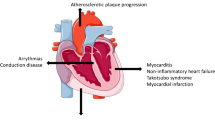
Abstract
Immune checkpoint inhibitor (ICI) therapy induces an immune response against cancer cells. Immune checkpoint inhibitor therapy has tremendously improved the prognosis for a large number of cancers, but is associated with considerable immune-related adverse events (irAEs). Cardiovascular complications from ICI therapy occur in a modest proportion of patients, but show the highest lethality rates of all ICI-related complications. While ICI-related myocarditis is the most dangerous complication, its clinical manifestation varies, e.g., asymptomatic reduction of left ventricular function, isolated increase in cardiac troponins, and arrhythmias. This review delineates current data on cardiovascular complications of ICI therapy. The effects of ICI therapy on the cardiovascular system are classified in the context of preclinical data on the biochemical and immunological function of the immune checkpoint signaling pathways in the heart and the vascular system. Incidence, suspected pathomechanisms, typical symptoms, as well as recommended diagnostics are summarized. Current therapy recommendations for ICI-related cardiotoxicity are outlined and innovative new approaches with high potential for improving outcome in ICI-related myocarditis are delineated. A better understanding of cardiovascular complications is essential for the best possible oncocardiology care of the growing number of patients undergoing ICI therapy.
Zusammenfassung
Das Ziel der Immuncheckpoint-Inhibitor(ICI)-Therapie ist die Induktion einer Immunreaktion gegen Krebszellen. Die ICI-Therapie hat bei einer Vielzahl von Tumorerkrankungen zu einer deutlich verbesserten Prognose geführt, ist jedoch mit erheblichen immunvermittelten Nebenwirkungen assoziiert. Kardiovaskuläre Komplikationen der ICI-Therapie treten bei einem geringen Anteil von Patienten auf, zeigen jedoch die höchste Letalität aller immunvermittelten Nebenwirkungen. Während die fulminante ICI-assoziierte Myokarditis die gefährlichste Komplikation darstellt, zeigen aktuelle Daten auch eine relevante Prävalenz von weiteren Nebenwirkungen, wie einer asymptomatischen Einschränkung der linksventrikulären Funktion, einer isolierten Erhöhung des kardialen Troponins oder einer bradykarden und tachykarden Herzrhythmusstörung. Diese Übersichtsarbeit behandelt die aktuelle Datenlage zu kardiovaskulären Komplikationen der ICI-Therapie. Es erfolgt eine Zusammenfassung von Inzidenz, vermutetem Pathomechanismus, typischen Symptomen sowie empfohlener Diagnostik und Therapieoptionen. Darüber hinaus werden die Effekte einer ICI-Therapie auf das kardiovaskuläre System im Kontext der verfügbaren präklinischen Daten zur biochemischen und immunologischen Funktion der Immuncheckpoint-Signalwege im Herzen und in Gefäßen eingeordnet. Aktuelle Therapieempfehlungen zur ICI-bedingten Kardiotoxizität werden dargestellt und innovative neue Ansätze mit hohem Potenzial für eine Verbesserung der Behandlung zusammengefasst. Das bessere Verständnis von kardiovaskulären Komplikationen ist essenziell für die bestmögliche onkokardiologische Betreuung des wachsenden Kollektivs von Patienten unter ICI-Therapie.

Similar content being viewed by others
References
Sharpe AH, Pauken KE (2018) The diverse functions of the PD1 inhibitory pathway. Nat Rev Immunol 18(3):153–167. https://doi.org/10.1038/nri.2017.108
Buchbinder EI, Desai A (2016) CTLA‑4 and PD‑1 pathways: similarities, differences, and implications of their inhibition. Am J Clin Oncol 39(1):98–106. https://doi.org/10.1097/COC.0000000000000239
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. https://doi.org/10.1016/j.cell.2011.02.013
Ribas A, Wolchok JD (2018) Cancer immunotherapy using checkpoint blockade. Science 359(6382):1350. https://doi.org/10.1126/science.aar4060
Sznol M, Powderly JD, Smith DC et al (2010) Safety and antitumor activity of biweekly MDX-1106 (Anti-PD‑1, BMS-936558/ONO-4538) in patients with advanced refractory malignancies. J Clin Oncol 28(15_suppl):2506–2506. https://doi.org/10.1200/jco.2010.28.15_suppl.2506
Schadendorf D, van Akkooi ACJ, Berking C et al (2018) Melanoma. Lancet 392(10151):971–984. https://doi.org/10.1016/s0140-6736(18)31559-9
Wei SC, Levine JH, Cogdill AP et al (2017) Distinct cellular mechanisms underlie anti-CTLA‑4 and anti-PD‑1 checkpoint blockade. Cell 170(6):1120–1133.e17. https://doi.org/10.1016/j.cell.2017.07.024
Postow MA, Chesney J, Pavlick AC et al (2015) Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med 372(21):2006–2017. https://doi.org/10.1056/NEJMoa1414428
Lyon AR, Yousaf N, Battisti NML et al (2018) Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol 19(9):e447–e458. https://doi.org/10.1016/s1470-2045(18)30457-1
Weber J, Mandala M, Del Vecchio M et al (2017) Adjuvant nivolumab versus Ipilimumab in resected stage III or IV melanoma. N Engl J Med 377(19):1824–1835. https://doi.org/10.1056/NEJMoa1709030
Tang J, Yu JX, Hubbard-Lucey VM et al (2018) Trial watch: The clinical trial landscape for PD1/PDL1 immune checkpoint inhibitors. Nat Rev Drug Discov 17(12):854–855. https://doi.org/10.1038/nrd.2018.210
Grabie N, Lichtman AH, Padera R (2019) T cell checkpoint regulators in the heart. Cardiovasc Res 115(5):869–877. https://doi.org/10.1093/cvr/cvz025
Grabie N, Gotsman I, DaCosta R et al (2007) Endothelial programmed death‑1 ligand 1 (PD-L1) regulates CD8+ T‑cell mediated injury in the heart. Circulation 116(18):2062–2071. https://doi.org/10.1161/circulationaha.107.709360
Tivol EA, Borriello F, Schweitzer AN et al (1995) Loss of CTLA‑4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA‑4. Immunity 3(5):541–547
Nishimura H, Okazaki T, Tanaka Y et al (2001) Autoimmune dilated cardiomyopathy in PD‑1 receptor-deficient mice. Science 291(5502):319–322. https://doi.org/10.1126/science.291.5502.319
Okazaki T, Tanaka Y, Nishio R et al (2003) Autoantibodies against cardiac troponin I are responsible for dilated cardiomyopathy in PD-1-deficient mice. Nat Med 9(12):1477–1483. https://doi.org/10.1038/nm955
Kaya Z, Goser S, Buss SJ et al (2008) Identification of cardiac troponin I sequence motifs leading to heart failure by induction of myocardial inflammation and fibrosis. Circulation 118(20):2063–2072. https://doi.org/10.1161/CIRCULATIONAHA.108.788711
Bockstahler M, Fischer A, Goetzke CC et al (2020) Heart-specific immune responses in an animal model of autoimmunerelated myocarditis mitigated by an Immunoproteasome inhibitor and genetic ablation. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.119.043171
Wang J, Okazaki IM, Yoshida T et al (2010) PD‑1 deficiency results in the development of fatal myocarditis in MRL mice. Int Immunol 22(6):443–452. https://doi.org/10.1093/intimm/dxq026
Lucas JA, Menke J, Rabacal WA et al (2008) Programmed death ligand 1 regulates a critical checkpoint for autoimmune myocarditis and pneumonitis in MRL mice. J Immunol 181(4):2513–2521. https://doi.org/10.4049/jimmunol.181.4.2513
Waterhouse P, Penninger JM, Timms E et al (1995) Lymphoproliferative disorders with early lethality in mice deficient in Ctla‑4. Science 270(5238):985–988
Klocke K, Sakaguchi S, Holmdahl R, Wing K (2016) Induction of autoimmune disease by deletion of CTLA‑4 in mice in adulthood. Proc Natl Acad Sci U S A 113(17):E2383–E2392. https://doi.org/10.1073/pnas.1603892113
Ruppert V, Meyer T, Struwe C et al (2010) Evidence for CTLA4 as a susceptibility gene for dilated cardiomyopathy. Eur J Hum Genet 18(6):694–699. https://doi.org/10.1038/ejhg.2010.3
Tarrio ML, Grabie N, Bu DX et al (2012) PD‑1 protects against inflammation and myocyte damage in T cell-mediated myocarditis. J Immunol 188(10):4876–4884. https://doi.org/10.4049/jimmunol.1200389
Fonseca R, Salgado RM, Borges da Silva H et al (2018) Programmed cell death protein 1‑PDL1 interaction prevents heart damage in chronic trypanosoma cruzi infection. Front Immunol 9:997–997. https://doi.org/10.3389/fimmu.2018.00997
Hofmann U, Frantz S (2015) Role of lymphocytes in myocardial injury, healing, and remodeling after myocardial infarction. Circ Res 116(2):354–367. https://doi.org/10.1161/CIRCRESAHA.116.304072
Baban B, Liu JY, Qin X et al (2015) Upregulation of programmed death‑1 and its Ligand in cardiac injury models: interaction with GADD153. PLoS ONE 10(4):e124059. https://doi.org/10.1371/journal.pone.0124059
Wolchok JD, Chiarion-Sileni V, Gonzalez R et al (2017) Overall survival with combined nNivolumab and Ipilimumab in advanced melanoma. N Engl J Med 377(14):1345–1356. https://doi.org/10.1056/NEJMoa1709684
Michot JM, Bigenwald C, Champiat S et al (2016) Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer 54:139–148. https://doi.org/10.1016/j.ejca .2015.11.016
Wang DY, Salem JE, Cohen JV et al (2018) Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol 4(12):1721–1728. https://doi.org/10.1001/jamaoncol.2018.3923
Heinzerling L, Ott PA, Hodi FS et al (2016) Cardiotoxicity associated with CTLA4 and PD1 blocking immunotherapy. J Immunother Cancer 4:50. https://doi.org/10.1186/s40425-016-0152-y
Johnson DB, Balko JM, Compton ML et al (2016) Fulminant mocarditis with combination immune checkpoint blockade. N Engl J Med 375(18):1749–1755. https://doi.org/10.1056/NEJMoa1609214
Mahmood SS, Fradley MG, Cohen JV et al (2018) Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.02.037
Moslehi JJ, Salem JE, Sosman JA et al (2018) Increased reporting of fatal immune checkpoint inhibitor-associated myocarditis. Lancet 391(10124):933. https://doi.org/10.1016/s0140-6736(18)30533-6
Mahmood SS, Fradley MG, Cohen JV et al (2018) Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol 71(16):1755–1764. https://doi.org/10.1016/j.jacc.2018.02.037
Michel L, Rassaf T, Totzeck M (2019) Cardiotoxicity from immune checkpoint inhibitors. Int J Cardiol Heart Vasc 25:100420. https://doi.org/10.1016/j.ijcha.2019.100420
Palaskas N, Lopez-Mattei J, Durand JB et al (2020) Immune checkpoint inhibitor myocarditis: Pathophysiological characteristics, diagnosis, and treatment. J Am Heart Assoc 9(2):e13757. https://doi.org/10.1161/JAHA.119.013757
Michel L, Rassaf T (2019) Cardio-oncology: need for novel structures. Eur J Med Res 24(1):1. https://doi.org/10.1186/s40001-018-0359-0
Michel L, Rassaf T, Totzeck M (2018) Biomarkers for the detection of apparent and subclinical cancer therapy-related cardiotoxicity. J Thorac Dis 10(Suppl 35):S4282–S4295. https://doi.org/10.21037/jtd.2018.08.15
Awadalla M, Mahmood SS, Groarke JD et al (2020) Global longitudinal strain and cardiac events in patients with immune checkpoint inhibitor-related myocarditis. J Am Coll Cardiol 75(5):467–478. https://doi.org/10.1016/j.jacc.2019.11.049
Awadalla M, Golden DLA, Mahmood SS et al (2019) Influenza vaccination and myocarditis among patients receiving immune checkpoint inhibitors. J Immunother Cancer 7(1):53. https://doi.org/10.1186/s40425-019-0535-y
Bonaca MP, Olenchock BA, Salem J‑E et al (2019) Myocarditis in the setting of cancer therapeutics. Circulation 140(1):80–91. https://doi.org/10.1161/CIRCULATIONAHA.118.034497
Rassaf T, Totzeck M, Backs J et al (2020) Onco-Cardiology: Consensus paper of the German Cardiac Society, the German Society for Pediatric Cardiology and Congenital Heart Defects and the German Society for Hematology and Medical Oncology. Clin Res Cardiol. https://doi.org/10.1007/s00392-020-01636-7
Zhang L, Awadalla M, Mahmood SS et al (2020) Cardiovascular magnetic resonance in immune checkpoint inhibitor-associated myocarditis. Eur Heart J. https://doi.org/10.1093/eurheartj/ehaa051
Caforio AL, Pankuweit S, Arbustini E et al (2013) Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 34(33):2636–2648. https://doi.org/10.1093/eurheartj/eht210
Champion SN, Stone JR (2020) Immune checkpoint inhibitor associated myocarditis occurs in both high-grade and low-grade forms. Mod Pathol 33(1):99–108. https://doi.org/10.1038/s41379-019-0363-0
Cooper LT, Baughman KL, Feldman AM et al (2007) The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology. Circulation 116(19):2216–2233. https://doi.org/10.1161/CIRCULATIONAHA.107.186093
Gallegos C, Rottmann D, Nguyen VQ, Baldassarre LA (2019) Myocarditis with checkpoint inhibitor immunotherapy: case report of late gadolinium enhancement on cardiac magnetic resonance with pathology correlate. Eur Heart J Case Rep. https://doi.org/10.1093/ehjcr/yty149
Brahmer JR, Lacchetti C, Schneider BJ et al (2018) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 36(17):1714–1768. https://doi.org/10.1200/JCO.2017.77.6385
Ganatra S, Neilan TG (2018) Immune checkpoint inhibitor-associated myocarditis. Oncologist 23(8):879–886. https://doi.org/10.1634/theoncologist.2018-0130
Wang DY, Okoye GD, Neilan TG et al (2017) Cardiovascular toxicities associated with cancer immunotherapies. Curr Cardiol Rep 19(3):21. https://doi.org/10.1007/s11886-017-0835-0
Jain V, Bahia J, Mohebtash M, Barac A (2017) Cardiovascular complications associated with novel cancer immunotherapies. Curr Treat Options Cardiovasc Med 19(5):36. https://doi.org/10.1007/s11936-017-0532-8
Frigeri M, Meyer P, Banfi C et al (2018) Immune checkpoint inhibitor-associated myocarditis: a new challenge for cardiologists. Can J Cardiol 34(1):92.e1–92.e3. https://doi.org/10.1016/j.cjca.2017.09.025
Norwood TG, Westbrook BC, Johnson DB et al (2017) Smoldering myocarditis following immune checkpoint blockade. J Immunother Cancer 5(1):91. https://doi.org/10.1186/s40425-017-0296-4
Centanni M, Moes D, Troconiz IF et al (2019) Clinical pharmacokinetics and pharmacodynamics of immune checkpoint inhibitors. Clin Pharmacokinet 58(7):835–857. https://doi.org/10.1007/s40262-019-00748-2
Salem JE, Allenbach Y, Vozy A et al (2019) Abatacept for severe immune checkpoint inhibitor-associated myocarditis. N Engl J Med 380(24):2377–2379. https://doi.org/10.1056/NEJMc1901677
Wang H, Tian R, Gao P et al (2020) Tocilizumab for fulminant programmed death 1 inhibitor-associated myocarditis. J Thorac Oncol 15(3):e31–e32. https://doi.org/10.1016/j.jtho.2019.09.080
Esfahani K, Buhlaiga N, Thebault P et al (2019) Alemtuzumab for immune-related myocarditis due to PD‑1 therapy. N Engl J Med 380(24):2375–2376. https://doi.org/10.1056/NEJMc1903064
Dolladille C, Ederhy S, Sassier M et al (2020) Immune checkpoint inhibitor rechallenge after immune-related adverse events in patients with cancer. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2020.0726
Jain V, Mohebtash M, Rodrigo ME et al (2018) Autoimmune myocarditis caused by immune checkpoint inhibitors treated with antithymocyte globulin. J Immunother 41(7):332–335. https://doi.org/10.1097/cji.0000000000000239
Ponikowski P, Voors AA, Anker SD et al (2016) 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 18(8):891–975. https://doi.org/10.1002/ejhf.592
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
L. Michel, M. Totzeck, L. Lehmann, and D. Finke declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Rights and permissions
About this article
Cite this article
Michel, L., Totzeck, M., Lehmann, L. et al. Emerging role of immune checkpoint inhibitors and their relevance for the cardiovascular system. Herz 45, 645–651 (2020). https://doi.org/10.1007/s00059-020-04954-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-020-04954-8
Keywords
- Heart failure
- Immunosuppressive therapy
- Drug-related side effects and adverse reactions
- Myocarditis
- Onco-cardiology