Abstract
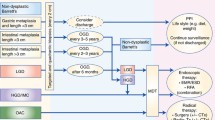
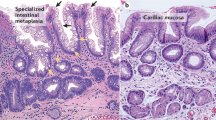
Gastric cancer has been declining for more than half a century, whereas the incidence of oesophageal cancer is increasing rapidly. The histopathological subtype is also changing with a predominance of oesophageal adenocarcinoma compared with squamous carcinoma. The reasons for these epidemiological changes are not clear, although population-based data have implicated gastro-oesophageal reflux disease as a risk factor. In susceptible individuals reflux of duodeno-gastric contents can lead to the development of a columnar-lined oesophagus, commonly called Barrett’s oesophagus. This can then progress to adenocarcinoma via a metaplasia-dysplasia-carcinoma sequence. At the current time, the mortality from oesophageal adenocarcinoma exceeds 80% at 5 years. Therefore, endoscopic surveillance programmes have been generally recommended for patients with Barrett’s oesophagus in an attempt to detect early, curable lesions. Unfortunately these programmes are cumbersome and costly and have not yet been proved to reduce population mortality. In order to improve patient outcomes we need to be able to identify patients at high risk and to understand the triggers for disease progression. There is mounting evidence that there is an underlying genetic susceptibility to Barrett’s oesophagus and oesophageal adenocarcinoma. However, this is likely to be as a result of multiple low penetrance susceptibility genes which have yet to be identified. Once patients are identified as having Barrett’s oesophagus their chance for developing adenocarcinoma is in the order of 0.5%-1% per year. The histological assessment of dysplasia as a predictor of cancer development is highly subjective. Therefore multiple, specific somatic mutations in the tissue have been investigated as potential biomarkers. The most promising markers to date are the presence of aneuploidy, loss of heterozygosity of p53 and cyclin D1 overexpression. However, a study of evolutionary relationships suggest that mutations occur in no obligate order. Combinatorial approaches are therefore being advocated which include genomic profiling or the use of a panel of molecular markers in order to define the common molecular signatures that can then be used to predict malignant progression. An alternative approach would be to use markers for the final common pathway following genetic instability, which is the loss of proliferative control. We have demonstrated an increase in the expression of a novel proliferation marker, Mcm2, which occurs during the malignant progression of Barrett’s oesophagus. These Mcm2-expressing cells are detectable on the surface, and hence a cytological approach may be applicable. In view of the role of reflux components in the pathogenesis of Barrett’s oesophagus the effect of acid and bile on the cell phenotype have been studied. These studies have demonstrated that pulsatile acid and bile exposure induce cell proliferation. The mechanism for the hyperproliferative response appears to involve p38 mitogen activated protein kinase (MAPK) pathways as well as protein kinase C (PKC) and cyclo-oxygenases. A clinical implication of the laboratory studies is that suppression of acid and bile may need to be profound in order to suppress cell proliferation and, by inference, ultimately prevent the development of dysplasia. There is some support for this concept from short-term clinical studies, and a large randomised chemoprevention trial is being instigated which will evaluate the effect of proton pump inhibitors with or without aspirin. Given the epidemic increase in oesophageal adenocarcinoma and the dismal 5-year mortality rate, a radical approach is necessary to prevent cancer development in individuals with pre-malignant lesions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA (1999) Cancer statistics, 1999. CA Cancer J Clin 49:8–31
Devesa SS, Blot WJ, Fraumeni JF Jr (1998) Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. Cancer 83:2049–2053
Berrino F, Capocaccia R, Esteve J, et al (1999) Survival of cancer patients in Europe— the EUROCARE-2 study. International Agency for Research on Cancer (IARC), vol. 151
Wild CP, Hardie LJ (2003) Reflux, Barrett’s oesophagus and adenocarcinoma: burning questions. Nat Rev Cancer 3:676–684
Souza RF, Morales CP, Spechler SJ (2001) A conceptual approach to understanding the molecular mechanisms of cancer development in Barrett’s oesophagus. Aliment Pharmacol Ther 15:1087–1100
Guindi M, Riddell RH (2003) Histology of Barrett’s esophagus and dysplasia. Gastrointest Endosc Clin N Am 13:349–368, viii
Lagergren J, Bergstrom R, Lindgren A, Nyren O (1999) Symptomatic gastroesophageal reflux as a risk factor for oesophageal adenocarcinoma. N Engl J Med 340:825–832
Cameron AJ, Zinsmeister AR, Ballard DJ, Carney JA (1990) Prevalence of columnarlined (Barrett’s) esophagus. Comparison of population-based clinical and autopsy findings. Gastroenterology 99:1918–1922
Cameron A, Kamath P, Carpenter H (1997) Prevalence of Barrett’s esophagus and intestinal metaplasia at the esophagogastric junction. Gastroenterology 112:A82
Shaheen NJ, Crosby MA, Bozymski EM, Sandler RS (2000) Is there publication bias in the reporting of cancer risk in Barrett’s esophagus? Gastroenterology 119:333–338
Conio M, Blanchi S, Lapertosa G, Ferraris R, Sablich R, Marchi S, D’Onofrio V, Lacchin T, Iaquinto G, Missale G, Ravelli P, Cestari R, Benedetti G, Macri G, Fiocca R, Munizzi F, Filiberti R (2003) Long-term endoscopic surveillance of patients with Barrett’s esophagus. Incidence of dysplasia and adenocarcinoma: a prospective study. Am J Gastroenterol 98:1931–1939
Murray L, Watson P, Johnston B, Sloan J, Mainie IM, Gavin A (2003) Risk of adeno carcinoma in Barrett’s oesophagus: population based study. Br Med J 327:534–535
Mandal A, Playford R, Wicks A (2003) Current practice in surveillance strategy for patients with Barrett’s oesophagus in the UK. Aliment Pharmacol Ther 15:1319–1324
Sampliner RE (2002) Updated guidelines for the diagnosis, surveillance, and therapy of Barrett’s esophagus. Am J Gastroenterol 97:1888–1895
Eckardt VF, Kanzler G, Bernhard G (2001) Life expectancy and cancer risk in patients with Barrett’s esophagus: a prospective controlled investigation. Am J Med 111:33–37
Macdonald CE, Wicks AC, Playford RJ (2000) Final results from 10 year cohort of patients undergoing surveillance for Barrett’s oesophagus: observational study. BMJ 321:1252–1255
Corley DA, Levin TR, Habel LA, Weiss NS, Buffler PA (2002) Surveillance and survival in Barrett’s adenocarcinomas: a population-based study. Gastroenterology 122:633–640
Lieberman D, Oehlke M, Helfand M (1997) Risk factors for Barrett’s eosphagus in community-based practice, GORGE consortium. Gastroenterology Outcomes Research Group in Endoscopy. Am J Gastroenterol 92:1293–1297
Spechler SJ, Barr H (2004) Screening and surveillance of Barrett’s oesophagus: what is a cost-effective framework? Aliment Pharmacol Ther 19 Suppl 1:49–53
Gerson LB, Edson R, Lavori PW, Triadafilopoulos G (2001) Use of a simple symptom questionnaire to predict Barrett’s esophagus in patients with symptoms of gastroesophageal reflux. Am J Gastroenterol 96:2005–2012
Locke GR, Zinsmeister AR, Talley NJ (2003) Can symptoms predict endoscopic findings in GERD? Gastrointest Endosc 58(5):661–670
Gerson LB, Shetler K, Triadafilopoulos G (2002) Prevalence of Barrett’s esophagus in asymptomatic individuals. Gastroenterology 123:461–467
Rex DK, Cummings OW, Shaw M, Cumings MD, Wong RK, Vasudeva RS, Dunne D, Rahmani EY, Helper DJ (2003) Screening for Barrett’s esophagus in colonoscopy patients with and without heartburn. Gastroenterology 125:1670–1677
Romero Y, Locke GR 3rd (1999) Is there a GERD gene? Am J Gastroenterol 94(5):1127–1129
Drovdlic CM, Goddard KA, Chak A, Brock W, Chessler L, King JF, Richter J, Falk GW, Johnston DK, Fisher JL, Grady WM, Lemeshow S, Eng C (2003) Demographic and phenotypic features of 70 families segregating Barrett’s oesophagus and oesophageal adenocarcinoma. J Med Genet 40:651–656
Romero Y, Cameron AJ, Locke GR 3rd, Schaid DJ, Slezak JM, Branch CD, Melton LJ 3rd (1997) Familial aggregation of gastroesophageal reflux in patients with Barrett’s esophagus and esophageal adenocarcinoma. Gastroenterology 113:1449–1456
Cameron AJ, Lagergren J, Henriksson C, Nyren O, Locke GR 3rd, Pedersen NL (2002) Gastroesophageal reflux disease in monozygotic and dizygotic twins. Gastroenterology 122:55–59
Hu FZ, Preston RA, Post JC, White GJ, Kikuchi LW, Wang X, Leal SM, Levenstien MA, Ott J, Self TW, Allen G, Stiffler RS, McGraw C, Pulsifer-Anderson EA, Ehrlich GD (2000) Mapping of a gene for severe pediatric gastroesophageal reflux to chromosome 13q14. Jama 284:325–334
Gopal DV, Lieberman DA, Magaret N, Fennerty MB, Sampliner RE, Garewal HS, Falk GW, Faigel DO (2003) Risk factors for dysplasia in patients with Barrett’s esophagus (BE): results from a multicenter consortium. Dig Dis Sci 48:1537–1541
Hillman LC, Chiragakis L, Clarke AC, Kaushik SP, Kaye GL (2003) Barrett’s esophagus: macroscopic markers and the prediction of dysplasia and adenocarcinoma. J Gastroenterol Hepatol 18:526–533
Barrett MT, Pritchard D, Palanca-Wessels C, Anderson J, Reid BJ, Rabinovitch PS (2003) Molecular phenotype of spontaneously arising 4 N (G2-tetraploid) intermediates of neoplastic progression in Barrett’s esophagus. Cancer Res 63:4211–4217
Prevo LJ, Sanchez CA, Galipeau PC, Reid BJ (1999) p53-mutant clones and field effects in Barrett’s esophagus. Cancer Res 59:4784–4787
Paulson TG, Galipeau PC, Reid BJ (1999) Loss of heterozygosity analysis using whole genome amplification, cell sorting, and fluorescence-based PCR. Genome Res 9:482–492
Reid BJ, Prevo LJ, Galipeau PC, Sanchez CA, Longton G, Levine DS, Blount PL, Rabinovitch PS (2001) Predictors of progression in Barrett’s esophagus II: baseline 17p (p53) loss of heterozygosity identifies a patient subset at increased risk for neoplastic progression. Am J Gastroenterol 96:2839–2848
Dolan K, Walker SJ, Gosney J, Field JK, Sutton R (2003) TP53 mutations in malignant and premalignant Barrett’s esophagus. Dis Esophagus 16:83–89
Reid BJ, Levine DS, Longton G, Blount PL, Rabinovitch PS (2000) Predictors of progression to cancer in Barrett’s esophagus: baseline histology and flow cytometry identify low-and high-risk patient subsets. Am J Gastroenterol 95:1669–1676
Iravani S, Zhang HQ, Yuan ZQ, Cheng JQ, Karl RC, Jove R, Coppola D (2003) Modification of insulin-like growth factor 1 receptor, c-Src, and Bcl-XL protein expression during the progression of Barrett’s neoplasia. Hum Pathol 34:975–982
Barrett MT, Sanchez CA, Prevo LJ, Wong DJ, Galipeau PC, Paulson TG, Rabinovitch PS, Reid BJ (1999) Evolution of neoplastic cell lineages in Barrett oesophagus. Nat Genet 22:106–109
Selaru FM, Zou T, Xu Y, Shustova V, Yin J, Mori Y, Sato F, Wang S, Olaru A, Shibata D, Greenwald BD, Krasna MJ, Abraham JM, Meltzer SJ (2002) Global gene expression profiling in Barrett’s esophagus and esophageal cancer: a comparative analysis using cDNA microarrays. Oncogene 21:475–478
Xu Y, Selaru FM, Yin J, Zou TT, Shustova V, Mori Y, Sato F, Liu TC, Olaru A, Wang S, Kimos MC, Perry K, Desai K, Greenwald BD, Krasna MJ, Shibata D, Abraham JM, Meltzer SJ (2002) Artificial neural networks and gene filtering distinguish between global gene expression profiles of Barrett’s esophagus and esophageal cancer. Cancer Res 62:3493–3497
Mei R, Galipeau PC, Prass C, Berno A, Ghandour G, Patil N, Wolff RK, Chee MS, Reid BJ, Lockhart DJ (2000) Genome-wide detection of allelic imbalance using human SNPs and high-density DNA arrays. Genome Res 10:1126–1137
Sirieix PS, O’Donovan M, Brown J, Save V, Coleman N, Fitzgerald RC (2003) Surface expression of mini-chromosome maintenance proteins provides a novel method for detecting patients at risk for developing adenocarcinoma in Barrett’s oesophagus. Clin Cancer Res 9:2560–2566
Going JJ, Keith WN, Neilson L, Stoeber K, Stuart RC, Williams GH (2002) Aberrant expression of minichromosome maintenance proteins 2 and 5, and Ki-67 in dysplastic squamous oesophageal epithelium and Barrett’s mucosa. Gut 50:373–377
Falk GW (2003) Cytology in Barrett’s esophagus. Gastrointest Endosc Clin N Am 13:335–348
Sonnenberg A, Fennerty MB (2003) Medical decision analysis of chemoprevention against esophageal adenocarcinoma. Gastroenterology 124:1758–1766
Rudolph RE, Vaughan TL, Kristal AR, Blount PL, Levine DS, Galipeau PC, Prevo LJ, Sanchez CA, Rabinovitch PS, Reid BJ (2003) Serum selenium levels in relation to markers of neoplastic progression among persons with Barrett’s esophagus. J Natl Cancer Inst 95:750–757
Schatzkin A, Gail M (2002) The promise and peril of surrogate end points in cancer research. Nat Rev Cancer 2:19–27
Fitzgerald RC, Omary MB, Triadafilopoulos G (1996) Dynamic effects of acid on Barrett’s esophagus: an ex vivo differentiation and proliferation model. J Clin Invest 98:2120–2128
Umansky M, Yasui W, Hallak A, Brill S, Shapira I, Halpern Z, Hibshoosh H, Rattan J, Meltzer S, Tahara E, Arber N (2001) Proton pump inhibitors reduce cell cycle abnormalities in Barrett’s esophagus. Oncogene 20:7987–7991
Fitzgerald R, Traidafilopoulos G (2001) Review article: Barrett’s oesophagus, dysplasia and pharmacologic acid suppression. Aliment Pharmacol Ther 15:269–276
Corey KE, Schmitz SM, Shaheen NJ (2003) Does a surgical antireflux procedure decrease the incidence of esophageal adenocarcinoma in Barrett’s esophagus? A metaanalysis. Am J Gastroenterol 98:2390–2394
Haigh CR, Attwood SE, Thompson DG, Jankowski JA, Kirton CM, Pritchard DM, Varro A, Dimaline R (2003) Gastrin induces proliferation in Barrett’s metaplasia through activation of the CCK2 receptor. Gastroenterology 124:615–625
Fitzgerald RC, Abdalla S (2003) Gastrin-induced hyperproliferation in Barrett’s esophagus. Gastroenterology 125:1921; author reply 1921-1922
Buttar NS, Wang KK, Leontovich O, Westcott JY, Pacifico RJ, Anderson MA, Krishnadath KK, Lutzke LS, Burgart LJ (2002) Chemoprevention of esophageal adenocarcinoma by COX-2 inhibitors in an animal model of Barrett’s esophagus. Gastroenterology 122:1101–1112
Lagorce C, Paraf F, Vidaud D, Couvelard A, Wendum D, Martin A, Flejou JF (2003) Cyclooxygenase-2 is expressed frequently and early in Barrett’s oesophagus and associated adenocarcinoma. Histopathology 42:457–465
Bosetti C, Talamini R, Franceschi S, Negri E, Garavello W, La Vecchia C (2003) Aspirin use and cancers of the upper aerodigestive tract. Br J Cancer 88:672–674
Corley DA, Kerlikowske K, Verma R, Buffler P (2003) Protective association of aspirin/NSAIDs and esophageal cancer: a systematic review and meta-analysis. Gastroenterology 124:47–56
Heath EI, Canto MI, Wu TT, Piantadosi S, Hawk E, Unalp A, Gordon G, Forastiere AA; CBET Research Group (2003) Chemoprevention for Barrett’s esophagus trial. Design and outcome measures. Dis Esophagus 16:177–186
Jankowski J, Sharma P (2004) Approaches to Barrett’s oesophagus treatment-the role of proton pump inhibitors and other interventions. Aliment Pharmacol Ther 19 Suppl 1:54–59
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2005 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Fitzgerald, R.C. (2005). Genetics and Prevention of Oesophageal Adenocarcinoma. In: Senn, HJ., Morant, R. (eds) Tumor Prevention and Genetics III. Recent Results in Cancer Research, vol 166. Springer, Berlin, Heidelberg. https://doi.org/10.1007/3-540-26980-0_3
Download citation
DOI: https://doi.org/10.1007/3-540-26980-0_3
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-22228-6
Online ISBN: 978-3-540-26980-9
eBook Packages: MedicineMedicine (R0)