Abstract
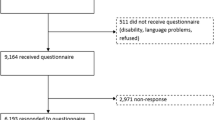
An important methodological issue in measuring health-related quality of life is whether instruments such as SF-36 and EQ can be used on an elderly population. This paper aims to test the completion, reliability and validity of the SF-36 and Euroqol on an elderly female population, and to compare them with the OPCS Disability Survey. Three hundred and eighty women aged 75 and over participated in a randomized controlled trial of the use of clodronate provided the sample. As part of the trial, patients were asked to complete the UK SF-36 and Euroqol, and the OPCS disability survey instrument administered by interview in a hospital clinic at baseline. A random subsample of respondents were retested six months later. The SF-36 achieved poorer levels of completion by dimension (68.1%–88.9%) than the OPCS (99.2%) and Euroqol (84%–93.5%) instruments. There were no major floor effects in the distribution of scores, except for the role dimensions of SF-36. Correlation between test-retest were significant for all instruments, but lower for the role dimensions and social functioning of SF-36, and these dimensions also had 95% Cls for the mean differences in excess of 10 points. There was substantial agreement between the three instruments, and evidence for their construct validity against age and recent use of health services. The sensitivities of the instruments were tested through hypothetical changes in health status. There was some evidence of greater sensitivity to lower levels of morbidity in the SF-36. Where brevity is required and the health changes are expected to be substantial, then EQ may be sufficient. For greater sensitivity SF-36 seems to have an advantage, however lower completion rates and problems with consistency suggest it requires adaptation. One solution would be to use interviewer administration. Another would be to change the SF-36 to make it more suitable for use in elderly people, although this may reduce its usefulness as a generic instrument.
Similar content being viewed by others
References
McHorney CA, Ware JE, Lu JFR, et al. The MOS 36 item Short-Form Health Survey (SF-36): III. Tests of data quality assumptions and reliability across diverse patient groups. Med Care 1994; 32: 40–52.
Stewart AL, Hays RD, Ware JE. The MOS short-form General Health Survey: II. reliability and validity in a patient population. Med Care 1988; 26(7): 724–735.
Brazier JE, Harper R, Jones NMB, OCathain A, Thomas KJ, Usherwood T, Westlake L. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. BMJ 1992; 305: 160–164.
McHorney CA, Ware JE, Raczek AK. The MOS 36 item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 1993; 31: 247–263.
Garratt AM, Ruta DA, Abdalla MI, et al. The SF-36 health survey questionnaire: an outcome measure suitable for routine use within the NHS. BMJ 1993; 306: 1440–1444.
The Euroqol Group. Euroqol—a facility for the measurement of health related quality of life. Health policy 1990; 16: 199–228.
Research Group on the Measurement & Valuation of Health. An interim tariff for Euroqol health states. York: University of York, 1994.
Brazier JE, Howard P, Williams BT. MRC Report: ‘Comparison of Outcome measures for patients’ with COPD. Grant no: G9203850. Sheffield: SCHARR, 1994.
Martin J, Meltzer H, Elliot D. OPCS Surveys of disability in Great Britain. Report 1. The prevalence of disability among adults. London: HMSO, 1988.
Ware JE, Sherbourne CD. The MOS 36 item Short-Form Health Survey (SF-36): I. Conceptual framework and item selection. Med Care 1992; 30: 473–483.
Ware JE, Brook RH, Williams KN et al. Conceptualisation and measurement of health for adults in the health insurance study. Vol 1: Model of health and methodology. Santa Monica CA: The RAND Corporation (Publication no. R-1987/1-HEW), 1980.
Streiner DL, Norman GR. Health measurement scales: a practical guide to their development and use. Oxford: Oxford University Press, 1989.
McDowell I, Newell C. Measuring health: a guide to rating scales and questionnaires. New York: Oxford University Press, 1987.
Kerlinger FN. Foundations of behavioural research. New York: Holt, Rinehart & Winston, 1973.
Bland JM, Altman DG. Statistical methods for assessing agreement two methods of clinical measurement. Lancet 1986; 1: 307–310.
Ziebland S. The short form 36 health status questionnaire: clues from the Oxford regions normative data about its usefulness in measuring health gain in population surveys. J Epidemiol Community Health 1995; 49: 102–105.
Kazis L, Anderson JA, Meenan RF. Effect sizes for interpreting changes in health status. Med Care 1989; 27(5): S178-S189.
Cohen J. Statistical Power analysis for the behavioural science. New York Academic Press, 1977.
OPCS. General Household Survey 1987. London: HMSO, 1988.
Medical Outcomes Trust. Scoring Exercise for the SF-36 Health Survey. 1994
Hayes V, Morris J, Wolfe C, Morgan M. The SF-36 Health Survey Questionnaire: Is it suitable for use with older adults? Age and Ageing 1995; 24: 120–125.
Brazier JE, Jones N, Kind P. Testing the validity of the Euroqol and comparing it with the SF-36 health survey questionnaire. Qual Life Res 1993; 2: 169–180.
Brazier JE. The Short-Form 36 (SF-36) Health Survey and its use in Pharmacoeconomic evaluation. Economics 1995; 7(5): 403–415.
Harries U, Hill S. Measuring Outcomes. Health Service Journal 1994; 22 September.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brazier, J.E., Walters, S.J., Nicholl, J.P. et al. Using the SF-36 and Euroqol on an elderly population. Qual Life Res 5, 195–204 (1996). https://doi.org/10.1007/BF00434741
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00434741