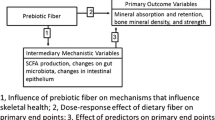
Abstract
Gut microbiota (GM) located within the intestinal tract lumen comprises the largest number of cells (10E14) in the human body. The gut microbiome refers to the collection of genomes and genes present in gut microbiota. GM can vary according to age, sex, genetic background, immune status, geography, diet, prebiotics, which are non-digestible fibers metabolized in the distal part of the gastrointestinal tract, probiotics, which are micro-organisms conferring a health benefit on the host when administered in adequate amounts, living conditions, diseases and drugs. A source of probiotics is fortified fermented dairy products, which in addition provide calcium, protein, phosphorus and various micronutrients. Bone homeostasis is influenced by GM composition and/or products. GM appears to be a major player in the various determinants of bone health. However, it remains to be demonstrated in well conducted long-term randomized controlled trials, whether interventions changing GM composition and/or function are capable of reducing fracture risk.


Similar content being viewed by others
References
Consortium HMP (2012) Structure, function and diversity of the healthy human microbiome. Nature 486:207–214
Gordon JI (2012) Honor thy gut symbionts redux. Science 336:1251–1253
Lucas S, Omata Y, Hofmann J et al (2018) Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat Commun 9:55
Cani PD (2017) Gut cell metabolism shapes the microbiome. Science 357:548–549
Valles Y, Artacho A, Pascual-Garcia A et al (2014) Microbial succession in the gut: directional trends of taxonomic and functional change in a birth cohort of Spanish infants. PLoS Genet 10:e1004406
Avershina E, Storro O, Oien T et al (2014) Major faecal microbiota shifts in composition and diversity with age in a geographically restricted cohort of mothers and their children. FEMS Microbiol Ecol 87:280–290
Cho I, Blaser MJ (2012) The human microbiome: at the interface of health and disease. Nat Rev Genet 13:260–270
Lahti L, Salojarvi J, Salonen A et al (2014) Tipping elements in the human intestinal ecosystem. Nat Commun 5:4344
Claesson MJ, Cusack S, O’Sullivan O et al (2011) Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc Natl Acad Sci USA 108:4586–4591
Claesson MJ, Jeffery IB, Conde S et al (2012) Gut microbiota composition correlates with diet and health in the elderly. Nature 488:178–184
Biagi E, Nylund L, Candela M et al (2010) Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PLoS One 5:e10667
David LA, Maurice CF, Carmody RN et al (2014) Diet rapidly and reproducibly alters the human gut microbiome. Nature 505:559–563
Beaumont M, Portune KJ, Steuer N et al (2017) Quantity and source of dietary protein influence metabolite production by gut microbiota and rectal mucosa gene expression: a randomized, parallel, double-blind trial in overweight humans. Am J Clin Nutr 106:1005–1019
Faith JJ, Guruge JL, Charbonneau M et al (2013) The long-term stability of the human gut microbiota. Science 341:1237439
Devkota S (2016) MICROBIOME. Prescription drugs obscure microbiome analyses. Science 351:452–453
Maier L, Pruteanu M, Kuhn M et al (2018) Extensive impact of non-antibiotic drugs on human gut bacteria. Nature 555:623–628
Sekirov I, Russell SL, Antunes LC et al (2010) Gut microbiota in health and disease. Physiol Rev 90:859–904
Ticinesi A, Lauretani F, Milani C et al (2017) Aging gut microbiota at the cross-road between nutrition, physical frailty, and sarcopenia: is there a gut-muscle axis? Nutrients 9:1303. https://doi.org/10.3390/nu9121303
Grosicki GJ, Fielding RA, Lustgarten MS (2018) Gut microbiota contribute to age-related changes in skeletal muscle size, composition, and function: biological basis for a gut-muscle axis. Calcif Tissue Int 102:433–442
Ticinesi A, Tana C, Nouvenne A (2019) The intestinal microbiome and its relevance for functionality in older persons. Curr Opin Clin Nutr Metab Care 22:4–12
Rizzoli R (2018) Diet, microbiota, and bone health. In: Weaver CM, Bischoff-Ferrari H, Daly R, Wong MS (eds) Nutritional influences on bone health. Springer Nature, Cham, pp 143–168
Sjogren K, Engdahl C, Henning P et al (2012) The gut microbiota regulates bone mass in mice. J Bone Miner Res 27:1357–1367
Schwarzer M, Strigini M, Leulier F (2018) Gut microbiota and host juvenile growth. Calcif Tissue Int 102:387–405. https://doi.org/10.1007/s00223-017-0368-y
Yan J, Herzog JW, Tsang K et al (2016) Gut microbiota induce IGF-1 and promote bone formation and growth. Proc Natl Acad Sci USA 113:E7554–E7563
Fransen F, Zagato E, Mazzini E et al (2015) BALB/c and C57BL/6 mice differ in polyreactive IgA abundance, which impacts the generation of antigen-specific IgA and microbiota diversity. Immunity 43:527–540
Weaver CM (2015) Diet, gut microbiome, and bone health. Curr Osteoporos Rep 13:125–130
Druart C, Alligier M, Salazar N et al (2014) Modulation of the gut microbiota by nutrients with prebiotic and probiotic properties. Adv Nutr 5:624 s–633 s
Denou E, Marcinko K, Surette MG et al (2016) High-intensity exercise training increases the diversity and metabolic capacity of the mouse distal gut microbiota during diet-induced obesity. Am J Physiol Endocrinol Metab 310:E982–E993
Suez J, Korem T, Zeevi D et al (2014) Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 514:181–186
Mitsou EK, Kakali A, Antonopoulou S et al (2017) Adherence to the Mediterranean diet is associated with the gut microbiota pattern and gastrointestinal characteristics in an adult population. Br J Nutr 117:1645–1655
De Filippis F, Pellegrini N, Vannini L et al (2016) High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 65:1812–1821
Tosti V, Bertozzi B, Fontana L (2018) Health benefits of the mediterranean diet: metabolic and molecular mechanisms. J Gerontol A Biol Sci Med Sci 73:318–326
Byberg L, Bellavia A, Larsson SC et al (2016) Mediterranean diet and hip fracture in Swedish men and women. J Bone Miner Res 31:2098–2105
Haring B, Crandall CJ, Wu C et al (2016) Dietary patterns and fractures in postmenopausal women: results from the women’s health initiative. JAMA Intern Med 176:645–652
Smith MI, Yatsunenko T, Manary MJ et al (2013) Gut microbiomes of Malawian twin pairs discordant for kwashiorkor. Science 339:548–554
Blanton LV, Charbonneau MR, Salih T et al (2016) Gut bacteria that prevent growth impairments transmitted by microbiota from malnourished children. Science. https://doi.org/10.1126/science.aad3311
Whisner CM, Castillo LF (2018) Prebiotics, bone and mineral metabolism. Calcif Tissue Int 102:443–479
Garcia-Vieyra MI, Del Real A, Lopez MG (2014) Agave fructans: their effect on mineral absorption and bone mineral content. J Med Food 17:1247–1255
Weaver CM, Martin BR, Nakatsu CH et al (2011) Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J Agric Food Chem 59:6501–6510
Ammann P, Rizzoli R, Fleisch H (1988) Influence of the disaccharide lactitol on intestinal absorption and body retention of calcium in rats. J Nutr 118:793–795
Lee C, Kim BG, Kim JH et al (2017) Sodium butyrate inhibits the NF-kappa B signaling pathway and histone deacetylation, and attenuates experimental colitis in an IL-10 independent manner. Int Immunopharmacol 51:47–56
Langlands SJ, Hopkins MJ, Coleman N et al (2004) Prebiotic carbohydrates modify the mucosa associated microflora of the human large bowel. Gut 53:1610–1616
Bindels LB, Delzenne NM, Cani PD et al (2015) Towards a more comprehensive concept for prebiotics. Nat Rev Gastroenterol Hepatol 12:303–310
Macfarlane S, Macfarlane GT, Cummings JH (2006) Review article: prebiotics in the gastrointestinal tract. Aliment Pharmacol Ther 24:701–714
Scholz-Ahrens KE, Ade P, Marten B et al (2007) Prebiotics, probiotics, and synbiotics affect mineral absorption, bone mineral content, and bone structure. J Nutr 137:838 s–846 s
Mitamura R, Hara H (2006) Ingestion of difructose anhydride III partially restores calcium absorption impaired by vitamin D and estrogen deficiency in rats. Eur J Nutr 45:242–249
Ohta A, Uehara M, Sakai K et al (2002) A combination of dietary fructooligosaccharides and isoflavone conjugates increases femoral bone mineral density and equol production in ovariectomized mice. J Nutr 132:2048–2054
Zafar TA, Weaver CM, Zhao Y et al (2004) Nondigestible oligosaccharides increase calcium absorption and suppress bone resorption in ovariectomized rats. J Nutr 134:399–402
Ammann P, Rizzoli R, Fleisch H (1986) Calcium absorption in rat large intestine in vivo: availability of dietary calcium. Am J Physiol 251:G14–G18
Mathey J, Puel C, Kati-Coulibaly S et al (2004) Fructooligosaccharides maximize bone-sparing effects of soy isoflavone-enriched diet in the ovariectomized rat. Calcif Tissue Int 75:169–179
Yang LC, Wu JB, Lu TJ et al (2013) The prebiotic effect of Anoectochilus formosanus and its consequences on bone health. Br J Nutr 109:1779–1788
Muhlbauer RC, Li F (1999) Effect of vegetables on bone metabolism. Nature 401:343–344
Muhlbauer RC, Lozano A, Reinli A (2002) Onion and a mixture of vegetables, salads, and herbs affect bone resorption in the rat by a mechanism independent of their base excess. J Bone Miner Res 17:1230–1236
Chaplin A, Parra P, Laraichi S et al (2016) Calcium supplementation modulates gut microbiota in a prebiotic manner in dietary obese mice. Mol Nutr Food Res 60:468–480
Charbonneau MR, O’Donnell D, Blanton LV et al (2016) Sialylated milk oligosaccharides promote microbiota-dependent growth in models of infant undernutrition. Cell 164:859–871
Sjogren YM, Tomicic S, Lundberg A et al (2009) Influence of early gut microbiota on the maturation of childhood mucosal and systemic immune responses. Clin Exp Allergy 39:1842–1851
He Y, Liu S, Leone S et al (2014) Human colostrum oligosaccharides modulate major immunologic pathways of immature human intestine. Mucosal Immunol 7:1326–1339
Peng L, Li ZR, Green RS et al (2009) Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J Nutr 139:1619–1625
Rizzoli R, Bianchi ML, Garabedian M et al (2010) Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone 46:294–305
Abrams SA, Griffin IJ, Hawthorne KM et al (2005) A combination of prebiotic short- and long-chain inulin-type fructans enhances calcium absorption and bone mineralization in young adolescents. Am J Clin Nutr 82:471–476
van den Heuvel EG, Muys T, van Dokkum W et al (1999) Oligofructose stimulates calcium absorption in adolescents. Am J Clin Nutr 69:544–548
Whisner CM, Martin BR, Schoterman MH et al (2013) Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: a double-blind cross-over trial. Br J Nutr 110:1292–1303
Whisner CM, Martin BR, Nakatsu CH et al (2014) Soluble maize fibre affects short-term calcium absorption in adolescent boys and girls: a randomised controlled trial using dual stable isotopic tracers. Br J Nutr 112:446–456
Slevin MM, Allsopp PJ, Magee PJ et al (2014) Supplementation with calcium and short-chain fructo-oligosaccharides affects markers of bone turnover but not bone mineral density in postmenopausal women. J Nutr 144:297–304
Whisner CM, Martin BR, Nakatsu CH et al (2016) Soluble corn fiber increases calcium absorption associated with shifts in the gut microbiome: a randomized dose-response trial in free-living pubertal females. J Nutr 146:1298–1306
McCabe LR, Parameswaran N (2018) Advances in probiotic regulation of bone and mineral metabolism. Calcif Tissue Int 102:480–488
McCabe LR, Irwin R, Schaefer L et al (2013) Probiotic use decreases intestinal inflammation and increases bone density in healthy male but not female mice. J Cell Physiol 228:1793–1798
Britton RA, Irwin R, Quach D et al (2014) Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J Cell Physiol 229:1822–1830
Zhang J, Motyl KJ, Irwin R et al (2015) Loss of bone and Wnt10b expression in male type 1 diabetic mice is blocked by the probiotic Lactobacillus reuteri. Endocrinology 156:3169–3182
Ohlsson C, Engdahl C, Fak F et al (2014) Probiotics protect mice from ovariectomy-induced cortical bone loss. PLoS One 9:e92368
Chiang SS, Pan TM (2011) Antiosteoporotic effects of Lactobacillus -fermented soy skim milk on bone mineral density and the microstructure of femoral bone in ovariectomized mice. J Agric Food Chem 59:7734–7742
Narva M, Rissanen J, Halleen J et al (2007) Effects of bioactive peptide, valyl-prolyl-proline (VPP), and lactobacillus helveticus fermented milk containing VPP on bone loss in ovariectomized rats. Ann Nutr Metab 51:65–74
Parvaneh K, Ebrahimi M, Sabran MR et al (2015) Probiotics (Bifidobacterium longum) increase bone mass density and upregulate sparc and Bmp-2 genes in rats with bone loss resulting from ovariectomy. Biomed Res Int 2015:897639
Li JY, Chassaing B, Tyagi AM et al (2016) Sex steroid deficiency-associated bone loss is microbiota dependent and prevented by probiotics. J Clin Investig 126:2049–2063
Rizzoli R, Biver E (2018) Effects of fermented milk products on bone. Calcif Tissue Int 102:489–500
Alvaro E, Andrieux C, Rochet V et al (2007) Composition and metabolism of the intestinal microbiota in consumers and non-consumers of yogurt. Br J Nutr 97:126–133
Sahni S, Tucker KL, Kiel DP et al (2013) Milk and yogurt consumption are linked with higher bone mineral density but not with hip fracture: the Framingham Offspring Study. Arch Osteoporos 8:119
Biver E, Durosier-Izart C, Merminod F et al (2018) Fermented dairy products consumption is associated with attenuated cortical bone loss independently of total calcium, protein, and energy intakes in healthy postmenopausal women. Osteoporos Int 29:1771–1782
Laird E, Molloy AM, McNulty H et al (2017) Greater yogurt consumption is associated with increased bone mineral density and physical function in older adults. Osteoporos Int 28:2409–2419
Michaelsson K, Wolk A, Lemming EW et al (2018) Intake of milk or fermented milk combined with fruit and vegetable consumption in relation to hip fracture rates: a cohort study of Swedish women. J Bone Miner Res 33:449–457
Michaëlsson K, Wolk A, Langenskiöld S et al (2014) Milk intake and risk of mortality and fractures in women and men: cohort studies. BMJ 349:g6015
Radavelli-Bagatini S, Zhu K et al (2014) Dairy food intake, peripheral bone structure, and muscle mass in elderly ambulatory women. J Bone Miner Res 29:1691–1700
Rizzoli R, Biver E, Bonjour JP et al (2018) Benefits and safety of dietary protein for bone health-an expert consensus paper endorsed by the European Society for Clinical and Economical Aspects of Osteoporosis, Osteoarthritis, and Musculoskeletal Diseases and by the International Osteoporosis Foundation. Osteoporos Int 29:1933–1948. https://doi.org/10.1007/s00198-018-4534-5
Jones ML, Martoni CJ, Prakash S (2013) Oral supplementation with probiotic L. reuteri NCIMB 30242 increases mean circulating 25-hydroxyvitamin D: a post hoc analysis of a randomized controlled trial. J Clin Endocrinol Metab 98:2944–2951
Nilsson AG, Sundh D, Backhed F et al (2018) Lactobacillus reuteri reduces bone loss in older women with low bone mineral density: a randomized, placebo-controlled, double-blind, clinical trial. J Intern Med. https://doi.org/10.1111/joim.12805
McNulty NP, Yatsunenko T, Hsiao A et al (2011) The impact of a consortium of fermented milk strains on the gut microbiome of gnotobiotic mice and monozygotic twins. Sci Transl Med 3:106ra106
Waterhouse M, Hope B, Krause L et al (2018) Vitamin D and the gut microbiome: a systematic review of in vivo studies. Eur J Nutr. https://doi.org/10.1007/s00394-018-1842-7
Kong J, Zhang Z, Musch MW et al (2008) Novel role of the vitamin D receptor in maintaining the integrity of the intestinal mucosal barrier. Am J Physiol Gastrointest Liver Physiol 294:G208–G216
Jin D, Wu S, Zhang YG et al (2015) Lack of vitamin D receptor causes dysbiosis and changes the functions of the murine intestinal microbiome. Clin Ther 37:996–1009.e1007
Luthold RV, Fernandes GR, Franco-de-Moraes AC et al (2017) Gut microbiota interactions with the immunomodulatory role of vitamin D in normal individuals. Metabolism 69:76–86
Wu S, Yoon S, Zhang YG et al (2015) Vitamin D receptor pathway is required for probiotic protection in colitis. Am J Physiol Gastrointest Liver Physiol 309:G341–G349
Bashir M, Prietl B, Tauschmann M et al (2016) Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur J Nutr 55:1479–1489
Collins FL, Schepper JD, Rios-Arce ND et al (2017) Immunology of gut-bone signaling. Adv Exp Med Biol 1033:59–94
Mineo H, Amano M, Minaminida K et al (2006) Two-week feeding of difructose anhydride III enhances calcium absorptive activity with epithelial cell proliferation in isolated rat cecal mucosa. Nutrition 22:312–320
Donohoe DR, Garge N, Zhang X et al (2011) The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab 13:517–526
Ma N, Guo P, Zhang J et al (2018) Nutrients mediate intestinal bacteria-mucosal immune crosstalk. Front Immunol 9:5
Beaumont M, Andriamihaja M, Armand L et al (2017) Epithelial response to a high-protein diet in rat colon. BMC Genom 18:116
Macpherson AJ, Harris NL (2004) Interactions between commensal intestinal bacteria and the immune system. Nat Rev Immunol 4:478–485
Ammann P, Rizzoli R, Bonjour JP et al (1997) Transgenic mice expressing soluble tumor necrosis factor-receptor are protected against bone loss caused by estrogen deficiency. J Clin Investig 99:1699–1703
Terashima A, Takayanagi H (2018) Overview of osteoimmunology. Calcif Tissue Int 102:503–511. https://doi.org/10.1007/s00223-018-0417-1
Li JY, Tawfeek H, Bedi B et al (2011) Ovariectomy disregulates osteoblast and osteoclast formation through the T-cell receptor CD40 ligand. Proc Natl Acad Sci USA 108:768–773
Lee HW, Suh JH, Kim AY et al (2006) Histone deacetylase 1-mediated histone modification regulates osteoblast differentiation. Mol Endocrinol 20:2432–2443
Katono T, Kawato T, Tanabe N et al (2008) Sodium butyrate stimulates mineralized nodule formation and osteoprotegerin expression by human osteoblasts. Arch Oral Biol 53:903–909
Albenberg LG, Wu GD (2014) Diet and the intestinal microbiome: associations, functions, and implications for health and disease. Gastroenterology 146:1564–1572
Rowland I, Gibson G, Heinken A et al (2018) Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr 57:1–24
Rizzoli R, Bonjour JP (2006) Physiology of calcium and phosphate homeostasis. In: Seibel MJ, Robins SP, Bilezikian JP (eds) Dynamics of bone and cartilage metabolism: principles and clinical applications. Academic Press, San Diego, pp 345–360
Yadav VK, Ryu JH, Suda N et al (2008) Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell 135:825–837
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
RR has received fees for consultancy or lecture from Danone, EffRx, Nestlé, ObsEva, Pfizer, Radius Health, Sandoz and TEVA/Theramex.
Statement of human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of paper, formal consent is not required.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rizzoli, R. Nutritional influence on bone: role of gut microbiota. Aging Clin Exp Res 31, 743–751 (2019). https://doi.org/10.1007/s40520-019-01131-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-019-01131-8