Summary
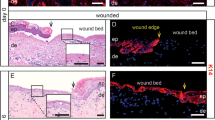
Reepithelialization of artificial partial thickness wounds made in biopsies of human skin was determined after 3, 5, or 7 d of incubation, submerged or elevated to the air-liquid interface. The biopsies were reepithelialized within 5–7 d, with a more complete epidermal healing in wounds exposed to air. Both types of wounds showed similar time-course in deposition of basement membrane components, as detected by immunofluorescence labeling. Laminin and collagen type VII were deposited underneath the migrating tips, whereas collagen type IV was detected after reepithelialization. Markers of terminal differentiation showed a pattern close to normal in the air-liquid incubated wounds after reepithelialization. Involucrin was detected in the suprabasal regions of the migrating epidermis and thereafter in the upper half of neo-epidermis in the air-liquid incubated wound. Filaggrin could not be detected in the submerged wounds at any time during healing, whereas wounds exposed to air showed a well-differentiated epidermis by Day 7. Tritiated thymidine-incorporation indicated proliferation of epidermal and dermal cells during reepithelialization and a maintained viability, as shown by cultivation of endothelial- and fibroblast-like cells obtained from the dermis 7 d after wounding.
Reepithelialization in this humanin vitro model is supported by a matrix close to normal with the possibility of extracellular influences and cell-cell interactions and, in addition, the technique is simple and reproducible. Therefore, we suggest this model for studies of regeneration in culture and as a complement toin vivo studies on epidermal healing.
Similar content being viewed by others
References
Alitalo, K.; Kuismanen, E.; Myllylä, R., et al. Extracellular matrix proteins of human epidermal keratinocytes and feeder 3T3 cells. J. Cell Biol. 94:497–505; 1982.
Asselineau, D.; Bernard, B. A.; Bailly, C., et al. Human epidermis reconstructed by culture: is it “normal”? J. Invest. Dermatol. 86:181–186; 1986.
Banks-Schlegel, S.; Green, H. Formation of epidermis by serially cultivated human epidermal cells transplanted as an epithelium to athymic mice. Transplantation. 29:308–313; 1980.
Banks-Schlegel, S.; Green, H. Involucrin synthesis and tissue assembly by keratinocytes in natural and cultured human epithelia. J. Cell Biol. 90:732–737; 1981.
Basset-Séguin, N.; Culard, J. F.; Kerai, C., et al. Reconstituted skin in culture: a simple method with optimal differentiation. Differentiation 44:232–238; 1990.
Bell, E.; Sher, S.; Hull, B., et al. The reconstitution of living skin. J. Invest. Dermatol. 81:436–438; 1983.
Bernard, B. A.; Robinson, S. M.; Vandaele, S., et al. Abnormal maturation pathway of keratinocytes in psoriatic skin. Br. J. Dermatol. 112:647–653; 1985.
Bosca, A. R.; Tinois, E.; Faure, M., et al. Epithelial differentiation of human skin equivalents after grafting onto nude mice. J. Invest. Dermatol. 91:136–141; 1988.
Briggaman, R. A.; Dalldorf, F. G.; Wheeler, C. E. J. Formation and origin of basal lamina and anchoring fibrils in adult human skin. J. Cell Biol. 51:384–395; 1971.
Clark, R. A. F.; Lanigan, J. M.; DellaPelle, P., et al. Fibronectin and fibrin provide a provisional matrix for epidermal cell migration during wound reepithelialization. J. Invest. Dermatol. 79:264–269; 1982.
Compton, C. C.; Gill, J. G.; Bradford, D. A., et al. Skin regenerated from cultured epithelial autografts on full-thickness burn wounds from 6 days to 5 years after grafting. A light, electron microscopic and immunohistochemical study. Lab. Invest. 60:600–612; 1989.
Coulomb, B.; Saiag, P.; Bell, E., et al. A new method for studying epidermalization in vitro. Br. J. Dermatol. 114:91–101; 1986.
Dale, B. A.; Ling, S. Y. Immunologic cross-reaction of stratum corneum basic protein and a keratohyalin granule protein. J. Invest. Dermatol. 72:257–261; 1979.
Demarchez, M.; Hartmann, D. J.; Herbage, D., et al. Wound healing of human skin transplanted onto the nude mouse. II. An immunohistological and ultrastructural study of the epidermal basement membrane zone reconstruction and connective tissue reorganization. Dev. Biol. 121:119–129; 1987.
Demarchez, M.; Sengel, P.; Prunieras, M. Wound healing of human skin transplanted onto the nude mouse. I. An immunohistological study of the reepithelialization process. Dev. Biol. 113:90–96; 1986.
Flaxman, B. A.; Harper, R. A. Organ culture of human skin in chemically defined medium. J. Invest. Dermatol. 64:96–99; 1975.
Foidart, J. M.; Bere, E. W.; Yaar, M., et al. Distribution and immunoelectron microscopic localization of laminin, a noncollagenous basement membrane glycoprotein. Lab. Invest. 42:336–342; 1980.
Freeman, A. E.; Igel, H. J.; Herrman, B. J., et al. Growth and characterization of human skin epithelial cell cultures. In Vitro 12:352–362; 1976.
Fuchs, E.; Green, H. Regulation of terminal differentiation of cultured human keratinocytes by vitamin A. Cell 25:617–625; 1981.
Garlick, J. A.; Taichmann, L. B. Fate of human keratinocytes during reepithelialization in an organotypic culture model. Lab. Invest. 70:916–924; 1994.
Green, H.; Kehinde, O.; Thomas, J. Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proc. Natl. Acad. Sci. USA 76:5665–5668; 1979.
Haegerstrand, A.; Gillis, C.; Bengtsson, L. Serial cultivation of adult human endothelium from the great saphenous vein. J. Vasc. Surg. 16:280–285; 1992.
Hennings, H.; Michael, D.; Cheng, C., et al. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 22:629–632; 1980.
Inoue, M.; Kratz, G.; Haegerstrand, A., et al. Collagenase expression is rapidly induced in wound edge keratinocytes after acute injury in human skin; persists during healing and stops at re-epithelialisation. J. Invest. Dermatol. 104:479–483; 1995.
Kratz, G.; Lake, M.; Gidlund, M. Insulin growth factor-1 and-2 and their role in the re-epithelialization of wounds; interactions with insulin like growth factor binding protein type 1. Scand. J. Plast. Reconstr. Surg. Hand Surg. 28:107–112; 1994.
Kratz, G.; Palmer, G.; Haegerstrand, A. Effects of keratinocyte conditioned medium, amniotic fluid and EGF in reepithelialization of human skin wounds in vitro. Eur. J. Plast. Surg. 18:209–213; 1995.
Limat, A.; Hunziker, C.; Boillat, K., et al. Post-mitotic human dermal fibroblasts efficiently support the growth of human follicular keratinocytes. J. Invest. Dermatol. 92:758–762; 1989.
Mansbridge, J. M.; Knapp, A. M. Changes in keratinocyte maturation during wound healing. J. Invest. Dermatol. 89:253–263; 1987.
McGrath, J. A.; Leigh, I. M.; Eady, R. A. J. Intracellular expression of type VII collagen during wound healing in severe recessive dystrophic epidermolysis bullosa and normal human skin. Br. J. Dermatol. 127:312–317; 1992.
Olerud, J. E.; Gown, A. M.; Bickenbach, J., et al. An assessment of human epidermal repair in elderly normal subjects using immunohistochemical methods. J. Invest. Dermatol. 90:845–850; 1988.
Ortonne, J.-P.; Löning, T.; Schmitt, D., et al. Immunomorphological and ultrastructural aspects of keratinocyte migration in epidermal wound healing. Virchows Archiv. A, Pathol. Anat. Histol. 392:217–230; 1981.
Prunieras, M.; Regnier, M.; Woodley, D. Methods for cultivation of keratinocytes with an air-liquid interface. J. Invest. Dermatol. (Supplement) 81:28s-33s; 1983.
Regauer, S.; Compton, C. C. Cultured keratinocyte sheets enhance spontaneous re-epithelialization in a dermal explant model of partial-thickness wound healing. J. Invest. Dermatol. 95:341–346; 1990.
Regauer, S.; Seiler, G. R.; Barrandon, Y., et al. Epithelial origin of cutaneous anchoring fibrils. J. Cell Biol. 111:2109–2115; 1990.
Regnier, M.; Prunieras, M.; Woodley, D. T. Growth and differentiation of adult human epidermal cells on dermal substrates. Front. Matrix Biol. 9:4–35; 1981.
Rheinwald, J. G.; Green, H. Serial cultivation of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 6:331–344; 1975.
Rigal, C.; Pieraggi, M.-T.; Serre, G., et al. Optimization of a model of full-thickness epidermal burns in the pig and immunohistochemical study of epidermodermal junction regeneration during burn healing. Dermatology 184:103–110; 1992.
Rigal, C.; Pieraggi, M.-T.; Vincent, C., et al. Healing of full-thickness cutaneous wounds in the pig. I. Immunohistochemical study of epidermodermal junction regeneration. J. Invest. Dermatol. 96:777–785; 1991.
Sakai, L. Y.; Keene, D. R.; Morris, N. P., et al. Type VII collagen is a major component of anchoring fibrils. J. Cell Biol. 106:1577–1586; 1986.
Shipley, G. D.; Pittelkow, M. R. Control of growth and differentiation in vitro of human keratinocytes cultured in serum-free medium (abstr). Arch. Dermatol. 123:1541a-1544a; 1987.
Smola, H.; Thiekötter, G.; Fusenig, N. E. Mutual induction of growth factor gene expression by epidermal-dermal cell interaction. J. Cell Biol. 122:417–429; 1993.
Stanley, J. R.; Alvarez, O. M.; Bere, W., et al. Detection of basement membrane zone antigens during epidermal wound healing in pigs. J. Invest. Dermatol. 77:240–243; 1981.
Woodley, D. T.; Peterson, H. D.; Herzog, S. R., et al. Burn wounds resurfaced by cultured epidermal autografts show abnormal reconstitution of anchoring fibrils. JAMA 259:2566–2571; 1988a.
Woodley, D. T.; Stanley, J. R.; Reese, M. J., et al. Human dermal fibroblasts synthesize laminin. J. Invest. Dermatol. 90:679–683; 1988b.
Yaoita, H.; Foidart, J. M.; Katz, S. I. Localization of the collagenous component in skin basement membrane. J. Invest. Dermatol. 70:191–193; 1978.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jansson, K., Kratz, G. & Haegerstrand, A. Characterization of a newin vitro model for studies of reepithelialization in human partial thickness wounds. In Vitro Cell.Dev.Biol.-Animal 32, 534–540 (1996). https://doi.org/10.1007/BF02722980
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02722980