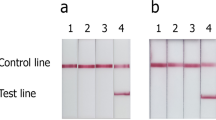
Abstract
For many years, microscopic examination of stool samples has been considered to be the “gold standard” for diagnosis of intestinal parasites although the Polymerase Chain Reaction (PCR) analysis is increasingly utilized due to its high accuracy. Recently, PCR has been approved by the World Health Organization as the current method of choice for the diagnosis of Entamoeba histolytica infection. In this study we evaluated a novel immunochromatographic antigen detection rapid test, ImmunoCardSTAT CGE (Meridian Bioscence, Milan, Italy), which has been proposed for the diagnosis of infections caused by Cryptosporidium parvum-Giardia intestinalis-Entamoeba histolytica. There is another rapid test with a similar name, the ImmunoCard STAT! Crypto/Giardia, but it is just for Cryptosporidium and Giardia. We aimed to compare E. histolytica results obtained from the rapid test with those of a rt-PCR for the detection of E. histolytica / E. dispar DNA. The new ImmunoCard rapid antigen detection test exhibited 88% sensitivity and 92% specificity (if assessed on rt-PCR negative samples) but showed a high proportion of cross-reaction between the pathogenic E. histolytica and the non pathogenic E. dispar.
Résumé
Depuis de nombreuses années, l’examen microscopique des selles a été considéré comme le « gold standard » pour le diagnostic des parasites intestinaux, bien que l’analyse de réaction en chaîne par polymérase (PCR) soit de plus en plus utilisée en raison de sa grande précision. Récemment, la PCR a été approuvée par l’Organisation mondiale de la santé comme méthode de choix pour le diagnostic de l’infection par Entamoeba histolytica. Dans cette étude, nous avons évalué un test rapide de détection d’antigène par immunochromatographie, ImmunoCardSTAT CGE (Meridian Bioscence, Milan, Italie), qui a été proposé pour le diagnostic des infections causées par Cryptosporidium parvum-Giardia intestinalis-Entamoeba histolytica. Nous avons cherché à comparer, pour E. histolytica, les résultats obtenus à partir du test rapide avec ceux d’une rt-PCR pour la détection de ADN de E. histolytica / E. dispar. Le nouveau test de détection rapide ImmunoCard présentait 88 % de sensibilité et 92 % de spécificité (si appréciée sur les échantillons négatifs en RT-PCR), mais a montré une forte proportion de réactions croisées entre la pathogène E. histolytica et la non pathogène E. dispar.
Similar content being viewed by others
References
Ali IK, Hossain MB, Roy S, et al (2003) Entamoeba moshkovskii infections in children, Bangladesh. Emerg Infect Di 9(5):580–4
Caballero-Salcedo A, Viveros-Rogel M, Salvatierra B, et al (1994) Seroepidemiology of amebiasis in Mexico. Am J Trop Med Hyg 50(4):412–9
Diamond LS, Clark CG (1993) A redescription of Entamoeba histolytica Shaudinn, 1903 (Emended Walker, 1911) separating from it from Entamoeba dispar Brumpt, 1925. J Eukaryot Microbiol 40(3):340–4
Fotedar R, Stark D, Beebe N, et al (2007) Laboratory diagnostic techniques for Entamoeba species. Clin Microbiol Rev 20(3):511–32
Fotedar R, Stark D, Beebe N, et al, (2007) PCR detection of Entamoeba histolytica, Entamoeba dispar, and Entamoeba moshkovskii in stool samples from Sydney, Australia. J Clin Microbiol 45(3):1035–7
Gatti S, Swierczynski G, Robinson F, et al (2002) Amebic infections due to the Entamoeba histolytica-Entamoeba dispar complex: a study of the incidence in a remote rural area of Ecuador. Am J Trop Med Hyg 67(1):123–7
González-Ruiz A, Haque R, Aguirre A, et al (1994) Value of microscopy in the diagnosis of dysentery associated with invasive Entamoeba histolytica. J Clin Pathol 47(3):236–9
Haque R, Neville LM, Hahn P, Petri WA Jr (1995) Rapid diagnosis of Entamoeba infection by using Entamoeba and Entamoeba histolytica stool antigen detection kits. J Clin Microbiol 33 (10):2558–61
Haque R, Faruque AS, Hahn P, et al (1997) Entamoeba histolytica and Entamoeba dispar infection in children in Bangladesh. J Infect Dis 175(3):734–6
Korpe PS, Stott BR, Nazib F, et al (2012) Evaluation of a rapid point-of-care fecal antigen detection test for Entamoeba histolytica. Am J Trop Med Hyg 86(6):980–1
Tanyuksel M, Petri WA Jr (2003) Laboratory diagnosis of amebiasis. Clin Microbiol Rev 16(4):713–29
Verweij J, Oostvogel F, Brienen EA, et al (2003) Short communication: Prevalence of Entamoeba histolytica and Entamoeba dispar in northern Ghana. Trop Med Int Health 8(12):1153–6
WHO (1997) Amoebiasis. Wkly Epidemiol Rec 72(14):97–9
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Formenti, F., Perandin, F., Bonafini, S. et al. Evaluation of the new ImmunoCard STAT!® CGE test for the diagnosis of Amebiasis. Bull. Soc. Pathol. Exot. 108, 171–174 (2015). https://doi.org/10.1007/s13149-015-0434-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13149-015-0434-5