Abstract
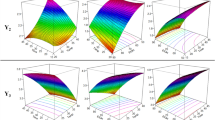
The objective of this study was to optimize the extraction conditions of phenolic and flavonoids compounds from quinoa (Chenopodium quinoa) seeds using ultrasound assistance technology. A randomized central composite face-centered design was used to evaluate the effect of extraction temperature, ethanol concentration in the solvent, and ultrasound power on the total phenolic content (TPC), total flavonoid content (TFC) and antioxidant activity by response surface analysis. Predicted model equations were obtained to describe the experimental data regarding TPC, TFC and antioxidant activity, with significant variation in the linear, quadratic, and interaction effects of the independent variables. Regression analysis showed that more than 88 % of the variability was explained by the models. The best extraction conditions obtained by simultaneous maximization of the responses were: extraction temperature of 60 °C, 80 % ethanol as solvent and non-application of ultrasounds. Under the optimal conditions, the corresponding predicted response values were 103.6 mg GAE/100 g dry weight (dw), 25.0 mg quercetin equiv./100 g dw and 28.6 % DPPH radical scavenging, for TPC, TFC and antioxidant activity, respectively. The experimental values agreed with those predicted within a 95 % confidence level, indicating the suitability of the employed model. HPLC analysis of the obtained extracts confirmed the highest phenolic compound yield in the extract obtained under optimal extraction conditions. Considering the characteristics of the antioxidant-rich extracts obtained, they could be consider for potential application in the food industry, as nutraceutical and functional foods ingredient or well as replacement of synthetic antioxidants.





Similar content being viewed by others
References
Abugoch James LE (2009) Quinoa (Chenopodium quinoa Willd.): composition, chemistry, nutritional, and functional properties. Adv Food Nutr Res 58:1–31
Alvarez-Jubete L, Wijngaard H, Arendt EK, Gallagher E (2010) Polyphenol composition and in vitro antioxidant activity of amaranth, quinoa, buckwheat and wheat as affected by sprouting and baking. Food Chem 119:770–778
Bhargava A, Shukla S, Ohri D (2006) Chenopodium quinoa—An Indian perspective. Ind Crop Prod 23:73–87
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28:25–30
Carciochi RA, Manrique GD, Dimitrov K (2014) Changes in phenolic composition and antioxidant activity during germination of quinoa seeds (Chenopodium quinoa Willd.). Int Food Res J 21:767–773
Carrera C, Ruiz-Rodríguez A, Palma M, Barroso CG (2012) Ultrasound assisted extraction of phenolic compounds from grapes. Anal Chim Acta 732:100–104
Galvan d’Alessandro L, Kriaa K, Nikov I, Dimitrov K (2012) Ultrasound assisted extraction of polyphenols from black chokeberry. Sep Purif Technol 93:42–47
Da Porto C, Porretto E, Decorti D (2013) Comparison of ultrasound-assisted extraction with conventional extraction methods of oil and polyphenols from grape (Vitis vinifera L.) seeds. Ultrason Sonochem 20:1076–1080
Dini I, Tenore GC, Dini A (2010) Antioxidant compound contents and antioxidant activity before and after cooking in sweet and bitter Chenopodium quinoa seeds. LWT Food Sci Technol 43:447–451
Durling NE, Catchpole OJ, Grey JB, Webby RF, Mitchell KA, Foo LY, Perry NB (2007) Extraction of phenolics and essential oil from dried sage (Salvia officinalis) using ethanol-water mixtures. Food Chem 101:1417–1424
Hamsaveni DR, Prapulla SG, Divakar S (2001) Response surface methodological approach for the synthesis of isobutyl isobutyrate. Process Biochem 36:1103–1109
Hirose Y, Fujita T, Ishii T, Ueno N (2010) Antioxidative properties and flavonoid composition of Chenopodium quinoa seeds cultivated in Japan. Food Chem 119:1300–1306
Koleva II, Van Beek TA, Linssen JPH, De Groot A, Evstatieva LN (2002) Screening of plant extracts for antioxidant activity: a comparative study on three testing methods. Phytochem Anal 13:8–17
Konishi Y, Hirano S, Tsuboi H, Wada M (2004) Distribution of minerals in quinoa (Chenopodium quinoa Willd.) seeds. Biosci Biotechnol Biochem 68:231–234
Liyana-Pathirana C, Shahidi F (2005) Optimization of extraction of phenolic compounds from wheat using response surface methodology. Food Chem 93:47–56
Ma YQ, Ye XQ, Fang ZX, Chen JC, Xu GH, Liu DH (2008) Phenolic compounds and antioxidant activity of extracts from ultrasonic treatment of satsuma mandarin (Citrus unshiu Marc.) peels. J Agric Food Chem 56:5682–5690
Madhujith T, Shahidi F (2006) Optimization of the extraction of antioxidative constituents of six barley cultivars and their antioxidant properties. J Agric Food Chem 54:8048–8057
Pingret D, Fabiano-Tixier AS, Chemat F (2013) Degradation during application of ultrasound in food processing: a review. Food Control 31:593–606
Repo-Carrasco-Valencia R, Hellström JK, Pihlava JM, Mattila PH (2010) Flavonoids and other phenolic compounds in Andean indigenous grains: Quinoa (Chenopodium quinoa), kañiwa (Chenopodium pallidicaule) and kiwicha (Amaranthus caudatus). Food Chem 120:128–133
Rice-Evans CA, Miller NJ, Paganga G (1997) Antioxidant properties of phenolic compounds. Trends Plant Sci 2:152–159
Silva EM, Rogez H, Larondelle Y (2007) Optimization of extraction of phenolics from Inga edulis leaves using response surface methodology. Sep Purif Technol 55:381–387
Singleton VL, Orthofer R, Lamuela-Raventós RM (1998) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol 299:152–178
Spigno G, De Faveri DM (2007) Antioxidants from grape stalks and marc: Influence of extraction procedure on yield, purity and antioxidant power of the extracts. J Food Eng 78:793–801
Vega-Gálvez A, Miranda M, Vergara J, Uribe E, Puente L, Martínez EA (2010) Nutrition facts and functional potential of quinoa (Chenopodium quinoa willd.), an ancient Andean grain: a review. J Sci Food Agric 90:2541–2547
Acknowledgments
The authors would like to thank the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina) and Eurotango II (Erasmus Mundus programme) for the parcial support of this project (Ph.D. fellowships granted to Carciochi).
Conflict of interest
The authors declare that they have no conflicts of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carciochi, R.A., Manrique, G.D. & Dimitrov, K. Optimization of antioxidant phenolic compounds extraction from quinoa (Chenopodium quinoa) seeds. J Food Sci Technol 52, 4396–4404 (2015). https://doi.org/10.1007/s13197-014-1514-4
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-014-1514-4