Abstract
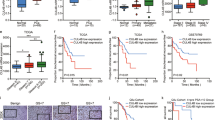
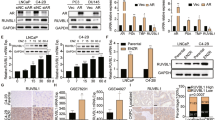
Thalidomide is experimentally used to treat various human cancers; however, clinical responses to thalidomide are sporadic. Here we demonstrate that CUL4A plays an oncogenic role in prostate cancer development and prostate cancer cells with higher level of CUL4A are particularly sensitive to thalidomide treatment. We show that CUL4A is frequently overexpressed in human primary prostate cancer and cell lines. Notably, subjects with tumors that highly expressed CUL4A had poor overall survival. CUL4A downregulation inhibited cell proliferation and induced apoptosis in vitro and in vivo, whereas CUL4A overexpression transformed human normal prostate epithelial cells and promoted invasion, which was attenuated by the extracellular signal-regulated kinase (ERK) inhibitor. We further show that the sensitivity to thalidomide is positively correlated with CUL4A expression in a panel of prostate cell lines. Ectopic CUL4A expression greatly enhanced sensitivity to thalidomide, while its downregulation conferred resistance to this drug. Mechanistically, thalidomide decreased CUL4A in a time- and dose-dependent manner, consequently leading to inaction of ERK pathway. Finally, we show that cereblon level is correlated with CUL4A expression and downregulated in thalidomide-resistant prostate cancer cell. Our results offer the first evidence that CUL4A is a potential therapeutic target for prostate cancer and may serve as a biomarker for assessing prognosis of human prostate cancer and response to thalidomide treatment.








Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Shen MM, Abate-Shen C (2010) Molecular genetics of prostate cancer: new prospects for old challenges. Genes Dev 24:1967–2000
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, Oudard S, Theodore C, James ND, Turesson I et al (2004) Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 351:1502–1512
Petrylak DP, Tangen CM, Hussain MH, Lara PN Jr, Jones JA, Taplin ME, Burch PA, Berry D, Moinpour C, Kohli M et al (2004) Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med 351:1513–1520
Bartlett JB, Dredge K, Dalgleish AG (2004) The evolution of thalidomide and its IMiD derivatives as anticancer agents. Nat Rev Cancer 4:314–322
Quach H, Ritchie D, Stewart AK, Neeson P, Harrison S, Smyth MJ, Prince HM (2010) Mechanism of action of immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia 24:22–32
Dauguet N, Fournie JJ, Poupot R, Poupot M (2010) Lenalidomide down regulates the production of interferon-gamma and the expression of inhibitory cytotoxic receptors of human natural killer cells. Cell Immunol 264:163–170
Hurteau JA, Brady MF, Darcy KM, McGuire WP, Edmonds P, Pearl ML, Ivanov I, Tewari KS, Mannel RS, Zanotti K et al (2010) Randomized phase III trial of tamoxifen versus thalidomide in women with biochemical-recurrent-only epithelial ovarian, fallopian tube or primary peritoneal carcinoma after a complete response to first-line platinum/taxane chemotherapy with an evaluation of serum vascular endothelial growth factor (VEGF): a Gynecologic Oncology Group Study. Gynecol Oncol 119:444–450
Reiriz AB, Richter MF, Fernandes S, Cancela AI, Costa TD, Di Leone LP, Schwartsmann G (2004) Phase II study of thalidomide in patients with metastatic malignant melanoma. Melanoma Res 14:527–531
Margulis V, Matin SF, Tannir N, Tamboli P, Shen Y, Lozano M, Swanson DA, Jonasch E, Wood CG (2009) Randomized trial of adjuvant thalidomide versus observation in patients with completely resected high-risk renal cell carcinoma. Urology 73:337–341
Dal Lago L, Richter MF, Cancela AI, Fernandes SA, Jung KT, Rodrigues AC, Costa TD, Di Leone LP, Schwartsmann G (2003) Phase II trial and pharmacokinetic study of thalidomide in patients with metastatic colorectal cancer. Invest New Drugs 21:359–366
Burris HA 3rd, Jones SF, Shipley D, Meluch AA, Greco FA, Barton JH, Yardley DA, Hainsworth JD (2010) Phase II study of capecitabine in combination with thalidomide in patients with metastatic breast cancer. Cancer Invest 28:408–412
Lee SM, Rudd R, Woll PJ, Ottensmeier C, Gilligan D, Price A, Spiro S, Gower N, Jitlal M, Hackshaw A (2009) Randomized double-blind placebo-controlled trial of thalidomide in combination with gemcitabine and carboplatin in advanced non-small-cell lung cancer. J Clin Oncol 27:5248–5254
Pacheco AV, Rasila K, Lee SJ, Rabinowitz I, Elias L, Lee FC, Verschraegen CF (2008) Phase II studies of antiangiogenic four drug regimens for the treatment of advanced renal cell carcinoma: FUNIL-retinoid and the FUNIL-thalidomide protocols. Urol Oncol 26:610–615
Boyd KD, Walker BA, Wardell CP, Ross FM, Gregory WM, Davies FE, Morgan GJ (2010) High expression levels of the mammalian target of rapamycin inhibitor DEPTOR are predictive of response to thalidomide in myeloma. Leukemia Lymphoma 51:2126–2129
Bagratuni T, Wu P, Gonzalez de Castro D, Davenport EL, Dickens NJ, Walker BA, Boyd K, Johnson DC, Gregory W, Morgan GJ et al (2010) XBP1s levels are implicated in the biology and outcome of myeloma mediating different clinical outcomes to thalidomide-based treatments. Blood 116:250–253
Brenne AT, Romstad LH, Gimsing P, Juliusson G, Turesson I, Romundstad P, Borset M, Sundan A, Waage A (2004) A low serum level of soluble tumor necrosis factor receptor p55 predicts response to thalidomide in advanced multiple myeloma. Haematologica 89:552–556
Nishitani H, Shiomi Y, Iida H, Michishita M, Takami T, Tsurimoto T (2008) CDK inhibitor p21 is degraded by a proliferating cell nuclear antigen-coupled Cul4-DDB1Cdt2 pathway during S phase and after UV irradiation. J Biol Chem 283:29045–29052
Li B, Jia N, Kapur R, Chun KT (2006) Cul4A targets p27 for degradation and regulates proliferation, cell cycle exit, and differentiation during erythropoiesis. Blood 107:4291–4299
Li J, Wang QE, Zhu Q, El-Mahdy MA, Wani G, Praetorius-Ibba M, Wani AA (2006) DNA damage binding protein component DDB1 participates in nucleotide excision repair through DDB2 DNA-binding and cullin 4A ubiquitin ligase activity. Cancer Res 66:8590–8597
Nag A, Bagchi S, Raychaudhuri P (2004) Cul4A physically associates with MDM2 and participates in the proteolysis of p53. Cancer Res 64:8152–8155
Chen LC, Manjeshwar S, Lu Y, Moore D, Ljung BM, Kuo WL, Dairkee SH, Wernick M, Collins C, Smith HS (1998) The human homologue for the Caenorhabditis elegans cul-4 gene is amplified and overexpressed in primary breast cancers. Cancer Res 58:3677–3683
Melchor L, Saucedo-Cuevas LP, Munoz-Repeto I, Rodriguez-Pinilla SM, Honrado E, Campoverde A, Palacios J, Nathanson KL, Garcia MJ, Benitez J (2009) Comprehensive characterization of the DNA amplification at 13q34 in human breast cancer reveals TFDP1 and CUL4A as likely candidate target genes. Breast Cancer Res 11:R86
Shinomiya T, Mori T, Ariyama Y, Sakabe T, Fukuda Y, Murakami Y, Nakamura Y, Inazawa J (1999) Comparative genomic hybridization of squamous cell carcinoma of the esophagus: the possible involvement of the DPI gene in the 13q34 amplicon. Genes Chromosomes Cancer 24:337–344
Dohna M, Reincke M, Mincheva A, Allolio B, Solinas-Toldo S, Lichter P (2000) Adrenocortical carcinoma is characterized by a high frequency of chromosomal gains and high-level amplifications. Gene Chromosome Canc 28:145–152
Michiels EMC, Weiss MM, Hoovers JMN, Baak JPA, Voute PA, Baas F, Hermsen MAJA (2002) Genetic alterations in childhood medulloblastoma analyzed by comparative genomic hybridization. J Pediat Hematol Onc 24:205–210
Yasui K, Arii S, Zhao C, Imoto I, Ueda M, Nagai H, Emi M, Inazawa J (2002) TFDP1, CUL4A, and CDC16 identified as targets for amplification at 13q34 in hepatocellular carcinomas. Hepatology 35:1476–1484
Schindl M, Gnant M, Schoppmann SF, Horvat R, Birner P (2007) Overexpression of the human homologue for Caenorhabditis elegans cul-4 gene is associated with poor outcome in node-negative breast cancer. Anticancer Res 27:949–952
Hung MS, Mao JH, Xu ZD, Yang CT, Yu JS, Harvard C, Lin YC, Bravo DT, Jablons DM, You LA (2011) Cul4A is an oncogene in malignant pleural mesothelioma. J Cell Mol Med 15:350–358
Paris PL, Andaya A, Fridlyand J, Jain AN, Weinberg V, Kowbel D, Brebner JH, Simko J, Watson JE, Volik S et al (2004) Whole genome scanning identifies genotypes associated with recurrence and metastasis in prostate tumors. Hum Mol Genet 13:1303–1313
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y, Yamaguchi Y, Handa H (2010) Identification of a primary target of thalidomide teratogenicity. Science 327:1345–1350
Bello D, Webber MM, Kleinman HK, Wartinger DD, Rhim JS (1997) Androgen responsive adult human prostatic epithelial cell lines immortalized by human papillomavirus 18. Carcinogenesis 18:1215–1223
Wang H, Zhai L, Xu J, Joo HY, Jackson S, Erdjument-Bromage H, Tempst P, Xiong Y, Zhang Y (2006) Histone H3 and H4 ubiquitylation by the CUL4-DDB-ROC1 ubiquitin ligase facilitates cellular response to DNA damage. Mol Cell 22: 383–394
Zhu YX, Braggio E, Shi CX, Bruins LA, Schmidt JE, Van Wier S, Chang XB, Bjorklund CC, Fonseca R, Bergsagel PL et al (2011) Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood 118:4771–4779
Higa LA, Wu M, Ye T, Kobayashi R, Sun H, Zhang H (2006) CUL4-DDB1 ubiquitin ligase interacts with multiple WD40-repeat proteins and regulates histone methylation. Nat Cell Biol 8:1277–1283
Acknowledgments
This work was supported by funding from the National Basic Research Program of China (no. 2012CB518300, YHS), the National Natural Science Foundation of China (no. 81101946, SCR), Key Basic Research Programme of Shanghai (no. 09DJ1400400, YHS), and the National Institutes of Health, National Cancer Institute grant R01 CA116481, Office of Biological & Environmental Research, of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231, and Laboratory Directed Research & Development Program (LDRD, JHM).
Conflict of interest
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors Shancheng Ren, Chuanliang Xu, and Zilian Cui contributed equally to this work.
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 540 kb)
Rights and permissions
About this article
Cite this article
Ren, S., Xu, C., Cui, Z. et al. Oncogenic CUL4A determines the response to thalidomide treatment in prostate cancer. J Mol Med 90, 1121–1132 (2012). https://doi.org/10.1007/s00109-012-0885-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-012-0885-0