Abstract
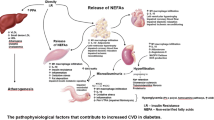
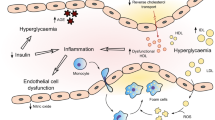
Diabetes is associated with the development of premature cardiovascular disease (CVD), which relates to the clustering of risk factors such as dyslipidaemia, hypertension, obesity and hyperglycaemia in the presence of insulin resistance. In addition, diabetes is associated with an inflammatory and pro-thrombotic environment, exacerbating the development of atherothrombosis. Insulin resistance and hyperglycaemia both contribute to the development of endothelial cell dysfunction and increased oxidative stress, culminating in accelerated atherosclerosis. Clot formation and function are also directly affected by insulin resistance and hyperglycaemia, with increased levels of coagulation factors and anti-fibrinolytic proteins and a fibrin network that is more resistant to lysis, coupled with increased platelet activation.
It is well recognised that the intensification of glycaemic control leads to a reduction in microvascular complications in type 1 and type 2 diabetes; however, the same is less clear with macrovascular disease. Several randomised studies have attempted to address the effect of short-, medium- and long-term glycaemic control on cardiovascular outcomes, with mixed results. The overall interpretation of these trials suggests that intensive glycaemic control in patients with a relatively short duration of diabetes, without very poor control and with no CVD, might be safe and associated with fewer cardiovascular events.
This review will summarise the effects of hyperglycaemia on the development of atherothrombosis and examine key cardiovascular outcome trials following intensive glucose control.
Zusammenfassung
Diabetes steht mit der Entwicklung von vorzeitig auftretenden kardiovaskulären Erkrankungen (cardiovascular disease, CVD) in Verbindung, die mit der Häufung von Risikofaktoren wie Dyslipidämie, Hypertonie, Adipositas und Hyperglykämie bei gleichzeitiger Insulinresistenz zusammenhängen. Diabetes geht außerdem mit einem entzündlichen und prothrombotischen Umfeld einher, was die Entwicklung von Atherothrombose exazerbiert. Insulinresistenz und Hyperglykämie tragen beide zur Entwicklung von Endothelzellendysfunktion und erhöhtem oxidativem Stress bei, was in beschleunigter Atherosklerose gipfelt. Bildung und Funktion von Gerinseln werden von Insulinresistenz und Hyperglykämie ebenfalls direkt beeinflusst, mit erhöhten Spiegeln von Blutgerinnungsfaktoren und antifibrinolytischen Proteinen und einem Fibrinnetzwerk, das Lyse gegenüber resistenter ist, verbunden mit einer erhöhten Plättchenaktivierung.
Es ist allgemein anerkannt, dass die Intensivierung der Blutzuckerkontrolle zu einer Verringerung mikrovaskulärer Komplikationen bei Diabetes Typ 1 und Typ 2 führt. Dies ist allerdings bei makrovaskulären Erkrankungen weniger deutlich. Verschiedene randomisierte Studien haben versucht, die Auswirkungen von kurz-, mittel- und langfristiger Blutzuckerkontrolle auf kardiovaskuläre Ergebnisse zu untersuchen, mit uneinheitlichen Ergebnissen. Die allgemeine Interpretation dieser Untersuchungen deutet darauf hin, dass die intensive Blutzuckerkontrolle bei Patienten mit einer vergleichsweise kurzen Dauer des Diabetes, ohne mangelhafte Kontrolle und ohne CVD, zuverlässig sein und mit weniger kardiovaskulären Ereignissen verbunden sein könnte.
Dieser Überblick fasst die Auswirkungen von Hyperglykämie auf die Entwicklung von Atherothrombose zusammen und untersucht zentrale kardiovaskuläre Endpunktstudien nach intensiver Blutzuckerkontrolle.


Similar content being viewed by others
References
Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M (1998) Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 339:229–234
Cubbon RM, Wheatcroft SB, Grant PJ et al (2007) Temporal trends in mortality of patients with diabetes mellitus suffering acute myocardial infarction: a comparison of over 3000 patients between 1995 and 2003. Eur Heart J 28:540–545
Kahn MB, Cubbon RM, Mercer B et al (2012) Association of diabetes with increased all-cause mortality following primary percutaneous coronary intervention for ST-segment elevation myocardial infarction in the contemporary era. Diabetes Vasc Dis Res 9:3–9
Anonymous (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 329:977–986
Anonymous (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352:837–853
Semple RK, Savage DB, Cochran EK, Gorden P, O’Rahilly S (2011) Genetic syndromes of severe insulin resistance. Endocr Rev 32:498–514
Steinberg HO, Chaker H, Leaming R, Johnson A, Brechtel G, Baron AD (1996) Obesity/insulin resistance is associated with endothelial dysfunction. Implications for the syndrome of insulin resistance. J Clin Invest 97:2601–2610
Perticone F, Ceravolo R, Candigliota M et al (2001) Obesity and body fat distribution induce endothelial dysfunction by oxidative stress: protective effect of vitamin C. Diabetes 50:159–165
Kohler HP, Grant PJ (2000) Plasminogen-Activator Inhibitor Type 1 and Coronary Artery Disease. N Engl J Med 342:1792–1801
Festa A, D’Agostino R Jr., Tracy RP, Haffner SM (2002) Elevated levels of acute-phase proteins and plasminogen activator inhibitor-1 predict the development of type 2 diabetes: the insulin resistance atherosclerosis study. Diabetes 51:1131–1137
Folsom AR, Wu KK, Davis CE, Conlan MG, Sorlie, Szklo M (1991) Population correlates of plasma fibrinogen and factor VII, putative cardiovascular risk factors. Atherosclerosis 91:191–205
Heywood DM, Mansfield MW, Grant PJ (1996) Factor VII gene polymorphisms, factor VII:C levels and features of insulin resistance in non-insulin-dependent diabetes mellitus. Thromb Haemost 75:401–406
Engstrom G, Hedblad B, Eriksson KF, Janzon L, Lindgarde F (2005) Complement C3 is a risk factor for the development of diabetes: a population-based cohort study. Diabetes 54:570–575
Thorand B, Lowel H, Schneider A et al (2003) C‑reactive protein as a predictor for incident diabetes mellitus among middle-aged men: results from the MONICa Augsburg cohort study, 1984–1998. Arch Intern Med 163:93–99
Balletshofer BM, Rittig K, Enderle MD et al (2000) Endothelial dysfunction is detectable in young normotensive first-degree relatives of subjects with type 2 diabetes in association with insulin resistance. Circulation 101:1780–1784
Mansfield MW, Heywood DM, Grant PJ (1996) Circulating levels of factor VII, fibrinogen, and von Willebrand factor and features of insulin resistance in first-degree relatives of patients with NIDDM. Circulation 94:2171–2176
Mansfield MW, Stickland MH, Grant PJ (1997) PAI-1 concentrations in first-degree relatives of patients with non-insulin-dependent diabetes: metabolic and genetic associations. Thromb Haemost 77:357–361
Naseem KM (2005) The role of nitric oxide in cardiovascular diseases. Mol Aspects Med 26:33–65
Tanner FC, Meier P, Greutert H, Champion C, Nabel EG, Luscher TF (2000) Nitric oxide modulates expression of cell cycle regulatory proteins: a cytostatic strategy for inhibition of human vascular smooth muscle cell proliferation. Circulation 101:1982–1989
Bohl KS, West JL (2000) Nitric oxide-generating polymers reduce platelet adhesion and smooth muscle cell proliferation. Biomaterials 21:2273–2278
Clapp BR, Hingorani AD, Kharbanda RK et al (2004) Inflammation-induced endothelial dysfunction involves reduced nitric oxide bioavailability and increased oxidant stress. Cardiovasc Res 64:172–178
Quyyumi AA, Dakak N, Andrews NP, Gilligan DM, Panza JA, Cannon ROIII (1995) Contribution of nitric oxide to metabolic coronary vasodilation in the human heart. Circulation 92:320–326
Tousoulis D, Davies G, Stefanadis C, Toutouzas P, Ambrose JA (2003) Inflammatory and thrombotic mechanisms in coronary atherosclerosis. Heart 89:993–997
Levi M, van der Poll T (2010) Inflammation and coagulation. Crit Care Med 38:S26–S34
Cirillo P, Golino P, Calabro P et al (2005) C‑reactive protein induces tissue factor expression and promotes smooth muscle and endothelial cell proliferation. Cardiovasc Res 68:47–55
Singh U, Jialal I (2006) Oxidative stress and atherosclerosis. Pathophysiology 13:129–142
Taylor AA (2001) Pathophysiology of hypertension and endothelial dysfunction in patients with diabetes mellitus. Endocrinol Metab Clin North Am 30:983–997
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Robertson RP, Harmon JS (2006) Diabetes, glucose toxicity, and oxidative stress: A case of double jeopardy for the pancreatic islet beta cell. Free Radic Biol Med 41:177–184
Lumeng CN, Saltiel AR (2011) Inflammatory links between obesity and metabolic disease. J Clin Invest 121:2111–2117
Goossens GH (2008) The role of adipose tissue dysfunction in the pathogenesis of obesity-related insulin resistance. Physiol Behav 94:206–218
Pasceri V, Willerson JT, Yeh ET (2000) Direct proinflammatory effect of C‑reactive protein on human endothelial cells. Circulation 102:2165–2168
Van Gaal LF, Mertens IL, De Block CE (2006) Mechanisms linking obesity with cardiovascular disease. Nature 444:875–880
Torimoto K, Okada Y, Mori H, Tanaka Y (2013) Relationship between fluctuations in glucose levels measured by continuous glucose monitoring and vascular endothelial dysfunction in type 2 diabetes mellitus. Cardiovasc Diabetol 12:1
Okada K, Hibi K, Gohbara M et al (2015) Association between blood glucose variability and coronary plaque instability in patients with acute coronary syndromes. Cardiovasc Diabetol 14:111
Monnier L, Mas E, Ginet C et al (2006) Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 295:1681–1687
Gerrits AJ, Koekman CA, van Haeften TW, Akkerman JW (2010) Platelet tissue factor synthesis in type 2 diabetic patients is resistant to inhibition by insulin. Diabetes 59:1487–1495
Grant PJ (2007) Diabetes mellitus as a prothrombotic condition. J Intern Med 262:157–172
Pinkney JH, Stehouwer CD, Coppack SW, Yudkin JS (1997) Endothelial dysfunction: cause of the insulin resistance syndrome. Diabetes 46(Suppl 2):9–13
Undas A, Wiek I, Stepien E, Zmudka K, Tracz W (2008) Hyperglycemia is associated with enhanced thrombin formation, platelet activation, and fibrin clot resistance to lysis in patients with acute coronary syndrome. Diabetes Care 31:1590–1595
Dunn EJ, Ariens RA (2004) Fibrinogen and fibrin clot structure in diabetes. Herz 29:470–479
Ajjan R, Grant PJ (2006) Coagulation and atherothrombotic disease. Atherosclerosis 186:240–259
Pieters M, van Zyl DG, Rheeder P et al (2007) Glycation of fibrinogen in uncontrolled diabetic patients and the effects of glycaemic control on fibrinogen glycation. Thromb Res 120:439–446
Hess K, Alzahrani SH, Mathai M et al (2011) A novel mechanism for hypofibrinolysis in diabetes: the role of complement C3. Diabetologia 55(4):1103–1113
Dunn EJ, Ariens RA, Grant PJ (2005) The influence of type 2 diabetes on fibrin structure and function. Diabetologia 48:1198–1206
Dunn EJ, Philippou H, Ariens RA, Grant PJ (2006) Molecular mechanisms involved in the resistance of fibrin to clot lysis by plasmin in subjects with type 2 diabetes mellitus. Diabetologia 49:1071–1080
Jorneskog G, Egberg N, Fagrell B et al (1996) Altered properties of the fibrin gel structure in patients with IDDM. Diabetologia 39:1519–1523
Pieters M, Covic N, van der Westhuizen FH et al (2008) Glycaemic control improves fibrin network characteristics in type 2 diabetes – a purified fibrinogen model. Thromb Haemost 99:691–700
Seljeflot I, Larsen JR, Dahlgren S, Hanssen KF, Arnesen H (2006) Fibrinolytic activity is highly influenced by long-term glycemic control in Type 1 diabetic patients. J Thromb Haemost 4:686–688
Ajjan RA, Gamlen T, Standeven KF et al (2013) Diabetes is associated with post-translational modifications in plasminogen resulting in reduced plasmin generation and enzyme specific activity. Blood 122(1):134–142
Agren A, Jorneskog G, Elgue G, Henriksson P, Wallen H, Wiman B (2014) Increased incorporation of antiplasmin into the fibrin network in patients with type 1 diabetes. Diabetes Care 37:2007–2014
Howes JM, Richardson VR, Smith KA et al (2012) Complement C3 is a novel plasma clot component with anti-fibrinolytic properties. Diabetes Vasc Dis Res 9:216–225
Ferreira IA, Mocking AI, Feijge MA et al (2006) Platelet inhibition by insulin is absent in type 2 diabetes mellitus. Arterioscler Thromb Vasc Biol 26:417–422
Hess K, Grant PJ (2011) Inflammation and thrombosis in diabetes. Thromb Haemost 105(Suppl 1):43–54
Watala C (2005) Blood platelet reactivity and its pharmacological modulation in (people with) diabetes mellitus. Curr Pharm Des 11:2331–2365
Angiolillo DJ, Suryadevara S (2009) Aspirin and clopidogrel: efficacy and resistance in diabetes mellitus. Best Pract Res Clin Endocrinol Metab 23:375–388
Trovati M, Anfossi G (1998) Insulin, insulin resistance and platelet function: similarities with insulin effects on cultured vascular smooth muscle cells. Diabetologia 41:609–622
Ferroni P, Basili S, Falco A, Davi G (2004) Platelet activation in type 2 diabetes mellitus. J Thromb Haemost 2:1282–1291
Kannel WB, Neaton JD, Wentworth D et al (1986) Overall and coronary heart disease mortality rates in relation to major risk factors in 325,348 men screened for the MRFIT. multiple risk factor intervention trial. Am Heart J 112:825–836
Jerums G, Panagiotopoulos S, Ekinci E, MacIsaac RJ (2015) Cardiovascular outcomes with antihypertensive therapy in type 2 diabetes: an analysis of intervention trials. J Hum Hypertens 29:473–477
Anonymous (2002) MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high–risk individuals: a randomised placebo-controlled trial. Lancet 360:7–22
Colhoun HM, Betteridge DJ, Durrington PN et al (2004) Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo-controlled trial. Lancet 364:685–696
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O (2003) Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 348:383–393
Gaede P, Lund-Andersen H, Parving HH, Pedersen O (2008) Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 358:580–591
Li G, Zhang P, Wang J et al (2014) Cardiovascular mortality, all-cause mortality, and diabetes incidence after lifestyle intervention for people with impaired glucose tolerance in the Da Qing Diabetes Prevention Study: a 23-year follow-up study. Lancet Diabetes Endocrinol 2:474–480
Wing RR, Bolin P, Brancati FL et al (2013) Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 369:145–154
Turnbull FM, Abraira C, Anderson RJ et al (2009) Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia 52:2288–2298
Green JB, Bethel MA, Armstrong PW et al (2015) Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 373:232–242
White WB, Bakris GL, Bergenstal RM et al (2011) EXamination of cArdiovascular outcoMes with alogliptIN versus standard of carE in patients with type 2 diabetes mellitus and acute coronary syndrome (EXAMINE): a cardiovascular safety study of the dipeptidyl peptidase 4 inhibitor alogliptin in patients with type 2 diabetes with acute coronary syndrome. Am Heart J 162:620–626
Zinman B, Wanner C, Lachin JM et al (2015) Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med 373:2117–2212
Gerstein HC, Miller ME, Byington RP et al (2008) Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 358:2545–2559
Patel A, MacMahon S, Chalmers J et al (2008) Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 358:2560–2572
Gerstein HC, Miller ME, Genuth S et al (2011) Long-term effects of intensive glucose lowering on cardiovascular outcomes. N Engl J Med 364:818–828
Duckworth W, Abraira C, Moritz T et al (2009) Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 360:129–139
Anonymous (2005) Intensive Diabetes Treatment and Cardiovascular Disease in Patients with Type 1 Diabetes. N Engl J Med 353:2643–2653
Anonymous (1998) Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352:854–865
Holman RR, Paul SK, Bethel MA, Neil HA, Matthews DR (2008) Long-term follow-up after tight control of blood pressure in type 2 diabetes. N Engl J Med 359:1565–1576
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R.J. King and P.J. Grant state that there are no conflicts of interest.
The accompanying manuscript does not include studies on humans or animals.
Rights and permissions
About this article
Cite this article
King, R.J., Grant, P.J. Diabetes and cardiovascular disease: pathophysiology of a life-threatening epidemic. Herz 41, 184–192 (2016). https://doi.org/10.1007/s00059-016-4414-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-016-4414-8