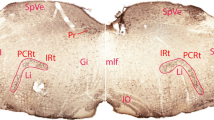
Summary
Single and double retrograde tracer techniques were employed in cats to investigate: (1) the topographical relationships between supraspinal neurons projecting to either the brachial or lumbosacral enlargement, (2) the distribution and relative frequency of single supraspinal neurons which project to both enlargements by means of axonal branching.
In one group of cats large injections of horseradish peroxidase (HRP) were made throughout either the brachial or lumbosacral enlargement. The results from these experiments support recent observations on the multiplicity of brainstem centers giving origin to descending spinal pathways and provide evidence for a population of corticospinal neurons in area 6.
In a second set of experiments, HRP was injected in one enlargement, and 3H-apo-HRP (enzymatically inactive) was injected in the other enlargement. Relatively large numbers of neurons with collateral projections to both enlargements (double-labeled) were observed in the medullary and pontine reticular formation, the medial and inferior vestibular nuclei bilaterally, the ipsilateral lateral vestibular nucleus, Edinger-Westphal nucleus, caudal midline raphe nuclei and nuclear regions surrounding the brachium conjunctivum. By contrast, double-labeled neurons were infrequently observed in the red nucleus and sensorimotor cortex, contralateral to the injections.
In the red nucleus, lateral vestibular nucleus and sensorimotor cortex, neurons projecting to the brachial enlargement were largely segregated topographically from neurons projecting to the lumbosacral enlargement. However, there was some overlap, and double-labeled neurons were consistently observed within the region of overlap. In the sensorimotor cortex, the overlap between brachial- and lumbar-projecting neurons was most prominent in areas 4 and 3a, along the cruciate sulcus, but also involved other cytoarchitectonic regions in the medial aspect of the hemisphere.
Similar content being viewed by others
Abbreviations
- AM:
-
nucleus ambiguus
- ap:
-
area postrema
- aq:
-
aqueduct
- BC:
-
brachium conjunctivum
- ci:
-
central inferior nucleus of the raphe
- cs:
-
central superior nucleus of the raphe
- Cun:
-
cuneate nucleus
- EC:
-
external cuneate nucleus
- EW:
-
Edinger-Westphal nucleus
- ETC:
-
central tegmental field
- FTG:
-
gigantocellular tegmental field
- FTL:
-
lateral tegmental field
- FTM:
-
magnocellular tegmental field
- FTP:
-
paralemniscal tegmental field
- Gr:
-
gracile nucleus
- IO:
-
inferior olive
- K-F:
-
Kölliker-Fuse nucleus
- LC:
-
nucleus locus coeruleus
- li:
-
rostral linear nucleus of the raphe
- LR:
-
lateral reticular nucleus
- mlf:
-
medial longitudinal fasciculus
- PAG:
-
periaqueductal gray
- PbL:
-
lateral parabrachial nucleus
- PG:
-
pontine gray
- PON:
-
preolivary nucleus
- ppr:
-
post-pyramidal nucleus of the raphe
- RB:
-
restiform body
- RNm:
-
red nucleus, magnocellular division
- RNp:
-
red nucleus, parvocellular division
- SC:
-
superior colliculus
- SN:
-
substantia nigra
- SOl:
-
lateral nucleus of the superior olive
- SOm:
-
medial nucleus of the superior olive
- Spin V:
-
spinal trigeminal nucleus
- SubC:
-
nucleus subcoeruleus
- TB:
-
trapezoid body
- tb:
-
nucleus of the trapezoid body
- trm:
-
tegmental reticular nucleus
- VInf:
-
inferior vestibular nucleus
- VLd:
-
lateral vestibular nucleus, dorsal division
- VLv:
-
lateral vestibular nucleus, ventral division
- VM:
-
medial vestibular nucleus
- VSm:
-
superior vestibular nucleus, medial division
- III:
-
oculomotor nucleus or nerve
- V:
-
sensory nucleus of the trigeminal nerve
- VI:
-
abducens nucleus
- VII I:
-
facial nucleus, lateral part
- VII m:
-
facial nucleus, medial part
- X:
-
vagus nucleus
- XII:
-
hypoglossal nucleus
References
Abzug C, Maeda M, Peterson BW, Wilson VJ (1973) Branching of individual vestibulospinal axons at different spinal cord levels. Brain Res 56: 327–330
Abzug C, Maeda M, Peterson BW, Wilson VJ (1974) Cervical branching of lumbar vestibulospinal axons. J Physiol (Lond) 243: 499–522
Armand J, Aurenty R (1977) Dual organization of motor corticospinal tract in the cat. Neurosci Lett 6: 1–7
Armand J, Padel Y, Smith AM (1974) Somatotopic organization of the corticospinal tract in cat motor cortex. Brain Res 74: 209–227
Basbaum A, Fields H (1979) The origin of descending pathways in the dorsolateral funiculus of the spinal cord of the cat and rat. Further studies on the anatomy of pain modulation. J Comp Neurol 187: 513–532
Basbaum A, Marley N, O'Keefe J (1976) Spinal cord pathways involved in the production of analgesia by brain stimulation. In: Bonica IJJ, Albe-Fessard D (eds) Advances in pain research, vol 1. Raven Press, New York, pp 511–515
Beall JE, Martin RF, Applebaum AE, Willis WD (1976) Inhibition of primate spinothalamic tract neurons by stimulation in the nucleus raphe magnus. Brain Res 114: 328–333
Berman AL (1968) The brainstem of the cat. University of Wisconsin Press, Madison
Berrevoets CE, Kuypers HGJM (1975) Pericruciate cortical neurons projecting to brain stem reticular formation, dorsal column nuclei, and spinal cord in the cat. Neurosci Lett 1: 257–262
Biedenbach MA, DeVito JL (1979) Cortical location of pyramidal tract neurons established with HRP. Neurosci Abstr 5: 113
Brodal A, Taber E, Walberg F (1960) The raphe nuclei of the brainstem in the cat. II. Efferent connections. J Comp Neurol 114: 239–259
Brodal A, Pompeiano O, Walberg F (1962) The vestibular nuclei and their connections, anatomy and functional correlations. Oliver and Boyd, Edinburgh
Burton H, Loewy AD (1977) Projections to the spinal cord from medullary somatosensory relay nuclei. J Comp Neurol 173: 773–792
Catsman-Berrevoets CE, Kuypers HGJM (1979) Differences in distribution of corticospinal axon collaterals to thalamus and midbrain tegmentum in cat, demonstrated by means of double retrograde fluorescent labeling of cortical neurons. Neurosci Lett [Suppl] 3: 133
Chambers WW, Liu CN (1957) Cortico-spinal tract of the cat. An attempt to correlate the pattern of degeneration with deficits in reflex activity following neocortical lesions. J Comp Neurol 108: 23–55
Chu N-S, Bloom FE (1974) The catecholamine-containing neurons in the cat dorsolateral pontine tegmentum; distribution of the cell bodies and some axonal projections. Brain Res 66: 1–21
Coulter JD, Ewing L, Carter C (1976) Origin of primary sensorimotor cortical projections to lumbar spinal cord of cat and monkey. Brain Res 103: 366–372
Dahlstrom A, Fuxe K (1964) Evidence of the existence of monoamine neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of neurons. Acta Physiol Scand 62 [Suppl] 232: 1–55
Delgado JMR (1952) Hidden motor cortex of the cat. Am J Physiol 170: 673–681
Delgado JMR, Livingston R (1955) Motor representation in the frontal sulci of the cat. Yale J Biol Med 28: 245–252
Eccles JC, Rantucci T, Sheid P, Táboříková H (1975) Somatotopic studies on red nucleus: spinal projection level and respective receptive fields. J Neurophysiol 38: 965–980
Engberg I, Ryall RW (1966) The inhibitory action of noradrenaline and other monoamines on spinal neurones. J Physiol (Lond) 185: 298–322
Engberg I, Lundberg A, Ryall RW (1968) Reticulospinal inhibition of interneurons. J Physiol (Lond) 194: 225–236
Feiten DL, Laties AM, Carpenter MB (1974) Monoamine- containing cell bodies in the squirrel monkey brain. Am J Anat 139: 153–166
Fields H, Basbaum A, Clanton C, Anderson SD (1977) Nucleus raphe magnus inhibition of spinal cord dorsal horn neurons. Brain Res 126: 441–453
Flindt-Egebak P (1977) Autoradiographical demonstration of the projections from the limb areas of the feline sensorimotor cortex to the spinal cord. Brain Res 136: 153–156
Futami T, Shinoda Y, Yokota J (1979) Spinal axon collaterals of corticospinal neurons identified by intracellular injection of horseradish peroxidase. Brain Res 164: 279–284
Fuxe K (1965) IV. Distribution of monoamine terminals in the central nervous system. Acta Physiol Scand 64 [Suppl] 247: 37–85
Garol HW (1942) The “motor” cortex of the cat. J Neuropathol 1: 139–145
Garver DL, Sladek JR (1975) Monoamine distribution in primate brain. Catecholamine-containing perikarya in the brain stem of Macaca speciosa. J Comp Neurol 159: 289–304
Groos WP, Ewing LK, Carter CW, Coulter JD (1978) Organization of corticospinal neurons in the cat. Brain Res 143: 393–419
Guilbaud G, Oliveras JL, Giesler G, Besson JM (1977) Effects induced by stimulation of the centralis inferior nucleus of the raphe on dorsal horn interneurons in cat's spinal cord. Brain Res 126: 355–360
Haber LH, Martin RF, Chatt AB, Willis WD (1978) Effects of stimulation in nucleus reticularis gigantocellularis on the activity of spinothalamic tract neurons in the monkey. Brain Res 153: 163–168
Hancock MB, Fougerousse CL (1976) Spinal projections from the nucleus locus coeruleus and nucleus subcoeruleus in the cat and monkey as demonstrated by the retrograde transport of horseradish peroxidase. Brain Res Bull 1: 229–334
Hanker JS, Yates PE, Metz CB, Rustioni A (1977) A new specific, sensitive, and non-carcinogenic reagent for the demonstration of horseradish peroxidase. Histochem J 9: 789–792
Hassler R (1966) Extrapyramidal motor areas of cat's frontal lobe. Their function and architectonic differentiation. Int J Neurol 5: 301–306
Hassler R, Muhs-Clement K (1964) Architektonischer Aufbau des sensorimotorischen und parietalen Cortex der Katze. J Hirnforsch 6: 377–420
Hayes NL, Rustioni A (1979a) Dual projections of single neurons are visualized simultaneously. Use of enzymatically inactive 3H-HRP. Brain Res 165: 321–326
Hayes NL, Rustioni A (1979b) Cells of origin of descending pathways to the cervical and lumbar spinal cord of the cat. The question of collateral projections. Anat Rec 193: 563
Hicks SP, D'Amato CJ (1977) Locating corticospinal neurons by retrograde axonal transport of horseradish peroxidase. Exp Neurol 56: 410–420
Illert M, Lundberg A, Padel Y, Tanaka R (1975) Convergence on propriospinal neurons which may mediate disynaptic corticospinal excitation to forelimb motoneurones in the cat. Brain Res 93: 530–534
Jordon LM, Kenshalo DR, Martin RF, Haber LH, Willis WD (1978) Depression of primate spinothalamic tract neurons by iontophoretic application of 5-hydroxytryptamine. Pain 5: 135–142
Jouvet M, Delorme F (1965) Locus coeruleus et sommeil paradoxal. C R Seances Soc Biol Paris 159: 895–899
Kneisley LW, Biber MP, LaVail JH (1978) A study of the origin of brain stem projections to monkey spinal cord using the retrograde transport method. Exp Neurol 60: 116–139
Kuypers HGJM (1963) The organization of the “motor system”. Int J Neurol 4: 78–91
Kuypers HGJM (1964) The descending pathways to the spinal cord, their anatomy and function. In: Eccles JC, Schadé JP (eds) Progress in brain research, vol 3. Elsevier, Amsterdam, pp 178–202
Kuypers HGJM, Maisky VA (1975) Retrograde axonal transport of horseradish peroxidase from spinal cord to brain stem cell groups in the cat. Neurosci Lett 1: 9–14
Kuypers HGJM, Maisky VA (1977) Funicular trajectories of descending brain stem pathways in cat. Brain Res 136: 159–165
LeBars D, Menetrey D, Besson J (1976) Effects of morphine upon lamina V type cell activities in the dorsal horn of the decerebrate cat. Brain Res 113: 293–310
Livingston A, Phillips CG (1957) Maps and thresholds for the sensorimotor cortex of the cat. Q J Exp Physiol 42: 190–205
Loewy AD, Saper CB (1978) Edinger-Westphal nucleus. Projections to the brainstem and spinal cord in the cat. Brain Res 150: 1–27
Lundberg A (1966) Integration in the reflex pathway. In: Granit R (ed) Nobel symposium. I. Muscular afferents and motor control. Almquist and Wikell, Stockholm, pp 275–305
Lundberg A (1967) The supraspinal control of transmission in spinal reflex pathways. In: Widen L (ed) Recent advances in clinical neurophysiology. Electroenceph Clin Neurophysiol [Suppl] 25: 35–46
MacKibben PS, Wheelis DR (1932) Experiments on the motor cortex of the cat. J Comp Neurol 56: 373–390
Maisky VA, Kuypers HGJM (1978a) Issledovanie provodyashchikh putei spinnogo mozga koshki s pomoshchu metoda eksperimental'nogo retrogradnogo aksonnogo transporta peroksidazy khrena. Neirofiziol 10: 125–132
Maisky VA, Kuypers HGJM (1978b) Issledovanie neironov supraspinal'nykh sistem mozga koshki s pomoshchu metoda retrogradnogo aksonnogo transporta peroksidazy khrena. Neirofiziol 10: 115–124
Martin RF (1977) Retrograde labeling of medullary raphe nuclei neurons in the monkey following spinal cord injections of horseradish peroxidase. Anat Rec 187: 645
Martin RF, Jordan LM, Willis WD (1978) Differential projections of cat medullary raphe neurons demonstrated by retrograde labeling following spinal cord lesions. J Comp Neurol 182: 77–88
Martin RF, Haber LH, Willis WD (1979) Primary afferent depolarization of identified cutaneous fibers following stimulation in medial brainstem. J Neurophysiol 42: 779–790
Mesulam MM (1978) Tetramethylbenzidine for horseradish peroxidase neurohistochemistry. A non-carcinogenic blue reaction product with superior sensitivity for visualizing neural afferents and efferents. J Histochem Cytochem 26: 106–117
Nieoullon A, Rispal-Padel L (1973) Organization spatiale du cortex somato-moteur du chat, etablie par stimulation intracorticale chez l'animal en preparation chronique. CR Acad Sci Paris 276: 2709–2712
Nieoullon A, Rispal-Padel L (1976) Somatotopic localization in cat motor cortex. Brain Res 105: 405–422
Nyberg-Hansen R, Brodal A (1963) Sites of termination of corticospinal fibers in the cat. An experimental study with silver impregnation methods. J Comp Neurol 120: 369–391
Nyberg-Hansen R, Brodal A (1964) Sites and mode of termination of rubrospinal fibers in the cat. J Anat 98: 235–253
Oliveras JL, Redjemi F, Guilbaud G, Besson JM (1975) Analgesia induced by electrical stimulation of the inferior centralis nucleus of the raphe in the cat. Pain 1: 139–145
Peterson BW, Coulter JD (1977) A new long spinal projection from the vestibular nuclei in the cat. Brain Res 122: 351–356
Peterson BW, Maunz RA, Pitts NG, Mackel RG (1975) Patterns of projection and branching of reticulospinal neurons. Exp Brain Res 23: 333–351
Pompeiano O (1973) Reticular formation. In: Iggo A (ed) Handbook of sensory physiology, vol 2. Springer, Berlin Heidelberg New York, pp 381–488
Pompeiano O, Brodal A (1957) Experimental demonstration of a somatotopical origin of rubrospinal fibers in the cat. J Comp Neurol 108: 225–251
Scheibel ME, Scheibel AB (1966) Terminal axonal patterns in cat spinal cord. I. The lateral corticospinal tract. Brain Res 2: 333–350
Shinoda Y, Yamaguchi T (1978) The intraspinal branching patterns of fast and slow pyramidal tract neurons in the cat. J Physiol (Paris) 74: 237–238
Shinoda Y, Arnold AP, Asanuma H (1976) Spinal branching of corticospinal axons in the cat. Exp Brain Res 26: 215–234
Shinoda Y, Ghez C, Arnold A (1977) Spinal branching of rubrospinal axons in the cat. Exp Brain Res 30: 203–218
Siegel JM, McGinty DJ (1977) Pontine reticular formation neurons. Relationship of discharge to motor activity. Science 196: 678–680
Sugimoto T, Itoh K, Mizuno N (1978) Direct projections from the Edinger-Westphal nucleus to the cerebellum and spinal cord in the cat: an HRP study. Neurosci Lett 9: 17–22
Taber E, Brodal A, Walberg F (1960) Raphe nuclei of the brainstem of the cat. I. Cytoarchitecture. J Comp Neurol 114: 161–188
Tohyama M, Sakai T, Touret M, Salvert D, Jouvet M (1979) Spinal projections from the lower brain stem in the cat as demonstrated by the horseradish peroxidase technique. II. Projections from the dorsolateral pontine tegmentum and raphe nuclei. Brain Res 176: 215–231
Ward JW, Clark SL (1975) Specific responses elicitable from subdivisions of the motor cortex of the cerebrum of the cat. J Comp Neurol 63: 49–64
Willis WD, Haber LH, Martin RF (1977) Inhibition of spinothalamic tract cells and interneurons by brainstem stimulation in the monkey. J Neurophysiol 40: 968–981
Wilson VJ, Peterson BW (1978) Peripheral and central substrates of vestibulospinal reflexes. Physiol Rev 58: 80–105
Wilson VJ, Kato M, Peterson BW, Wylie RM (1967) A single unit analysis of the organization of Deiters' nucleus. J Neurophysiol 30: 603–618
Woolsey CN (1958) Organization of somatic sensory and motor areas of the cerebral cortex. In: Harlow H, Woolsey CN (eds) Biological and biochemical bases of behavior. University of Wisconsin Press, Madison, pp 63–81
Author information
Authors and Affiliations
Additional information
The research was supported by USPHS grants NS 12440 and MH 14277. 3H-apo-HRP was generously provided by New England Nuclear
Rights and permissions
About this article
Cite this article
Hayes, N.L., Rustioni, A. Descending projections from brainstem and sensorimotor cortex to spinal enlargements in the cat. Exp Brain Res 41, 89–107 (1981). https://doi.org/10.1007/BF00236598
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00236598