Summary
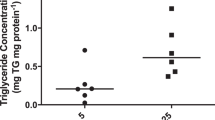
Rats were made diabetic by the injection of streptozotocin on day 12 of pregnancy and were investigated on days 17 and 20 of pregnancy. A significant correlation between both maternal plasma triglyceride and nonesterified fatty acid levels with placental or fetal triglyceride content was found, although fetal weight did not change significantly. In order to investigate the source of the placental and fetal fat the rats received, intragastrically 24 h earlier, 1-14C-triolein (as a preformed fatty acid tracer) and tritiated water (as a marker of de novo fatty acid synthesis). Several maternal tissues, placenta, and fetuses were extracted and analysed for fatty acid radioactivity. Compared with non-diabetic pregnant rats, maternal 14C-label storage was reduced. In contrast, the placental and fetal tissues demonstrated a significant rise in 14C-label, similar in magnitude to the rise in their triglyceride content. The pattern of 3H incorporation also indicated a pronounced decrease in maternal fatty acid synthesis, but no increase in de novo fatty acid synthesis in the fetus or placenta. The findings show that fetal lipids in the pregnant rat originate both from maternal fatty acids and those synthesized in situ. The diabetes-induced increment in fetal triglyceride content is derived, however, from preformed maternal triglycerides or non-esterified fatty acids secondary to the increase in their concentration in the maternal circulation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pedersen J (1977) The pregnant diabetic and her newborn, 2nd edn. Williams & Wilkins, Baltimore, pp 106–122
Szabo AJ, Szabo O (1974) Placental free fatty acid transfer and fetal adipose tissue development: An explanation of fetal adiposity in infants of diabetic mothers. Lancet 2: 498–499
Szabo AJ, Grimaldi RD (1970) The metabolism of the placenta. Adv Metab Disorders 4: 186–228
Robertson AF, Sprecher H (1968) A review of placental lipid metabolism and' transport. Acta Paediatr Scand 183 (Suppl): 3–18
Hull D, Elphick MC (1979) Evidence for fatty acid transfer across the human placenta. In: Beard RW, Hoet JJ (eds) Pregnancy metabolism, diabetes and the fetus. Ciba Foundation Symposium, Excerpta Medica, Amsterdam, pp 75–86
Hull D, Elphick M (1979) Transfer of fatty acids. In: Chamberlain GVP, Wilkinson AW (eds) Placental transfer. Pitman, London, pp 159–165
Foster BW, Bloom B (1963) The synthesis of fatty acids by rat liver slices in tritiated water. J Biol Chem 238: 882–892
Ho RJ (1970) Radiochemical assay of long-chain fatty acids using 68 Ni as tracer. Anal Biochem 36: 105–113
Folch J, Less M, Sloane-Stanley GH (1951) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 225: 497–509
Hershfield MS, Nemeth AM (1968) Placental transport of free palmitic and linoleic acids in the guinea pig. J Lipid Res 9: 460–468
Thomas CR, Lowy C, Bacchus RA, Yeoman DK (1977) Transfer of fatty acid across the guinea pig placenta. Diabetologia 13: 436 (Abstract)
Van Duyne CM, Havel RJ, Felts YM (1962) Placental transfer of palmitic acid-1-C14 in rabbits. Am J Obstet Gynecol 84: 1069–1074
Edson JL, Hull D (1975) Evidence for increased fatty acid transfer across the placenta during a maternal fast in rabbits. Biol Neonate 27: 50–55
Van Duyne GM, Parker HR, Havel RJ, Holm LW (1960) Free fatty acid metabolism in fetal and newborn sheep. Am J Physiol 199: 987–990
Koren Z, Shafrir E (1964) Placental transfer of free fatty acids in the pregnant rat. Proc Soc Exp Biol Med 116: 411–414
Hummel L, Schirrmeister N, Zimmermann T, Wagner H (1974) Studies on the lipid metabolism using 1-14C-palmitate in fetal rats. Biol Neonate 24: 298–305
Elphick MC, Filshie GM, Hull D (1978) The passage of fat emulsion across human placenta. Br J Obstet Gynaecol 85: 610–618
Diamant YZ, Diamant S, Freinkel N (1980) Lipid deposition and metabolism in rat placenta during gestation. Placenta 1: 319–325
Mallov S, Alousi AA (1965) Lipoprotein lipase activity of rat and human placenta. Proc Soc Exp Biol Med 119: 301–306
Kervran A, Girard JR (1974) Glucose induced increase of plasma insulin in the rat foetus in utero. J Endocrinol 62: 545–551
Diamant YZ, Shafrir E (1979) Placental enzymes of glycolysis, gluconeogenesis and lipogenesis in the diabetic rat and in starvation. Comparison with maternal and fetal liver. Diabetologia 15: 481–485
Prager R, Abramovici A, Liban E, Laron Z (1974) Histopathological changes in the placenta of streptozotocin induced diabetic rats. Diabetologia 10: 89–91
Kim YS, Jatoi I, Kim Y (1980) Neonatal macrosomia in maternal diabetes. Diabetologia 18: 407–411
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shafrir, E., Khassis, S. Maternal-fetal fat transport versus new fat synthesis in the pregnant diabetic rat. Diabetologia 22, 111–117 (1982). https://doi.org/10.1007/BF00254839
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00254839