Abstract
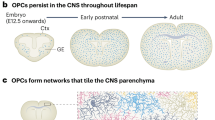
Oligodendrocytes are the myelin-forming cells in the central nervous system (CNS). These cells originate from oligodendrocyte precursor cells (OPCs) during development, and they migrate extensively from oligodendrogliogenic niches along the neural tube to colonise the entire CNS. Like many other such events, this migratory process is precisely regulated by a battery of positional and signalling cues that act via their corresponding receptors and that are expressed dynamically by OPCs. Here, we will review the cellular and molecular basis of this important event during embryonic and postnatal development, and we will discuss the relevance of the substantial number of OPCs existing in the adult CNS. Similarly, we will consider the behaviour of OPCs in normal and pathological conditions, especially in animal models of demyelination and of the demyelinating disease, multiple sclerosis. The spontaneous remyelination observed after damage in demyelinating pathologies has a limited effect. Understanding the cellular and molecular mechanisms underlying the biology of OPCs, particularly adult OPCs, should help in the design of neuroregenerative strategies to combat multiple sclerosis and other demyelinating diseases.



Similar content being viewed by others
References
Del Rio-Hortega P (2012) Estudios sobre la neuroglia. La glia de escasas radiaciones-oligodendroglia. Boletin Real Soc Hist Nat 21:63–92
Barres BA, Barde Y (2000) Neuronal and glial cell biology. Curr Opin Neurobiol 10:642–648
Zalc B, Colman DR (2000) Origins of vertebrate success. Science 14(288):271–272
Zalc B, Goujet D, Colman D (2008) The origin of the myelination program in vertebrates. Curr Biol 18:R511–R512
Emery B (2010) Regulation of oligodendrocyte differentiation and myelination. Science 330:779–782
Fields RD (2005) Myelination: an overlooked mechanism of synaptic plasticity? Neuroscientist 11:528–531
Bjartmar C, Hildebrand C, Loinder K (1994) Morphological heterogeneity of rat oligodendrocytes: electron microscopic studies on serial sections. Glia 11:235–244
Peters A, Palay S, Webster HF (1991) The fine structure of the nervous system: Neurons and their supporting cells, 3rd edn. Oxford University Press, Oxford
Frohlich N, Nagy B, Hovhannisyan A, Kukley M (2011) Fate of neuron-glia synapses during proliferation and differentiation of NG2 cells. J Anat 219:18–32
Noll E, Miller RH (1993) Oligodendrocyte precursors originate at the ventral ventricular zone dorsal to the ventral midline region in the embryonic rat spinal cord. Development 118:563–573
Warf BC, Fok-Seang J, Miller RH (1991) Evidence for the ventral origin of oligodendrocyte precursors in the rat spinal cord. J Neurosci 11:2477–2488
Cai J, Qi Y, Hu X et al (2005) Generation of oligodendrocyte precursor cells from mouse dorsal spinal cord independent of Nkx6 regulation and Shh signaling. Neuron 45:41–53
Fogarty M, Richardson WD, Kessaris N (2005) A subset of oligodendrocytes generated from radial glia in the dorsal spinal cord. Development 132:1951–1959
Kessaris N, Fogarty M, Iannarelli P, Grist M, Wegner M, Richardson WD (2006) Competing waves of oligodendrocytes in the forebrain and postnatal elimination of an embryonic lineage. Nat Neurosci 9:173–179
Vallstedt A, Klos JM, Ericson J (2005) Multiple dorsoventral origins of oligodendrocyte generation in the spinal cord and hindbrain. Neuron 45:55–67
DE Castro F, Zalc B (2013) Migration of myelin-forming cells in the CNS. In: Cellular migration and formation of neuronal connections: comprehensive developmental neuroscience. Elsevier, Amsterdam (in press)
Rowitch DH (2004) Glial specification in the vertebrate neural tube. Nat Rev Neurosci 5:409–419
Furusho M, Kaga Y, Ishii A, Hebert JM, Bansal R (2011) Fibroblast growth factor signaling is required for the generation of oligodendrocyte progenitors from the embryonic forebrain. J Neurosci 31:5055–5066
Richardson WD, Kessaris N, Pringle N (2006) Oligodendrocyte wars. Nat Rev Neurosci 7:11–18
Tripathi RB, Clarke LE, Burzomato V et al (2011) Dorsally and ventrally derived oligodendrocytes have similar electrical properties but myelinate preferred tracts. J Neurosci 31:6809–6819
de Castro F, Bribian A (2005) The molecular orchestra of the migration of oligodendrocyte precursors during development. Brain Res Rev 49:227–241
Decker L, Avellana-Adalid V, Nait-Oumesmar B, Durbec P, Baron-Van Evercooren A (2000) Oligodendrocyte precursor migration and differentiation: combined effects of PSA residues, growth factors, and substrates. Mol Cell Neurosci 16:422–439
Dubois-Dalcq M, Murray K (2000) Why are growth factors important in oligodendrocyte physiology? Pathol Biol 48:80–86
Rogister B, Ben-Hur T, Dubois-Dalcq M (1999) From neural stem cells to myelinating oligodendrocytes. Mol Cell Neurosci 14:287–300
Demerens C, Stankoff B, Logak M et al (1996) Induction of myelination in the central nervous system by electrical activity. Proc Natl Acad Sci USA 93:9887–9892
Stevens B, Porta S, Haak LL, Gallo V, Fields RD (2002) Adenosine: a neuron-glial transmitter promoting myelination in the CNS in response to action potentials. Neuron 36:855–868
Stritt C, Stern S, Harting K et al (2009) Paracrine control of oligodendrocyte differentiation by SRF-directed neuronal gene expression. Nat Neurosci 12:418–427
Wake H, Lee PR, Fields RD (2011) Control of local protein synthesis and initial events in myelination by action potentials. Science 333:1647–1651
Ono K, Yasui Y, Rutishauser U, Miller RH (1997) Focal ventricular origin and migration of oligodendrocyte precursors into the chick optic nerve. Neuron 19:283–292
Small RK, Riddle P, Noble M (1987) Evidence for migration of oligodendrocyte–type-2 astrocyte progenitor cells into the developing rat optic nerve. Nature 328:155–157
Spassky N, de Castro F, Le Bras B et al (2002) Directional guidance of oligodendroglial migration by class 3 semaphorins and netrin-1. J Neurosci 22:5992–6004
Leber SM, Sanes JR (1995) Migratory paths of neurons and glia in the embryonic chick spinal cord. J Neurosci 15:1236–1248
Leber SM, Yamagata M, Sanes JR (1996) Gene transfer using replication-defective retroviral and adenoviral vectors. Methods Cell Biol 51:161–183
Miller RH, Ono K (1998) Morphological analysis of the early stages of oligodendrocyte development in the vertebrate central nervous system. Microsc Res Tech 41:441–453
Ono K, Bansal R, Payne J, Rutishauser U, Miller RH (1995) Early development and dispersal of oligodendrocyte precursors in the embryonic chick spinal cord. Development 121:1743–1754
Olivier C, Cobos I, Perez Villegas EM et al (2001) Monofocal origin of telencephalic oligodendrocytes in the anterior entopeduncular area of the chick embryo. Development 128:1757–1769
Garcia-Lopez R, Martinez S (2010) Oligodendrocyte precursors originate in the parabasal band of the basal plate in prosomere 1 and migrate into the alar prosencephalon during chick development. Glia 58:1437–1450
Mecklenburg N, Garcia-Lopez R, Puelles E, Sotelo C, Martinez S (2011) Cerebellar oligodendroglial cells have a mesencephalic origin. Glia 59:1946–1957
Sugimoto Y, Taniguchi M, Yagi T, Akagi Y, Nojyo Y, Tamamaki N (2001) Guidance of glial precursor cell migration by secreted cues in the developing optic nerve. Development 128:3321–3330
Ueda H, Levine JM, Miller RH, Trapp BD (1999) Rat optic nerve oligodendrocytes develop in the absence of viable retinal ganglion cell axons. J Cell Biol 146:1365–1374
Cohen RI (2005) Exploring oligodendrocyte guidance: ‘to boldly go where no cell has gone before’. Cell Mol Life Sci 62:505–510
Bribián A, Barallobre MJ, Soussi-Yanicostas N, de Castro F (2006) Anosmin-1 modulates the FGF-2-dependent migration of oligodendrocyte precursors in the developing optic nerve. Mol Cell Neurosci 33:2–14
Simpson PB, Armstrong RC (1999) Intracellular signals and cytoskeletal elements involved in oligodendrocyte progenitor migration. Glia 26:22–35
Fruttiger M, Karlsson L, Hall AC et al (1999) Defective oligodendrocyte development and severe hypomyelination in PDGF-A knockout mice. Development 126:457–467
Milner R, Frost E, Nishimura S et al (1997) Expression of alpha vbeta3 and alpha vbeta8 integrins during oligodendrocyte precursor differentiation in the presence and absence of axons. Glia 21:350–360
Merchan P, Bribian A, Sanchez-Camacho C, Lezameta M, Bovolenta P, de Castro F (2007) Sonic hedgehog promotes the migration and proliferation of optic nerve oligodendrocyte precursors. Mol Cell Neurosci 36:355–368
Bribián A, Esteban PF, Clemente D et al (2008) A novel role for anosmin-1 in the adhesion and migration of oligodendrocyte precursors. Dev Neurobiol 68:1503–1516
Endo Y, Ishiwata-Endo H, Yamada KM (2012) Extracellular matrix protein anosmin promotes neural crest formation and regulates FGF, BMP, and WNT activities. Dev Cell 23:305–316
Gadea A, Aguirre A, Haydar TF, Gallo V (2009) Endothelin-1 regulates oligodendrocyte development. J Neurosci 29:10047–10062
Osterhout DJ, Ebner S, Xu J, Ornitz DM, Zazanis GA, McKinnon RD (1997) Transplanted oligodendrocyte progenitor cells expressing a dominant-negative FGF receptor transgene fail to migrate in vivo. J Neurosci 17:9122–9132
Frost E, Kiernan BW, Faissner A, ffrench-Constant C (1996) Regulation of oligodendrocyte precursor migration by extracellular matrix: evidence for substrate-specific inhibition of migration by tenascin-C. Dev Neurosci 18:266–273
Murcia-Belmonte V, Esteban PF, Garcia-Gonzalez D, de Castro F (2010) Biochemical dissection of Anosmin-1 interaction with FGFR1 and components of the extracellular matrix. J Neurochem 115:1256–1265
Clemente D, Esteban PF, Del Valle I et al (2008) Expression pattern of Anosmin-1 during pre- and postnatal rat brain development. Dev Dyn 237:2518–2528
Armendariz BG, Bribian A, Perez-Martinez E et al (2012) Expression of semaphorin 4F in neurons and brain oligodendrocytes and the regulation of oligodendrocyte precursor migration in the optic nerve. Mol Cell Neurosci 49:54–67
Taniguchi Y, Amazaki M, Furuyama T et al (2009) Sema4D deficiency results in an increase in the number of oligodendrocytes in healthy and injured mouse brains. J Neurosci Res 87:2833–2841
Goldberg JL, Vargas ME, Wang JT et al (2004) An oligodendrocyte lineage-specific semaphorin, Sema5A, inhibits axon growth by retinal ganglion cells. J Neurosci 24:4989–4999
Li X, Lee AY (2010) Semaphorin 5A and plexin-B3 inhibit human glioma cell motility through RhoGDIalpha-mediated inactivation of Rac1 GTPase. J Biol Chem 285:32436–32445
Petros TJ, Williams SE, Mason CA (2006) Temporal regulation of EphA4 in astroglia during murine retinal and optic nerve development. Mol Cell Neurosci 32:49–66
Prestoz L, Chatzopoulou E, Lemkine G et al (2004) Control of axonophilic migration of oligodendrocyte precursor cells by Eph-ephrin interaction. Neuron Glia Biol 1:73–83
Nakada M, Niska JA, Miyamori H et al (2004) The phosphorylation of EphB2 receptor regulates migration and invasion of human glioma cells. Cancer Res 64:3179–3185
Zhang H, Vutskits L, Calaora V, Durbec P, Kiss JZ (2004) A role for the polysialic acid-neural cell adhesion molecule in PDGF-induced chemotaxis of oligodendrocyte precursor cells. J Cell Sci 117:93–103
Zhang H, Vutskits L, Pepper MS, Kiss JZ (2003) VEGF is a chemoattractant for FGF-2-stimulated neural progenitors. J Cell Biol 163:1375–1384
Ortega MC, Cases O, Merchan P, Kozyraki R, Clemente D, de Castro F (2012) Megalin mediates the influence of sonic hedgehog on oligodendrocyte precursor cell migration and proliferation during development. Glia 60:851–866
Jarjour AA, Manitt C, Moore SW, Thompson KM, Yuh SJ, Kennedy TE (2003) Netrin-1 is a chemorepellent for oligodendrocyte precursor cells in the embryonic spinal cord. J Neurosci 23:3735–3744
Tsai HH, Tessier-Lavigne M, Miller RH (2003) Netrin 1 mediates spinal cord oligodendrocyte precursor dispersal. Development 130:2095–2105
Tsai HH, Macklin WB, Miller RH (2006) Netrin-1 is required for the normal development of spinal cord oligodendrocytes. J Neurosci 26:1913–1922
Tsai HH, Macklin WB, Miller RH (2009) Distinct modes of migration position oligodendrocyte precursors for localized cell division in the developing spinal cord. J Neurosci Res 87:3320–3330
Tsai HH, Miller RH (2002) Glial cell migration directed by axon guidance cues. Trends Neurosci 25:173–175
Ortega MC, Bribian A, Peregrin S, Gil MT, Marin O, de Castro F (2012) Neuregulin-1/ErbB4 signaling controls the migration of oligodendrocyte precursor cells during development. Exp Neurol 235:610–620
Tsai HH, Frost E, To V et al (2002) The chemokine receptor CXCR2 controls positioning of oligodendrocyte precursors in developing spinal cord by arresting their migration. Cell 110:373–383
Padovani-Claudio DA, Liu L, Ransohoff RM, Miller RH (2006) Alterations in the oligodendrocyte lineage, myelin, and white matter in adult mice lacking the chemokine receptor CXCR2. Glia 54:471–483
Dziembowska M, Tham TN, Lau P, Vitry S, Lazarini F, Dubois-Dalcq M (2005) A role for CXCR4 signaling in survival and migration of neural and oligodendrocyte precursors. Glia 50:258–269
Levison SW, Goldman JE (1993) Both oligodendrocytes and astrocytes develop from progenitors in the subventricular zone of postnatal rat forebrain. Neuron 10:201–212
Young KM, Psachoulia K, Tripathi RB et al (2013) Oligodendrocyte Dynamics in the Healthy Adult CNS: evidence for Myelin Remodeling. Neuron 77:873–885
Zhu X, Hill RA, Nishiyama A (2008) NG2 cells generate oligodendrocytes and gray matter astrocytes in the spinal cord. Neuron Glia Biol 4:19–26
Gonzalez-Perez O (2011) Alvarez-Buylla A (2011) Oligodendrogenesis in the subventricular zone and the role of epidermal growth factor. Brain Res Rev 67:147–156
Richardson WD, Young KM, Tripathi RB, McKenzie I (2011) NG2-glia as multipotent neural stem cells: fact or fantasy? Neuron 70:661–673
Etxeberria A, Mangin JM, Aguirre A, Gallo V (2010) Adult-born SVZ progenitors receive transient synapses during remyelination in corpus callosum. Nat Neurosci 13:287–289
Fancy SP, Zhao C, Franklin RJ (2004) Increased expression of Nkx2.2 and Olig2 identifies reactive oligodendrocyte progenitor cells responding to demyelination in the adult CNS. Mol Cell Neurosci 27:247–254
Gensert JM, Goldman JE (1997) Endogenous progenitors remyelinate demyelinated axons in the adult CNS. Neuron 19:197–203
Menn B, Garcia-Verdugo JM, Yaschine C, Gonzalez-Perez O, Rowitch D, Alvarez-Buylla A (2006) Origin of oligodendrocytes in the subventricular zone of the adult brain. J Neurosci 26:7907–7918
Nait-Oumesmar B, Decker L, Lachapelle F, vellana-Adalid v, Bachelin c, Baron-Van Evercooren A (1999) Progenitor cells of the adult mouse subventricular zone proliferate, migrate and differentiate into oligodendrocytes after demyelination. Eur J Neurosci 11:4357–4366
Assanah M, Lochhead R, Ogden A, Bruce J, Goldman J, Canoll P (2006) Glial progenitors in adult white matter are driven to form malignant gliomas by platelet-derived growth factor-expressing retroviruses. J Neurosci 26:6781–6790
Dawson MR, Polito A, Levine JM, Reynolds R (2003) NG2-expressing glial progenitor cells: an abundant and widespread population of cycling cells in the adult rat CNS. Mol Cell Neurosci 24:476–488
Roy NS, Wang S, Harrison-Restelli C et al (1999) Identification, isolation, and promoter-defined separation of mitotic oligodendrocyte progenitor cells from the adult human subcortical white matter. J Neurosci 19:9986–9995
LeVine SM, Goldman JE (1988) Ultrastructural characteristics of GD3 ganglioside-positive immature glia in rat forebrain white matter. J Comp Neurol 277:456–464
Mangin JM, Gallo V (2011) The curious case of NG2 cells: transient trend or game changer? ASN Neuro 3:e00052
Nishiyama A, Watanabe M, Yang Z, Bu J (2002) Identity, distribution, and development of polydendrocytes: NG2-expressing glial cells. J Neurocytol 31:437–455
Pringle NP, Mudhar HS, Collarini EJ, Richardson WD (1992) PDGF receptors in the rat CNS: during late neurogenesis, PDGF alpha-receptor expression appears to be restricted to glial cells of the oligodendrocyte lineage. Development 115:535–551
Stallcup WB, Beasley L (1987) Bipotential glial precursor cells of the optic nerve express the NG2 proteoglycan. J Neurosci 7:2737–2744
Fang X, Burg MA, Barritt D, hlin-Huppe K, Nishiyama A, Stallcup WB (1999) Cytoskeletal reorganization induced by engagement of the NG2 proteoglycan leads to cell spreading and migration. Mol Biol Cell 10:3373–3387
Lin XH, hlin-Huppe k, Stallcup WB (1996) Interaction of the NG2 proteoglycan with the actin cytoskeleton. J Cell Biochem 63:463–477
Majumdar M, Vuori K, Stallcup WB (2003) Engagement of the NG2 proteoglycan triggers cell spreading via rac and p130cas. Cell Signal 15:79–84
Tillet E, Gential B, Garrone R, Stallcup WB (2002) NG2 proteoglycan mediates beta1 integrin-independent cell adhesion and spreading on collagen VI. J Cell Biochem 86:726–736
Doetsch F, Caille I, Lim DA, Garcia-Verdugo JM, Alvarez-Buylla A (1999) Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97:703–716
Blakemore WF, Gilson JM, Crang AJ (2000) Transplanted glial cells migrate over a greater distance and remyelinate demyelinated lesions more rapidly than endogenous remyelinating cells. J Neurosci Res 61:288–294
Franklin RJ, Gilson JM, Blakemore WF (1997) Local recruitment of remyelinating cells in the repair of demyelination in the central nervous system. J Neurosci Res 50:337–344
Nishiyama A, Komitova M, Suzuki R, Zhu X (2009) Polydendrocytes (NG2 cells): multifunctional cells with lineage plasticity. Nat Rev Neurosci 10:9–22
Butt AM, Duncan A, Hornby MF et al (1999) Cells expressing the NG2 antigen contact nodes of Ranvier in adult CNS white matter. Glia 26:84–91
Butt AM, Kiff J, Hubbard P, Berry M (2002) Synantocytes: new functions for novel NG2 expressing glia. J Neurocytol 31:551–565
Wigley R, Butt AM (2009) Integration of NG2-glia (synantocytes) into the neuroglial network. Neuron Glia Biol 5:21–28
Gudz TI, Komuro H, Macklin WB (2006) Glutamate stimulates oligodendrocyte progenitor migration mediated via an alphav integrin/myelin proteolipid protein complex. J Neurosci 26:2458–2466
Yuan X, Eisen AM, McBain CJ, Gallo V (1998) A role for glutamate and its receptors in the regulation of oligodendrocyte development in cerebellar tissue slices. Development 125:2901–2914
Gallo V, Mangin JM, Kukley M, Dietrich D (2008) Synapses on NG2-expressing progenitors in the brain: multiple functions? J Physiol 586:3767–3781
Gallo V, Zhou JM, McBain CJ, Wright P, Knutson PL, Armstrong RC (1996) Oligodendrocyte progenitor cell proliferation and lineage progression are regulated by glutamate receptor-mediated K + channel block. J Neurosci 16:2659–2670
Mangin JM, Li P, Scafidi J, Gallo V (2012) Experience-dependent regulation of NG2 progenitors in the developing barrel cortex. Nat Neurosci 15:1192–1194
Karadottir R, Cavelier P, Bergersen LH, Attwell D (2005) NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438:1162–1166
Wang C, Pralong WF, Schulz MF et al (1996) Functional N-methyl-D-aspartate receptors in O-2A glial precursor cells: a critical role in regulating polysialic acid-neural cell adhesion molecule expression and cell migration. J Cell Biol 135:1565–1581
Micu I, Jiang Q, Coderre E et al (2006) (2005) NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia. Nature 439:988–992
Salter MG, Fern R (2005) NMDA receptors are expressed in developing oligodendrocyte processes and mediate injury. Nature 438:1167–1171
Lin SC, Bergles DE (2004) Synaptic signaling between neurons and glia. Glia 47:290–298
Tong XP, Li XY, Zhou B et al (2009) Ca(2+) signaling evoked by activation of Na(+) channels and Na(+)/Ca(2+) exchangers is required for GABA-induced NG2 cell migration. J Cell Biol 186:113–128
Gregg C, Shikar V, Larsen P et al (2007) White matter plasticity and enhanced remyelination in the maternal CNS. J Neurosci 27:1812–1823
Simon C, Gotz M, Dimou L (2011) Progenitors in the adult cerebral cortex: cell cycle properties and regulation by physiological stimuli and injury. Glia 59:869–881
Aguirre A, Gallo V (2004) Postnatal neurogenesis and gliogenesis in the olfactory bulb from NG2-expressing progenitors of the subventricular zone. J Neurosci 24:10530–10541
Gonzalez-Perez O, Romero-Rodriguez R, Soriano-Navarro M, Garcia-Verdugo JM, Alvarez-Buylla A (2009) Epidermal growth factor induces the progeny of subventricular zone type B cells to migrate and differentiate into oligodendrocytes. Stem Cells 27:2032–2043
Azim K, Raineteau O, Butt AM (2012) Intraventricular injection of FGF-2 promotes generation of oligodendrocyte-lineage cells in the postnatal and adult forebrain. Glia 60:1977–1990
Clemente D, Ortega MC, Arenzana FJ, de Castro F (2011) FGF-2 and Anosmin-1 are selectively expressed in different types of multiple sclerosis lesions. J Neurosci 31:14899–14909
Loulier K, Ruat M, Traiffort E (2006) Increase of proliferating oligodendroglial progenitors in the adult mouse brain upon sonic hedgehog delivery in the lateral ventricle. J Neurochem 98:530–542
Bouslama-Oueghlani L, Wehrle R, Doulazmi M et al (2012) Purkinje cell maturation participates in the control of oligodendrocyte differentiation: role of sonic hedgehog and vitronectin. PLoS ONE 7:e49015
Colak D, Mori T, Brill MS et al (2008) Adult neurogenesis requires Smad4-mediated bone morphogenic protein signaling in stem cells. J Neurosci 28:434–446
Franklin RJ, ffrench-Constant C (2008) Remyelination in the CNS: from biology to therapy. Nat Rev Neurosci 9:839–855
Levine JM, Reynolds R, Fawcett JW (2001) The oligodendrocyte precursor cell in health and disease. Trends Neurosci 24:39–47
Bradl M, Lassmann H (2010) Oligodendrocytes: biology and pathology. Acta Neuropathol 119:37–53
Polito A, Reynolds R (2005) NG2-expressing cells as oligodendrocyte progenitors in the normal and demyelinated adult central nervous system. J Anat 207:707–716
Patrikios P, Stadelmann C, Kutzelnigg A et al (2006) Remyelination is extensive in a subset of multiple sclerosis patients. Brain 129:3165–3172
Compston A, Coles A (2008) Multiple sclerosis. Lancet 372:1502–1517
Piaton G, Williams A, Seilhean D, Lubetzki C (2009) Remyelination in multiple sclerosis. Prog Brain Res 175:453–464
Bramow S, Frischer JM, Lassmann H et al (2010) Demyelination versus remyelination in progressive multiple sclerosis. Brain 133:2983–2998
Piaton G, Aigrot MS, Williams A et al (2011) Class 3 semaphorins influence oligodendrocyte precursor recruitment and remyelination in adult central nervous system. Brain 134:1156–1167
Lucchinetti C, Bruck W, Parisi J, Scheithauer B, Rodriguez M, Lassmann H (1999) A quantitative analysis of oligodendrocytes in multiple sclerosis lesions. A study of 113 cases. Brain 122(Pt 12):2279–2295
Wolswijk G, Noble M (1989) Identification of an adult-specific glial progenitor cell. Development 105:387–400
Miller RH (2002) Regulation of oligodendrocyte development in the vertebrate CNS. Prog Neurobiol 67:451–467
Blakemore WF, Franklin RJ (2008) Remyelination in experimental models of toxin-induced demyelination. Curr Top Microbiol Immunol 318:193–212
Matsushima GK, Morell P (2001) The neurotoxicant, cuprizone, as a model to study demyelination and remyelination in the central nervous system. Brain Pathol 11:107–116
Omari KM, John GR, Sealfon SC, Raine CS (2005) CXC chemokine receptors on human oligodendrocytes: implications for multiple sclerosis. Brain 128:1003–1015
Moreno B, Espejo C, Mestre L et al (2012) Guidelines on the appropriate use of animal models for developing therapies in multiple sclerosis. Rev Neurol 54:114–124
Pachner AR (2011) Experimental models of multiple sclerosis. Curr Opin Neurol 24:291–299
Wang Y, Imitola J, Rasmussen S, O’Connor KC, Khoury SJ (2008) Paradoxical dysregulation of the neural stem cell pathway sonic hedgehog-Gli1 in autoimmune encephalomyelitis and multiple sclerosis. Ann Neurol 64:417–427
Williams A, Piaton G, Aigrot MS et al (2007) Semaphorin 3A and 3F: key players in myelin repair in multiple sclerosis? Brain 130:2554–2565
Butt AM, Dinsdale J (2005) Fibroblast growth factor 2 induces loss of adult oligodendrocytes and myelin in vivo. Exp Neurol 192:125–133
Rottlaender A, Villwock H, Addicks K, Kuerten S (2011) Neuroprotective role of fibroblast growth factor-2 in experimental autoimmune encephalomyelitis. Immunology 133:370–378
Messersmith DJ, Murtie JC, Le TQ, Frost EE, Armstrong RC (2000) Fibroblast growth factor 2 (FGF2) and FGF receptor expression in an experimental demyelinating disease with extensive remyelination. J Neurosci Res 62:241–256
Ruffini F, Furlan R, Poliani PL et al (2001) Fibroblast growth factor-II gene therapy reverts the clinical course and the pathological signs of chronic experimental autoimmune encephalomyelitis in C57BL/6 mice. Gene Ther 8:1207–1213
Armstrong RC, Le TQ, Frost EE, Borke RC, Vana AC (2002) Absence of fibroblast growth factor 2 promotes oligodendroglial repopulation of demyelinated white matter. J Neurosci 22:8574–8585
Armstrong RC, Le TQ, Flint NC, Vana AC, Zhou YX (2006) Endogenous cell repair of chronic demyelination. J Neuropathol Exp Neurol 65:245–256
Goddard DR, Berry M, Kirvell SL, Butt AM (2001) Fibroblast growth factor-2 inhibits myelin production by oligodendrocytes in vivo. Mol Cell Neurosci 18:557–569
Frohman EM, Racke MK, Raine CS (2006) Multiple sclerosis–the plaque and its pathogenesis. N Engl J Med 354:942–955
Mastronardi FG, Min W, Wang H et al (2004) Attenuation of experimental autoimmune encephalomyelitis and nonimmune demyelination by IFN-beta plus vitamin B12: treatment to modify notch-1/sonic hedgehog balance. J Immunol 172:6418–6426
Seifert T, Bauer J, Weissert R, Fazekas F, Storch MK (2005) Differential expression of sonic hedgehog immunoreactivity during lesion evolution in autoimmune encephalomyelitis. J Neuropathol Exp Neurol 64:404–411
Alvarez JI, Dodelet-Devillers A, Kebir H et al (2011) The Hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science 334:1727–1731
de Wit J, Toonen RF, Verhaagen J, Verhage M (2006) Vesicular trafficking of semaphorin 3A is activity-dependent and differs between axons and dendrites. Traffic 7:1060–1077
Kerstetter AE, Padovani-Claudio DA, Bai L, Miller RH (2009) Inhibition of CXCR2 signaling promotes recovery in models of multiple sclerosis. Exp Neurol 220:44–56
Canoll PD, Musacchio JM, Hardy R, Reynolds R, Marchionni MA, Salzer JL (1996) GGF/neuregulin is a neuronal signal that promotes the proliferation and survival and inhibits the differentiation of oligodendrocyte progenitors. Neuron 17:229–243
Cannella B, Hoban CJ, Gao YL et al (1998) The neuregulin, glial growth factor 2, diminishes autoimmune demyelination and enhances remyelination in a chronic relapsing model for multiple sclerosis. Proc Natl Acad Sci USA 95:10100–10105
Cannella B, Pitt D, Marchionni M, Raine CS (1999) Neuregulin and erbB receptor expression in normal and diseased human white matter. J Neuroimmunol 100:233–242
Norton N, Moskvina V, Morris DW et al (2006) Evidence that interaction between neuregulin 1 and its receptor erbB4 increases susceptibility to schizophrenia. Am J Med Genet B 141B:96–101
Cayre M, Canoll P, Goldman JE (2009) Cell migration in the normal and pathological postnatal mammalian brain. Prog Neurobiol 88:41–63
Mei L, Xiong WC (2008) Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nat Rev Neurosci 9:437–452
Charles P, Hernandez MP, Stankoff B et al (2000) Negative regulation of central nervous system myelination by polysialylated-neural cell adhesion molecule. Proc Natl Acad Sci USA 97:7585–7590
Lubetzki C, Demerens C, Anglade P et al (1993) Even in culture, oligodendrocytes myelinate solely axons. Proc Natl Acad Sci USA 90:6820–6824
Kremer D, Aktas O, Hartung HP, Kury P (2011) The complex world of oligodendroglial differentiation inhibitors. Ann Neurol 69:602–618
Mi S, Miller RH, Lee X et al (2005) LINGO-1 negatively regulates myelination by oligodendrocytes. Nat Neurosci 8:745–751
Mi S, Hu B, Hahm K et al (2007) LINGO-1 antagonist promotes spinal cord remyelination and axonal integrity in MOG-induced experimental autoimmune encephalomyelitis. Nat Med 13:1228–1233
Mi S, Miller RH, Tang W et al (2009) Promotion of central nervous system remyelination by induced differentiation of oligodendrocyte precursor cells. Ann Neurol 65:304–315
Jepson S, Vought B, Gross CH et al (2012) LINGO-1, a transmembrane signaling protein, inhibits oligodendrocyte differentiation and myelination through intercellular self-interactions. J Biol Chem 287:22184–22195
Farmer SF, Harrison LM, Mayston MJ, Parekh A, James LM, Stephens JA (2004) Abnormal cortex-muscle interactions in subjects with X-linked Kallmann’s syndrome and mirror movements. Brain 127:385–397
Steffenhagen C, Dechant FX, Oberbauer E et al (2012) Mesenchymal stem cells prime proliferating adult neural progenitors toward an oligodendrocyte fate. Stem Cells Dev 21:1838–1851
Rivers LE, Young KM, Rizzi M, et al (2008) PDGFRA/NG2 glia generate myelinating oligodendrocytes and piriform projection neurons in adult mice. Nat Neurosci 11:1392–1401
Acknowledgments
Our work at the GNDe was and/or is supported by grants from the Spanish Ministerio de Economía y Competitividad-MINECO (SAF2009-07842, ADE10-0010, SAF2012-40023, RD07-0060-2007, and RD12-0032-12, partially co-financed by F.E.D.E.R.; European Union, “Una manera de hacer Europa”), the Fundación Eugenio Rodríguez Pascual (Spain), and the Fondation ARSEP (France) to F.dC. F.dC is a senior researcher at SESCAM. A.B. holds a postdoctoral contract with the “Sara Borrell” program from the FIS-ISCIII/Spanish Ministerio de Economía y Competitividad-MINECO. M.C.O. held a pre-doctoral fellowship from the Gobierno de Castilla-La Mancha (FISCAM MOV2007-JI/20), was employed under RD07-0060-2007 and is currently supported by the grant from the Fondation ARSEP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Castro, F., Bribián, A. & Ortega, M.C. Regulation of oligodendrocyte precursor migration during development, in adulthood and in pathology. Cell. Mol. Life Sci. 70, 4355–4368 (2013). https://doi.org/10.1007/s00018-013-1365-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-013-1365-6