Abstract
Introduction
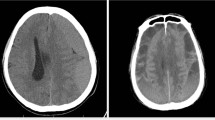
Chronic subdural haematoma (CSH) has multifactorial mechanisms involved in its development and progression. Identifying readily available inflammatory and coagulation indices that can predict the prognosis of CSH will help in clinical care, prognosis, generating objective criteria for assessing efficacy of treatment strategies and comparisons of treatment efficacy between clinical studies. We conducted a study in which we evaluated the impact value of neutrophil to lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), inflammatory biomarkers (erythrocyte sedimentation rate and C-reactive protein), activated partial thromboplastin time (APTT), prothrombin time (PT) and international normalized ratio (INR) at presentation on CSH severity and outcome using Glasgow outcome scale (GOS), Markwalder grading scale (MGS) and Lagos brain disability examination scale (LABDES).
Methods
We prospectively studied patients in a single healthcare system with clinical and radiological features of chronic subdural haematoma. Only patients who were managed surgically were recruited. The primary outcome was predictors of severe form CSH (using admission Glasgow coma scale score, MGS grade, inflammatory cells and biomarkers, APTT, PT, and INR) and outcome (using GOS, MGS and LABDES grades 3 months) with secondary outcome being mortality rate, 3 months following surgery. Good outcome was defined as GOS score > 3, LABDES score ≥ 40 at three months and MGS score < 2. Differences in categorical and continuous variables between groups were compared using Fisher’s exact test or Chi-square test (χ2) analysis, one-way ANOVA or Kruskal–Wallis test (in case of non-normal distribution).
Results
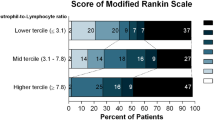
We included 61 patients with a male-to-female ratio of 2.6:1 and mean age of 57.5 ± 13.3 years (median 58 years; 27–83 years). The pre-op MGS grade was significantly associated with the LABDES (p = 0.034), GOS (p = 0.011) and post-op MGS (p = 0.007) grade. All the patients that died had elevated APTT and PT with low PLR. A low admission PLR was significantly associated with a poor outcome using the GOS (p = 0.001), MGS (p = 0.011) and LABDES grade (p = 0.006) (Table 3). A high APTT was also significantly associated with a worse outcome using GOS (p = 0.007), MGS (p = 0.007) and LABDES grade (p = 0.003). There were three (4.9%) deaths with post-mortem diagnosis of pulmonary embolism, chronic renal failure and irreversible craniocaudal herniation syndrome. All the patients that died had elevated APTT and PT with low PLR.
Conclusion
Patients’ admission APTT, PT, INR and PLR are good predictors of outcome using the GOS. A high admission INR is also associated with a worse outcome using MGS and LABDES grade.

Similar content being viewed by others
References
Stoodley M, Weir B. Contents of chronic subdural hematoma. NeurosurgClin N Am. 2000;11(3):425–34.
Aronowski J, Zhao X. Molecular pathophysiology of cerebral hemorrhage: Secondary brain injury. Stroke. 2011;42:1781–6.
Wang J, Dore S. Inflammation after intracerebral hemorrhage. J Cereb Blood Flow Metab. 2007;27:894–908.
Silva Y, Leira R, Tejada J, Lainez JM, Castillo J, Dávalos A. Molecular signatures of vascular injury are associated with early growth of intracerebral hemorrhage. Stroke. 2005;36:86–91.
Liu X, Shen Y, Wang H, Ge Q, Fei A, Pan S. Prognostic significance of neutrophil-to lymphocyte ratio in patients with sepsis: a prospective observational study. Mediators Inflamm. 2016;2016:8191254.
Hwang SY, Shin TG, Jo IJ, Jeon K, Suh GY, Lee TR, Yoon H, Cha WC, Sim MS. Neutrophil-to-lymphocyte ratio as a prognostic marker in critically-ill septic patients. Am J Emerg Med. 2017;35(2):234–9.
Riché F, Gayat E, Barthélémy R, Le Dorze M, Matéo J, Payen D. Reversal of neutrophil-to-lymphocyte count ratio in early versus late death from septic shock. Crit Care. 2015;19(1):439.
Sato S, Suzuki J. Ultrastructural observations of the capsule of chronic subdural haematoma in various clinical stages. J Neurosurg. 1975;43:569–78.
Xue P, Kanai M, Mori Y, Nishimura T, Uza N, Kodama Y, Kawaguchi Y, Takaori K, Matsumoto S, Uemoto S, Chiba T. Neutrophil-to-lymphocyte ratio for predicting palliative chemotherapy outcomes in advanced pancreatic cancer patients. Cancer Med. 2014;3:406–15.
Wang X, Zhang G, Jiang X, Zhu H, Lu Z, Xu L. Neutrophil to lymphocyte ratio in relation to risk of all-cause mortality and cardiovascular events among patients undergoing angiography or cardiac revascularization: a meta-analysis of observational studies. Atherosclerosis. 2014;234:206–13.
Kundi H, Balun A, Cicekcioglu H, Cetin M, Kiziltunc E, Cetin ZG, Mansuroglu C, Ornek E. The relation between platelet-to-lymphocyte ratio and Pulmonary Embolism Severity Index in acute pulmonary embolism. Heart Lung. 2015;44:340–3.
Ozcan Cetin EH, Cetin MS, Aras D, Topaloglu S, Temizhan A, Kisacik HL, Aydogdu S. Platelet to lymphocyte ratio as a prognostic marker of in-hospital and long-term major adverse cardiovascular events in ST-segment elevation myocardial infarction. Angiology. 2016;67:336–45.
Yang W, Liu Y. Platelet-lymphocyte ratio is a predictor of venous thromboembolism in cancer patients. Thromb Res. 2015;136:212–5.
Tokgoz S, Kayrak M, Akpinar Z, Seyithanoglu A, Guney F, Yuruten B. Neutrophil lymphocyte ratio as a predictor of stroke. J Stroke Cerebrovasc Dis. 2013;22:1169–74.
Wang F, Hu S, Ding Y, et al. Neutrophil-to-lymphocyte ratio and 30-day mortality in patients with acute intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2016;25:182–7.
Idowu OE, Oshinaike O, Ogun SA, Arabambi B, Orungbeja TO, Malomo OO, Aisudo CK, Oyeleye OO. Predicting outcome in traumatic brain injury using a new outcome assessment scale—The Lagos Brain Disability Examination Scale (LABDES). Trauma. 2019;22(2):148–54.
Chen JCT, Levy ML. Causes, epidemiology, and risk factors of chronic subdural hematoma. Neurosurg Clin N Am. 2000;11(3):399–406.
Sajanti J, Majamaa K. High concentrations of procollagen propeptides in chronic subdural haematoma and effusion. J Neurol Neurosurg Psychiatry. 2003;74:522.
Ito H, Yamamoto S, Saito K, Ikeda K, Hisada K. Quantitative estimation of haemorrhage in chronic subdural haematoma using the 51Cr erythrocyte labelling method. J Neurosurg. 1987;66:862–4.
Traynelis VC. Chronic subdural haematoma in the elderly. Clin Geriatr Med. 1991;7:583–98.
Sambasivan M. An overview of chronic subdural hematoma: experience with 2300 cases. Surg Neurol. 1997;47:418–22.
Hohenstein A, Erber R, Schilling L, Weigel R. Increased mRNA expression of VEGF within the hematoma and imbalance of angiopoietin-1 and -2 mRNA within the neomembranes of chronic subdural hematoma. J Neurotrauma. 2005;22(5):518–28.
Hara M, Tamaki M, Aoyagi M, et al. Possible role of cyclooxygenase-2 in developing chronic subdural haematoma. J Med Dent Sci. 2009;56:101–6.
Shono T, Inamura T, Morioka T. Vascular endothelial growth factor in chronic subdural haematomas. J Clin Neurosci. 2001;8(5):411–5.
Nanko N, Tanikawa M, Mase M, Fujita M, Tateyama H, Miyati T, Yamada K. Involvement of hypoxia-induciblefactor-1a and VEGF in the mechanism and development of chronic subdural haematoma. Neurol Med Chir (Tokyo). 2009;49(9):379–85.
Kalamatianos T, Stavrinou LC, Koutsarnakis C, Psachoulia C, Sakas DE, Stranjalis G. PlGF and sVEGFR-1 in chronic subdural hematoma: implications for hematoma development. J Neurosurg. 2013;118(2):353–7.
Hua C, Zhao G, Feng Y, Yuan H, Song H, Bie L, Ogawa A. Role of matrix metalloproteinase-2, matrix metalloproteinase-9, and vascular endothelial growth factor in the development of chronic subdural hematoma. J Neurotrauma. 2016;33(1):65–70.
Vaquero J, Zurita M, Cincu R. Vascular endothelial growth-permeability factor in granulation tissue of chronic subdural haematomas. Acta Neurochir (Wien). 2002;144(4):343–6.
Jung S, Moon KS, Jung TY, Kim IY, Lee YH, Rhu HH, Sun HS, Jeong YI, Kim K, Kang S. Possible pathophysiological role of vascular endothelial growth factor (VEGF) and matrix metalloproteinases (MMPs) in metastatic brain tumor-associated intracerebral hemorrhage. J Neurooncol. 2006;76(3):257–63.
Hong HJ, Kim YJ, Yi HJ, Yi H, Ko Y, Oh S, Kim J. Role of angiogenic growth factors and inflammatory cytokine on recurrence of chronic subdural hematoma. Surg Neurol. 2009;71(2):161–5.
Stanisic M, Lyngstadaas SP, Pripp AH, Aasen AO, Lindegaard KF, Ivanovic J, Ilstad E, Konglund A, Sandell T, Ellingsen O, Saehle T. Chemokines as markers of local inflammation and angiogenesis in patients with chronic subdural haematoma: a prospective study. Acta Neurochir. 2012;154:113–20.
Kitazono M, Yokota H, Satoh H, Onda H, Matsumoto G, Fuse A, Teramoto A. Measurement of inflammatory cytokines and thrombomodulin in chronic subdural hematoma. Neurol Med Chir (Tokyo). 2012;52(11):810–5.
Wada T, Kuroda K, Yoshida Y, Ogasawara K, Ogawa A, Endo S. Local elevation of the anti-inflammatory interleukin-10 in the pathogenesis of chronic subdural hematoma. Neurosurg Rev. 2006;29(3):242–5.
Kitazono M, Yokota H, Satoh H, Onda H, Matsumoto G, Fuse A, Akira T. Measurement of inflammatory cytokines and thrombomodulin in chronic subdural hematoma. Neurol Med Chir. 2012;52(11):810–5.
Balser D, Farooq S, Mehmood T, Quigley MR. Actual and projected incidence rates for chronic subdural hematomas in United States Veterans Administration and civilian populations. J Neurosurg. 2015;123:1209–15.
Miranda LB, Braxton E, Hobbs J, et al. Chronic subdural hematoma in the elderly: not a benign disease. J Neurosurg. 2011;114:72–6.
Ramachandran R, Hegde T. Chronic subdural hematomas–causes of morbidity and mortality. Surg Neurol. 2007;67:367–72.
Waqas M, Vakhari K, Weimer PV, Hashmi E, Davies JM, Siddiqui AH. Safety and effectiveness of embolization for chronic subdural hematoma: systematic review and case series. World Neurosurg. 2019;126:228–36.
Wakai S, Hashimoto K, Watanabe N, Inoh S, Ochiai C, Nagai M. Efficacy of closed system drainage in treating chronic subdural hematoma: a prospective comparative study. Neurosurgery. 1990;26:771–3.
Hellwig D, Kuhn TJ, Bauer BL, List-Hellwig E. Endoscopic treatment of septated chronic subdural hematoma. Surg Neurol. 1996;45:272–7.
Smely C, Madlinger A, Scheremet R. Chronic subdural hematoma—a comparison of two different treatment modalities. Acta Neurochir. 1997;139:818–25.
Yoshimoto Y, Kwak S. Frontal small craniostomy and irrigation for treatment of chronic subdural haematoma. Br J Neurosurg. 1997;11:150–1.
Tschoeke SK, Ertel W. Immunoparalysis after multiple trauma. Injury. 2007;38(12):1346–57.
Xu CS, Lu M, Liu LY, Yao MY, Cheng GL, Tian XY, Xiao F, Wan Q, Chen F. Chronic subdural hematoma management: clarifying the definitions of outcome measures to better understand treatment efficacy—a systematic review and metaanalysis. Eur Rev Med Pharmacol Sci. 2017;21:809–18.
Gernsback J, Kolcun JP, Jagid J. To drain or two drains: recurrences in chronic subdural hematomas. World Neurosurg. 2016;95:447–50.
Ban SP, Hwang G, Byoun HS, et al. Middle meningeal artery embolization for chronic subdural hematoma. Radiology. 2018;286:992–9.
Author information
Authors and Affiliations
Contributions
OEI contributed to the conceptualization, study design, data collection, data curation, writing and editing of the manuscript and is the primary writer of the article. SOO and JMV contributed to the study design, data collection, review and revision of the manuscript, and coordination of the study.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflict of interest, financial or otherwise.
Rights and permissions
About this article
Cite this article
Idowu, O.E., Oyeleke, S.O. & Vitowanu, J.M. Impact of inflammatory cell ratio, biomarkers, activated partial thromboplastin time and prothrombin time on chronic subdural haematoma severity and outcome. Eur J Trauma Emerg Surg 48, 1085–1092 (2022). https://doi.org/10.1007/s00068-021-01665-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-021-01665-5