 121:2794–2807
Van den Eynden GG, Majeed AW, Illemann M, Vermeulen PB, Bird NC, Hoyer-Hansen G, Eefsen RL, Reynolds AR, Brodt P (2013) The multifaceted role of the microenvironment in liver metastatsis: biology and clinical implications. Cancer Res 73:2031–2043
Hess KR, Varadhachary GR, Taylor SH, Wei W, Raber MN, Lenzi R, Abbruzzese JL (2006) Metastatic patterns in adenocarcinoma. Cancer 106:1624–1633
Jaques DP, Coit DG, Casper ES ES, Brennan MF (1995) Hepatic metastases from soft-tissue sarcoma. Ann Surg 221:392–397
Amankwah EK, Conley AP, Reed DR (2013) Epidemiology and therapies for metastatic sarcoma. Clin Epidemiol 5:147–162
Ryu SW, Saw R, Scolyer RA, Crawford M, Thompson JF, Sandroussi C (2013) Liver resection for metastatic melanoma: equivalent survival for cutaneous and ocular primaries. J Surg Oncol 108:129–135
Bross ID, Viadana E, Pickren JW (1975) The metastatic spread of myeloma and leukemias in men. Virchows Arch A Pathol Anat Histol 365:91–101
Burke D, Allen-Mersh TG (1996) Colorectal liver metastases. Postgrad Med J 72:464–469
Helling TS, Martin M (2014) Cause of death from liver metastases in colorectal cancer. Ann Surg Oncol 21:501–506
Mogrovejo E, Manickam P, Amin M, Cappell MS (2014) Characterization of the syndrome of acute liver failure caused by metastases from breast carcinoma. Dig Dis Sci 59:724–736
Chambers AF, Groom AC, MacDonald IC (2002) Metastasis: dissemination and growth of cancer cells in metastatic sites. Nature Rev Cancer 2:563–572
Vidal-Vanaclocha F (2011) Architectural and functional aspects of the liver with implications for cancer metastasis. In: P. Brodt (ed.) Liver metastasis: biology and clinical management, cancer metastasis—biology and treatment 16, DOI 10.1007/978-94-007-0292-9_2, Springer Science + Business Media B.V 9-42
Paschos KA, Majeed AW, Bird NC (2014) Natural history of hepatic metastases from colorectal cancer—pathobiological pathways with clinical significance. World J Gastroenterol 20:3719–3737
Enns A, Gassmann P, Schlüter K, Korb T, Spiegel HU, Senninger N, Haier J (2004) Integrins can directly mediate metastatic tumor cell adhesion within the liver sinusoids. J Gastrointest Surg 8:1049–1059
Vollmar B, Menger MD (2009) The hepatic microcirculation: mechanistic contributions and therapeutic targets in liver injury and repair. Physiol Rev 89:1269–1339
Höchst B, Schildberg FA, Sauerborn P, Gäbel YA, Gevensleben H, Goltz D, Heukamp LC, Türler A, Ballmaier M, Gieseke F et al (2013) Activated human hepatic stellate cells induce myeloid derived suppressor cells from peripheral blood monocytes in a CD44-dependent fashion. J Hepatol 59:528–535
Pancione M, Giordano G, Remo A, Febbraro A, Sabatino L, Manfrin E, Ceccarelli M, Colantuoni V (2014) Immune escape mechanisms in colorectal cancer pathogenesis and liver metastasis. J Immunol Res 2014:686879
Vidal-Vanaclocha F (2008) The prometastatic environment of the liver. Cancer Microenviron 1:113–129
Schirrmacher V, Bosslet K (1980) Tumor metastases and cell-mediated immunity in a model system in DBA/2 mice. X. Immunoselection of tumor variants differing in tumor antigen expression and metastatic capacity. Int J Cancer 25:781–788
Krüger A, Schirrmacher V, von Hoegen P (1994) Scattered micrometastases visualized at the single-cell level: detection and re-isolation of lacZ-labeled metastasized lymphoma cells. Int J Cancer 58:275–284
Krüger A, Schirrmacher V, Khokha R (1998-1999) The bacterial lacZ gene: an important tool for metastasis research and evaluation of new cancer therapies. Cancer Metastasis Rev 17:285-294
Weinspach D, Seubert B, Schaten S, Honert K, Sebens S, Altevogt P, Krüger A (2014) Role of L1 cell adhesion molecule (L1CAM) in the metastatic cascade: promotion of dissemination, colonization, and metastatic growth. Clin Exp Metastasis 31:87–100
Zhang Y, Davis C, Ryan J, Janney C, Pena MM (2013) Development and characterization of a reliable mouse model of colorectal cancer metastasis to the liver. Clin Exp Metastasis 30:903–918
Kim S, Takahashi H, Lin WW, Descargues P, Grivennikov S, Kim Y, Luo JL, Karin M (2009) Carcinoma produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 457:102–106
Khatib AM, Auguste P, Fallavollita L, Wang N, Samani A, Kontogiannea M, Meterissian S, Brodt P (2005) Characterization of the host inflammatory response to tumor cells during the initial stages of liver metastasis. Am J Pathol 167:749–759
Hingorani SR, Wang L, Multani AS, Combs C, Deramaudt TB, Hruban RH, Rustgi AK, Chang S, Tuveson DA (2005) Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 7:469–483
Hermann PC, Sancho P, Canamero M, Martinelli P, Madriles F, Michl P, Gress T, de Pascual R, Gandia L, Guerra C et al (2014) Nicotine promotes initiation and progression of KRAS-induced pancreatic cancer via Gata6-dependent dedifferentiation of acinar cells in mice. Gastroenterology 147:1119–1133
Kowanetz M, Wu X, Lee J, Tan M, Hagenbeek T, Qu X, Yu L, Ross J, Korsisaari N, Cao T et al (2010) Granulocyte-colony stimulating factor promotes lung metastasis through mobilization of Ly6G + Ly6C+ granulocytes. Proc Natl Acad Sci 107:21248–21255
Ilkovitch D, Lopez DM (2009) The liver is a site for tumor-induced myeloid-derived suppressor cell accumulation and immunosuppression. Cancer Res 69:5514–5521
Connolly MK, Mallen-St Clair J, Bedrosian AS, Malhotra A, Vera V, Ibrahim J, Henning J, Pachter HL, Bar-Sagi D, Frey AB et al (2010) Distinct populations of metastases-enabling myeloid cells expand in the liver of mice harboring invasive and preinvasive intra-abdominal tumor. Leukoc Biol 87:713–725
Kapanadze T, Gamrekelashvili J, Ma C, Chan C, Zhao F, Hewitt S, Zender L, Kapoor V, Felsher DW, Manns MP et al (2013) Regulation of accumulation and function of myeloid derived suppressor cells in different murine models of hepatocellular carcinoma. J Hepatol 59:1007–1013
Eggert T, Medina-Echeverz J, Kapanadze T, Kruhlak MJ, Korangy F, Greten TF (2014) Tumor induced hepatic myeloid derived suppressor cells can cause moderate liver damage. PLoS One 9, e112717
Li H, Fan X, Stoicov C, Liu JH, Zubair S, Tsai E, Ste Marie R, Wang TC, Lyle S, Kurt-Jones E et al (2009) Human and mouse colon cancer utilizes CD95 signaling for local growth and metastatic spread to liver. Gastroenterology 137:934–944
Ballas ZK, Buchta CM, Rosean TR, Heusel JW, Shey MR (2013) Role of NK cell subsets in organ-specific murine melanoma metastasis. PLoS One 8, e65599
Sato S, Hanibuchi M, Kuramoto T, Yamamori N, Goto H, Ogawa H, Mitsuhashi A, Van TT, Kakiuchi S et al (2013) Macrophage stimulating protein promotes liver metastases of small cell lung cancer cells by affecting the organ microenvironment. Clin Exp Metastasis 30:333–344
Park YJ, Song B, Kim YS, Kim EK, Lee JM, Lee GE, Kim JO, Kim YJ, Chang WS, Kang CY (2013) Tumor microenvironmental conversion of natural killer cells into myeloid-derived suppressor cells. Cancer Res 73:5669–5681
Sato Y, Shimzu K, Shinga J, Hidaka M, Kawano F, Kakimi K, Yamasaki S, Asakura M, Fujii SI (2015) Characterization of the myeloid-derived suppressor cell subset regulated by NK cells in malignant lymphoma. Oncoimmunology 4, e995541
Ling X, Spaeth E, Chen Y, Shi Y, Zhang W, Schober W, Hail N, Konopleva M, Andreeff M (2013) The CXCR4 antagonist AMD3465 regulates oncogenic signalling and invasiveness in vitro and prevents breast cancer growth and metastasis in vivo. PLoS One 8, e58426
Seubert B, Grünwald B, Kobuch J, Cui H, Schelter F, Schaten S, Siveke JT, Lim NH, Nagase H, Simonavicius N et al (2015) TIMP1 creates a pre-metastatic niche in the liver through SDF/CXCR4-dependent neutrophil recruitment in mice. Hepatology 61:238–248
Sanford DE, Belt BA, Panni RZ, Mayer A, Deshpande AD, Carpenter D, Mitchem JB, Plambeck-Suess SM, Worley LA, Goetz BD et al (2013) Inflammatory monocyte mobilization decreases patient survival in pancreatic cancer: a role for targeting the CCL2/CCR2 axis. Clin Cancer Res 19:3404–3415
Würtz SØ, Møller S, Mouridsen H, Hertel PB, Friis E, Brünner N (2008) Plasma and serum levels of tissue inhibitor of metalloproteinases-1 are associated with prognosis in node-negative breast cancer: a prospective study. Mol Cell Proteomics 7:424–430
Holten-Andersen MN, Nielsen HJ, Sørensen S, Jensen V, Brünner N, Christensen IJ (2006) Tissue inhibitor of metalloproteinases-1 in the postoperative monitoring of colorectal cancer. Eur J Cancer 42:1889–1896
Pesta M, Kulda V, Kucera R, Pesek M, Vrzalova J, Liska V, Pecen L, Treska V, Safranek J, Prazakova M et al (2011) Prognostic significance of TIMP-1 in non-small cell lung cancer. Anticancer Res 31:4031–4038
Pennanen H, Kuittinen O, Soini Y, Turpeenniemi-Hujanen T (2004) Clinicopathological correlations of TIMP-1 and TIMP-2 in Hodgkin’s lymphoma. Eur J Haematol 72:1–9
Bunatova K, Pesta M, Kulda V, Topolcan O, Vrzalova J, Sutnar A, Treska V, Pecen L, Liska V (2012) Plasma TIMP1 level is a prognostic factor in patients with liver metastases. Anticancer Res 32:4601–4606
Chirco R, Liu XW, Jung KK, Kim HR (2006) Novel functions of TIMPs in cell signalling. Cancer Metastasis Rev 25:99–113
Lee SJ, Yoo HJ, Bae YS, Kim HJ, Lee ST (2003) TIMP-1 inhibits apoptosis in breast carcinoma cells via a pathway involving pertussis toxin-sensitive G protein and c-Src. Biochem Biophys Res Commun 312:1196–1201
Porter JF, Shen S, Denhardt D (2004) Tissue inhibitor of metalloproteinase-1 stimulates proliferation of human cancer cells by inhibiting a metalloproteinase. Br J Cancer 90:463–470
Egea V, Zahler S, Rieth N, Neth P, Popp T, Kehe K, Jochum M, Ries C (2012) Tissue inhibitor of metalloproteinase-1 (TIMP-1) regulates mesenchymal stem cells through let-7f microRNA and Wnt/β-catenin signaling. Proc Natl Acad Sci U S A 109:E309–E316
Cui H, Seubert B, Stahl E, Dietz H, Reuning U, Moreno-Leon L, Ilie M, Hofman P, Nagase H, Mari B et al (2014) Tissue inhibitor of metalloproteinase-1 induces a pro-tumorigenic increase of miR-210 in lung adenocarcinoma cells and their exosomes. Oncogene. doi:10.1038/onc.2014.300
Schelter F, Grandl M, Seubert B, Schaten S, Hauser S, Gerg M, Boccaccio C, Comoglio P, Krüger A (2011) Tumor cell-derived Timp-1 is necessary for maintaining metastasis-promoting Met-signaling via inhibition of Adam-10. Clin Exp Metastasis 28:793–802
Kopitz C, Gerg M, Bandapalli OR, Ister D, Pennington CJ, Hauser S, Flechsig C, Krell HW, Antolovic D, Brew K et al (2007) Tissue inhibitor of metalloproteinases-1 promotes liver metastasis by induction of hepatocyte growth factor signalling. Cancer Res 67:8615–8623
Schrötzlmair F, Kopitz C, Halbgewachs B, Lu F, Algül H, Brünner N, Gänsbacher B, Krüger A (2010) Tissue inhibitor of metalloproteinases-1-induced scattered liver metastasis is mediated by host-derived urokinase-type plasminogen activator. J Cell Mol Med 14:2760–2770
Kobuch J, Cui H, Grünwald B, Saftig P, Knolle PA, Krüger A (2015) TIMP-1 signaling via CD63 triggers granulopoiesis and neutrophilia in mice. Haematologica 100:1005–1013
Yamamoto M, Kikuchi H, Ohta M, Kawabata T, Hiramatsu Y, Kondo K, Baba M, Kamiya K, Tanaka T, Kitagawa M et al (2008) TSU68 prevents liver metastasis of colon cancer xenografts by modulating the premetastatic niche. Cancer Res 68:9754–9762
Medina-Echeverz J, Fioravanti J, Díaz-Valdés N, Frank K, Aranda F, Gomar C, Ardaiz N, Dotor J, Umansky V, Prieto J et al (2014) Harnessing high density lipoproteins to block transforming growth factor beta and to inhibit the growth of liver tumor metastases. PLoS One 9, e96799
Pan S, Chen R, Crispin DA, May D, Stevens T, McIntosh MW, Bronner MP, Ziogas A, Anton-Culver H, Brentnall TA (2011) Protein alterations associated with pancreatic cancer and chronic pancreatitis found in human plasma using global quantitative proteomics profiling. J Proteome Res 10:2359–2376
Pan S, Chen R, Brand RE, Hawley S, Tamura Y, Gafken PR, Milless BP, Goodlett DR, Rush J, Brentnall TA (2012) A multiplex targeted proteomic assay for biomarker detection in plasma: a pancreatic cancer biomarker case study. J Proteome Res 11:1937–1948
Pan S, Brentnall TA, Chen R (2015) Proteomics analysis of bodily fluids in pancreatic cancer. Proteomics. doi:10.1002/pmic.201400476
Acknowledgment
This work was supported by grants from Deutsche Forschungsgemeinschaft (KR2047 1–2 and KR2047 1–3). Ulrike Ludwig is acknowledged for the support in the artwork.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krüger, A. Premetastatic niche formation in the liver: emerging mechanisms and mouse models. J Mol Med 93, 1193–1201 (2015). https://doi.org/10.1007/s00109-015-1342-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1342-7