Summary
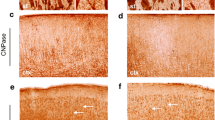
Immunocytochemical investigations were performed on Jimpy and control mouse brains using three specific anti-myelin proteolipids antisera: immunoaffinity purified multivalent anti-(PLP + DM-20) proteolipid antibodies, anti-C-terminal hexapeptide 271–276 and anti-tridecapeptide 117–129 antisera. The results show that oligodendrocytes and myelin sheaths in normal mouse brain are labelled to the same extent by the three specific antisera; in contrast, in Jimpy brain these cellular structures are only stained by the multivalent antibodies and the site-specific, anti-tridecapeptide antiserum. The absence of labelling with C-terminal hexapeptide antiserum in mutant brain is interpreted as the result of either a large deletion or a point mutation producing a frameshift in the C-terminal part of the sequences of the proteolipids PLP and DM-20. Furthermore, we show that this mutation prevents the normal transport of proteolipid molecules through the Golgi apparatus. The existence of a minor, extra-Golgi apparatus metabolic route for proteolipids to myelin structures is also discussed.
Similar content being viewed by others
References
Benjamins, J. A. &Smith, M. E. (1984) Metabolism of myelin. InMyelin (edited byMorell, P.), pp. 225–58. New York, London: Plenum Press.
Bizzozero, O. A., Soto, E. F. &Pasquini, J. M. (1983) Myelin proteolipid protein is not esterified at the site of synthesis.Neurochemistry International 5, 729–36.
Bizzozero, O. A., Soto, E. F. &Pasquini, J. M. (1984) Simultaneous entry of palmitic acid into proteolipid protein in different myelin subfractions of rat brain.Neurochemistry International 6, 659–64.
Dautigny, A., Mattei, M. G., Morello, D., Alliel, P. M., Pham-Dinh, D., Amar, L., Arnaud, D., Simon, D., Mattei, J. F., Guenet, J. L., Jolles, P. &Avner, P. (1986) The structural gene coding for myelin-associated proteolipid protein is mutated in Jimpy mice.Nature 321, 867–9.
Farkas-Bargeton, E., Robain, O. &Mandel, P. (1972) Abnormal glial maturation in the white matter of Jimpy mice: an optical study.Acta neuropathologica 21, 272–81.
Hogan, E. L. &Greenfield, S. (1984) Animal models of genetic disorders of myelin. InMyelin (edited byMorell, P.), pp. 489–534. New York, London: Plenum Press.
Kirschner, D. A., Ganser, A. L. &Caspar, D. L. D. (1984) Diffraction studies of molecular organization and membrane interactions in myelin. InMyelin (edited byMorell, P.), pp. 51–95. New York, London: Plenum Press.
Laursen, R. A., Samiullah, M. &Lees, M. B. (1984) The structure of bovine brain myelin proteolipid and its organization in myelin.Proceedings of the National Academy of Sciences USA 81, 2912–16.
Nussbaum, J. L. &Mandel, P. (1973) Brain proteolipids in neurological mutant mice.Brain Research 61, 295–310.
Nussbaum, J. L. &Roussel, G. (1983) Immunocytochemical demonstration of the transport of myelin proteolipids through the Golgi apparatus.Cell and Tissue Research 234, 547–59.
Nussbaum, J. L., Roussel, G., Wünsch, E. &Jolles, P. (1985) Site-specific antibodies to rat myelin proteolipids directed against the C-terminal hexapeptide.Journal of the Neurological Sciences 68, 89–100.
Privat, A., Drian, M. J. &Escaig, J. (1980) A freeze fracture approach of normal and pathological myelination. InNeurological Mutations Affecting Myelination, INSERM Symposium 14 (edited byBaumann, N.), pp. 131–40. Amsterdam, New York, Oxford: Elsevier/North-Holland Biochemical Press.
Privat, A., Robain, O. &Mandel, P. (1972) Aspects ultrastructuraux du corps calleux chez la souris Jimpy.Acta neuropathologica 21, 282–95.
Roach, A., Takahashi, N., Pravtcheva, D., Ruddle, F. &Hood, L. (1985) Chromosomal mapping of mouse myelin basic protein gene and structure and transcription of the partially deleted gene in shiverer mutant mice.Cell 42, 149–55.
Roussel, G. &Nussbaum, J. L. (1981) Comparative localization of Wolfgram W1 and myelin basic proteins in the rat brain during ontogenesis.Histochemical Journal 13, 1029–47.
Sidman, R. L., Dickie, M. M. &Appel, S. H. (1964) Mutant mice (Quaking and Jimpy) with deficient myelination in the central nervous system.Science 144, 309–11.
Skoff, R. P. (1982) Increased proliferation of oligodendrocytes in the hypomyelinated mouse mutant Jimpy.Brain Research 248, 19–31.
Stoffel, W., Hillen, H. &Giersiefen, H. (1984) Structure and molecular arrangement of proteolipid, protein of central nervous system myelin.Proceedings of the National Academy of Sciences USA 81, 5012–16.
Stoffel, W., Hillen, H., Schroeder, W. &Deutzmann, R. (1983) The primary structure of bovine brain myelin lipophilin (proteolipid apoprotein).Hoppe-Seyler's Zeitschrift für Physiologische Chemie 364, 1455–66.
Trifilieff, E., Luu, B., Nussbaum, J. L., Roussel, G., Espinosa De Los Monteros, A., Sabatier, J. M. &Van Rietschoten, J. (1986) A specific immunological probe for the major myelin proteolipid. Confirmation of a deletion in DM-20.FEBS letters 198, 235–9.
Trifilieff, E., Skalidis, G., Helynck, G., Lepage, P., Sorokine, O., Van Dorsselaer, A. &Luu, B. (1985) Données structurales sur le protéolipide de la myéline de masse moléculaire apparente 20 KDa (DM-20).Comptes rendus de l'Académie des Sciences Paris, Séries III 300, 241–6.
Webster, H. Def &Sternberger, N. H. (1980) Morphological features of myelin formation. InNeurological Mutations Affecting Myelination, INSERM Symposium 14 (edited byBaumann, N.), pp. 73–86. Amsterdam, New York, Oxford: Elsevier/North-Holland Biochemical Press.
Wolf, M. K. &Holden, A. B. (1969) Tissue culture analysis of the inborn defect of CNS myelination in Jimpy mice.Journal of Neuropathology and Experimental Neurology 28, 195–213.
Author information
Authors and Affiliations
Additional information
This article is dedicated to the memory of Dr A. Porte, who died on 29 August 1986 and with whom we shared many fruitful discussions.
Rights and permissions
About this article
Cite this article
Roussel, G., Neskovic, N.M., Trifilieff, E. et al. Arrest of proteolipid transport through the Golgi apparatus in Jimpy brain. J Neurocytol 16, 195–204 (1987). https://doi.org/10.1007/BF01795303
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01795303